Market Overview
The Canada medical devices market has demonstrated substantial growth driven by increasing demand for advanced healthcare solutions. Based on recent historical assessments, the market size is projected to reach approximately USD ~ billion, supported by government-backed healthcare initiatives and the rising prevalence of chronic conditions that necessitate sophisticated medical devices. A surge in technological innovations, including AI-driven diagnostic tools and minimally invasive surgical equipment, is also contributing to market growth. Additionally, increasing investments from both private and public sectors aim to modernize the country’s healthcare infrastructure.
The market is dominated by key cities and regions such as Ontario, British Columbia, and Quebec, which house a high concentration of healthcare facilities, research institutions, and innovation hubs. These regions are at the forefront of medical device adoption due to their well-established healthcare systems, high healthcare spending, and access to state-of-the-art technologies. Furthermore, government policies that promote the integration of digital health technologies and advancements in healthcare infrastructure also play a crucial role in these areas’ market dominance.
Market Segmentation
By Product Type
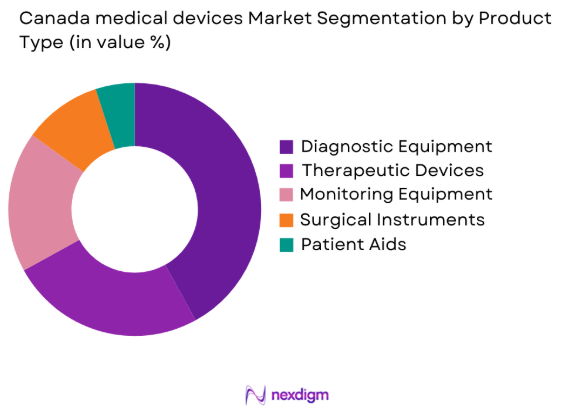
The Canadian medical devices market is segmented by product type into diagnostic equipment, therapeutic devices, monitoring equipment, surgical instruments, and patient aids. Recently, diagnostic equipment has dominated the market share, driven by the rising demand for early disease detection and ongoing advancements in imaging technologies, such as MRI and CT scanners. The growing number of chronic disease cases, including cancer, diabetes, and cardiovascular diseases, has further propelled the demand for these diagnostic solutions, supported by both government funding and private sector investments.
By Platform Type
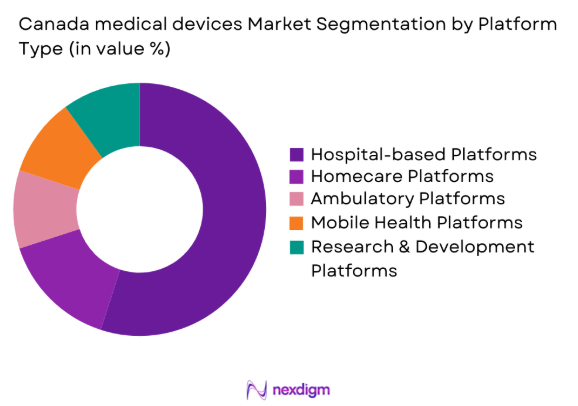
The Canada medical devices market is segmented by platform type into hospital-based platforms, homecare platforms, ambulatory platforms, mobile health platforms, and research & development platforms. Among these, hospital-based platforms have the largest market share due to the significant investments in infrastructure modernization and increasing demand for advanced medical devices in hospitals. The need for high-quality and accurate diagnostic equipment, therapeutic devices, and surgical tools in healthcare settings is crucial for delivering optimal patient care and improving healthcare outcomes.
Competitive Landscape
The Canada medical devices market is competitive, with numerous established global players and a few local companies vying for market share. Major players dominate the landscape, contributing to technological advancements, regulatory compliance, and product innovation. Consolidation is prominent as companies merge with or acquire smaller firms to enhance their market position and expand their technological capabilities. Key players, particularly those investing heavily in AI, robotics, and diagnostics, maintain strong influence over the industry’s direction.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Additional Parameter |
| Medtronic | 1949 | Dublin, Ireland | ~ | ~ | ~ | ~ | ~ |
| Stryker Corporation | 1941 | Kalamazoo, USA | ~ | ~ | ~ | ~ | ~ |
| GE Healthcare | 1892 | Chicago, USA | ~ | ~ | ~ | ~ | ~ |
| Philips Healthcare | 1891 | Amsterdam, Netherlands | ~ | ~ | ~ | ~ | ~ |
| Siemens Healthineers | 1847 | Erlangen, Germany | ~ | ~ | ~ | ~ | ~ |
Canada medical devices Market Analysis
Growth Drivers
Technological Advancements
The continuous development and integration of new technologies have been a significant growth driver in the Canadian medical devices market. Technological innovations, such as AI-powered diagnostic tools, robotic surgery systems, and minimally invasive treatment devices, are revolutionizing the healthcare sector. These advancements enhance the accuracy and speed of diagnosis, reduce recovery times, and improve overall patient outcomes. Additionally, the growing acceptance of telemedicine and remote patient monitoring, coupled with increasing healthcare system digitalization, supports the broader integration of innovative medical devices. As a result, healthcare providers are more inclined to adopt state-of-the-art technologies that promise higher efficiency and better patient care.
Government Investments in Healthcare Infrastructure
Government funding for the expansion and modernization of healthcare infrastructure is a significant growth driver for the Canadian medical devices market. Substantial investments have been made to improve hospital facilities, clinics, and diagnostic centers across the country. These investments allow healthcare providers to adopt advanced medical devices, thereby enhancing the overall quality of healthcare services. In addition, increasing government support for health IT systems, digital healthcare platforms, and telehealth initiatives plays a crucial role in driving market growth. This support not only improves healthcare delivery but also encourages private sector participation in the development of innovative medical solutions. As a result, the medical device market is benefiting from enhanced infrastructure and a more robust healthcare system.
Market Challenges
High Regulatory Compliance Costs
A major challenge for the Canadian medical devices market is the complex and expensive regulatory environment. Manufacturers must adhere to stringent regulations set by Health Canada to ensure product safety and efficacy. While these regulations protect public health, they can also delay product approval, increase development costs, and limit innovation. The regulatory requirements for labeling, clinical trials, and post-market surveillance demand significant resources, which can be particularly challenging for smaller companies with limited financial capacity. These regulatory burdens can slow market entry for new products and innovations, making it difficult for smaller players to compete with larger, more established companies that have the resources to manage compliance and approval processes effectively.
High Initial Capital Investment
The capital-intensive nature of medical device manufacturing presents a significant challenge in the market. Developing advanced technologies requires substantial investments in research and development, testing, production facilities, and skilled labor. For companies entering the market or expanding their product lines, these financial burdens can be overwhelming. Moreover, continuous investments in upgrading manufacturing facilities and maintaining compliance with industry standards add to the financial strain. Small and medium-sized enterprises (SMEs) often face difficulty securing the necessary capital to compete with larger, well-established players. This financial barrier can limit their ability to innovate, scale operations, and effectively address market demand, giving larger corporations a competitive edge in terms of resources and technological advancements.
Opportunities
Homecare Medical Devices
The growing trend of home healthcare services presents significant opportunities for the Canadian medical devices market. As the aging population increases, more individuals are opting for homecare services that allow them to receive medical care in the comfort of their homes. This shift has resulted in higher demand for home medical devices, such as blood glucose monitors, oxygen concentrators, and wearable heart rate monitors. The COVID-19 pandemic has further accelerated this trend, driving demand for devices that enable remote monitoring and management of health conditions. Companies that focus on developing and providing user-friendly, cost-effective homecare devices are well-positioned to capture this growing market segment.
Integration of AI in Medical Devices
The integration of artificial intelligence (AI) in medical devices offers substantial opportunities in Canada. AI enhances diagnostic accuracy, streamlines workflows, and optimizes healthcare delivery. AI-driven imaging systems and diagnostic tools enable early disease detection, which improves patient outcomes. Furthermore, AI-powered predictive analytics aid healthcare providers in making informed decisions, improving patient care and resource allocation. AI applications, such as automated analysis of medical imaging, can speed up diagnoses, reducing waiting times and improving efficiency. AI technology advances, Canadian companies that focus on integrating AI into medical devices will be well-positioned to lead the market, offering solutions that improve healthcare quality and drive better operational efficiency in medical facilities. This trend is expected to shape the future of healthcare in Canada.
Future Outlook
Over the next five years, the Canadian medical devices market is expected to continue its growth trajectory, fueled by advancements in technology, increased demand for homecare solutions, and ongoing government investments in healthcare infrastructure. The adoption of AI, telemedicine, and digital health platforms will play a pivotal role in shaping the market, creating new opportunities for innovation and market expansion. Regulatory support will continue to enhance the integration of these technologies, ensuring the safety and effectiveness of medical devices in the Canadian healthcare ecosystem.
Major Players
- Medtronic
- Stryker Corporation
- GE Healthcare
- Philips Healthcare
- Siemens Healthineers
- Johnson & Johnson
- Abbott Laboratories
- Boston Scientific
- Zimmer Biomet
- Baxter International
- B. Braun Melsungen
- Intuitive Surgical
- Smith & Nephew
- Terumo Corporation
- Hologic
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Hospitals and healthcare providers
- Medical device manufacturers
- Healthcare technology developers
- Private equity firms
- Insurance companies
- Pharmaceutical companies
Research Methodology
Step 1: Identification of Key Variables
This step involves identifying the key variables that influence the Canadian medical devices market, such as technological trends, regulatory frameworks, and healthcare infrastructure developments. These variables are crucial for accurately analyzing market dynamics.
Step 2: Market Analysis and Construction
Market analysis is performed through comprehensive data collection and segmentation, evaluating factors like product type, platform type, end-user segments, and key drivers and challenges. This helps in constructing a clear market structure.
Step 3: Hypothesis Validation and Expert Consultation
Market hypotheses are validated through expert consultations and industry insights to ensure that assumptions and models accurately reflect the current and future landscape of the medical devices market in Canada.
Step 4: Research Synthesis and Final Output
The final output synthesizes all data, analysis, and insights into a cohesive report that reflects market trends, growth opportunities, challenges, and competitive dynamics.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Increase in Chronic Diseases
Aging Population
Technological Advancements - Market Challenges
High Regulatory Barriers
Cost of Advanced Technology
Supply Chain Disruptions - Market Opportunities
Demand for Homecare Devices
Technological Integration in Healthcare
Expansion of Telemedicine Platforms - Trends
Rise in Minimally Invasive Procedures
Increase in Wearable Medical Devices
Shift Towards Patient-Centered Care - Government Regulations
- SWOT Analysis of Key Competitors
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Diagnostic Equipment
Therapeutic Equipment
Monitoring Equipment
Surgical Instruments
Patient Aids - By Platform Type (In Value%)
Hospital-based Platforms
Home Care Platforms
Ambulatory Platforms
Mobile Health Platforms
Research & Development Platforms - By Fitment Type (In Value%)
Fixed Systems
Portable Systems
Wearable Systems
Point-of-Care Systems
Modular Systems - By End User Segment (In Value%)
Hospitals & Clinics
Homecare Patients
Diagnostic Labs
Ambulatory Surgery Centers
Research Institutes - By Procurement Channel (In Value%)
Direct Procurement
Distributors
Online Procurement
B2B Marketplaces
Government Tendering
- Market Share Analysis
- Cross Comparison Parameters (System Type, Platform Type, Procurement Channel, End User Segment, Fitment Type, Technology Integration, Regulatory Compliance, Market Accessibility, Cost of Innovation, Product Customization)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Medtronic
Stryker Corporation
GE Healthcare
Philips Healthcare
Siemens Healthineers
Boston Scientific
Johnson & Johnson
Abbott Laboratories
Zimmer Biomet
Baxter International
B. Braun Melsungen
Intuitive Surgical
Smith & Nephew
Terumo Corporation
Hologic
- Growing Demand in Rural Areas
- Technological Adoption in Hospitals
- Surge in Homecare Applications
- Shift to Personalized Healthcare Solutions
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035