Market Overview
The France medical devices market is valued at approximately USD ~ billion based on a recent historical assessment. This growth is driven by an aging population, an increasing prevalence of chronic diseases, and technological advancements in medical equipment. The government’s healthcare investments and a growing demand for innovative treatments and medical procedures further support this market. Moreover, France’s strong healthcare system, complemented by its private and public healthcare sectors, ensures the continuous growth of the medical devices industry.
Paris, Lyon, and Marseille are dominant cities in France’s medical devices market due to their substantial healthcare infrastructure and established medical device manufacturing centers. Paris, as the capital, hosts the largest concentration of healthcare providers, hospitals, and medical research institutions. Lyon and Marseille are known for their contributions to medical technology, with advanced research in biotechnology and a growing number of medical device companies. The prominence of these cities, along with their established healthcare ecosystems, makes them key contributors to France’s medical devices sector.
Market Segmentation
By Product Type
The France medical devices market is segmented by product type into diagnostic devices, therapeutic devices, and monitoring devices. Recently, diagnostic devices have dominated the market share due to the increasing demand for early disease detection and monitoring. The high demand for diagnostic imaging technologies such as MRI machines, ultrasound devices, and X-ray systems is driving this trend. Hospitals, clinics, and diagnostic centers continue to invest in advanced diagnostic equipment to provide more accurate and timely results. Additionally, advancements in non-invasive diagnostic tools and personalized medicine have further fueled the growth of diagnostic devices in France.

By Application
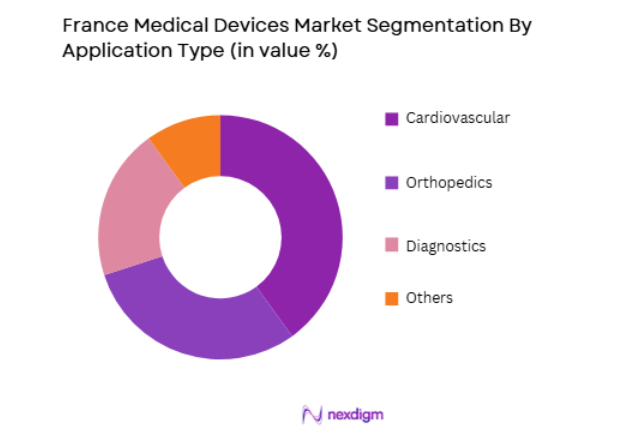
The France medical devices market is segmented by application into cardiovascular, orthopedics, diagnostics, and others. Recently, cardiovascular devices have dominated the market share due to the high prevalence of heart disease and related conditions in France. The aging population and increasing risk factors such as high blood pressure, smoking, and obesity have led to a surge in demand for cardiovascular devices such as pacemakers, stents, and heart valves. The need for these devices in both emergency and routine care settings continues to drive their dominance in the French market.
Competitive Landscape
The competitive landscape of France’s medical devices market is highly consolidated, with leading multinational companies holding significant market share. Major players in the medical device industry continue to expand their product portfolios and leverage technological innovations to maintain their competitive edge. The market also includes several domestic companies that focus on niche medical device segments, such as diagnostics and orthopedics. Government regulations ensure that all products meet strict quality and safety standards, which drives the reliability of the sector. The market is also characterized by partnerships between healthcare providers and medical device companies to enhance patient outcomes.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Additional Parameter |
| Medtronic | 1949 | Dublin, Ireland | ~ | ~ | ~ | ~ | ~ |
| Siemens Healthineers | 1847 | Erlangen, Germany | ~ | ~ | ~ | ~ | ~ |
| Philips Healthcare | 1891 | Amsterdam, Netherlands | ~ | ~ | ~ | ~ | ~ |
| Johnson & Johnson | 1886 | New Brunswick, USA | ~ | ~ | ~ | ~ | ~ |
| Stryker Corporation | 1941 | Kalamazoo, USA | ~ | ~ | ~ | ~ | ~ |
France Medical Devices Market Analysis
Growth Drivers
Aging Population and Rising Incidence of Chronic Diseases
One of the major growth drivers for France’s medical devices market is the aging population, which leads to a greater need for medical devices across various specialties. As the elderly population increases, the demand for healthcare services and related medical devices, such as those for cardiovascular care, diagnostics, and orthopedics, has surged. The rise in chronic diseases, particularly heart disease, diabetes, and arthritis, is contributing significantly to the demand for specialized medical devices. With an aging population more likely to require medical interventions, the market for diagnostic and therapeutic devices will continue to expand. Additionally, the prevalence of lifestyle diseases, including obesity and hypertension, further boosts the demand for medical devices, particularly in diagnostics and cardiovascular segments. France’s robust healthcare infrastructure, combined with increased government investment in healthcare reforms, ensures that medical devices will remain a crucial element in the country’s healthcare delivery system.
Technological Advancements and Innovation
Another major driver for the growth of France’s medical devices market is the continuous advancement in medical technologies. Innovations in diagnostic imaging, minimally invasive surgical instruments, and wearable medical devices have revolutionized healthcare delivery, allowing for more efficient and precise treatments. France’s medical device industry benefits from significant investments in research and development (R&D), which drive the introduction of new and improved devices. For example, the development of advanced MRI systems, robotic surgical tools, and remote monitoring devices has expanded the range of treatments available to patients. Furthermore, the integration of artificial intelligence (AI) and machine learning in medical devices is enhancing diagnostic accuracy and patient outcomes. With ongoing technological advancements and the integration of digital health solutions, the demand for innovative medical devices in France is expected to continue to rise.
Market Challenges
Regulatory Challenges and Compliance
One of the key challenges facing the French medical devices market is the complex regulatory environment and compliance requirements. Medical device manufacturers must adhere to strict regulations set by the European Medicines Agency (EMA) and national authorities to ensure that their products meet safety and performance standards. The regulatory processes for approval and certification of medical devices can be lengthy and costly, which can delay product entry into the market. Additionally, the regulatory landscape is constantly evolving, with new standards and regulations being introduced to address emerging risks and innovations in medical technologies. Manufacturers must continuously invest in meeting these regulatory demands, which can place a significant financial burden on smaller companies. The complexity of these regulatory requirements presents a barrier to entry for new players and can slow down innovation in the sector.
Rising Healthcare Costs and Budget Constraints
Rising healthcare costs, particularly in the public healthcare system, pose another challenge to the French medical devices market. France’s healthcare system is publicly funded, and with the increasing demand for medical devices driven by an aging population and the prevalence of chronic diseases, the costs associated with healthcare delivery are rising. Hospitals and clinics must balance their budgets while ensuring access to the latest medical technologies, often resulting in delays in acquiring new devices. The increasing cost of healthcare equipment, especially high-tech diagnostic and therapeutic devices, places further pressure on healthcare providers. Additionally, cost-consciousness among consumers and healthcare providers, as well as the strain on public healthcare budgets, limits the extent to which new devices can be adopted. Medical device manufacturers must navigate these financial constraints while maintaining affordability and accessibility for the healthcare system.
Opportunities
Expansion of Medical Tourism
The growth of medical tourism presents a significant opportunity for France’s medical devices market. France is already a leading destination for medical tourists, particularly for specialized procedures in fields such as cosmetic surgery, orthopedic treatments, and fertility services. The country’s world-class healthcare infrastructure, highly skilled medical professionals, and advanced medical technologies make it an attractive destination for patients from other countries. As the medical tourism industry continues to expand, the demand for high-quality medical devices, particularly in specialized healthcare services, is expected to grow. French medical device companies can capitalize on this trend by offering cutting-edge technologies to meet the needs of international patients. Additionally, the growing trend of cross-border healthcare provides opportunities for collaboration between French healthcare providers and medical device manufacturers to create tailored solutions for medical tourists.
Integration of Digital Health and Wearable Devices
The integration of digital health solutions and wearable medical devices is another opportunity for growth in France’s medical devices market. Digital health technologies, such as remote patient monitoring, telemedicine, and mobile health applications, are becoming increasingly important in managing chronic diseases and providing preventive care. The rise of wearable medical devices, such as fitness trackers, ECG monitors, and continuous glucose monitors, is driving the demand for more advanced and personalized healthcare solutions. These devices allow patients to monitor their health in real-time and provide healthcare professionals with valuable data to improve treatment plans. As France continues to invest in digital health infrastructure and emphasizes the use of data-driven healthcare, the market for digital health devices and wearables will likely experience significant growth. This shift toward personalized and remote healthcare solutions represents a major opportunity for medical device manufacturers to innovate and meet the evolving needs of both patients and healthcare providers.
Future Outlook
The future outlook for France’s medical devices market is promising, with continued growth driven by technological advancements, an aging population, and increasing healthcare demand. The market will benefit from the ongoing expansion of medical tourism and the increasing adoption of digital health solutions. The French government’s support for healthcare reforms, along with the growing emphasis on preventive care and chronic disease management, will further fuel the demand for innovative medical devices. With continued investment in research and development, France is expected to remain a key player in the global medical devices market, providing new opportunities for manufacturers and healthcare providers.
Major Players
- Medtronic
- Siemens Healthineers
- Philips Healthcare
- Johnson & Johnson
- Stryker Corporation
- GE Healthcare
- Boston Scientific
- Abbott Laboratories
- Zimmer Biomet
- B. Braun Melsungen
- Roche Diagnostics
- Cardinal Health
- Biotronik
- Baxter International
- Terumo Corporation
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Healthcare providers
- Medical device manufacturers
- Pharmaceutical companies
- Hospitals and clinics
- Medical research organizations
Research Methodology
Step 1: Identification of Key Variables
The research identifies key market variables such as healthcare demand, technological advancements, market challenges, and government policies that influence the growth of the medical devices market.
Step 2: Market Analysis and Construction
Market data is collected from credible sources, and analytical models are used to construct a comprehensive market forecast for the medical devices market in France.
Step 3: Hypothesis Validation and Expert Consultation
Hypotheses regarding market trends, growth drivers, and opportunities are validated through consultations with industry experts and key stakeholders in the medical devices sector.
Step 4: Research Synthesis and Final Output
The final report synthesizes all data and insights to provide an in-depth analysis of the France medical devices market, including growth drivers, challenges, and opportunities.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Growth Drivers
Technological Advancements in Medical Devices
Aging Population Driving Healthcare Demand
Increased Adoption of Minimally Invasive Procedures - Market Challenges
Regulatory and Compliance Barriers
High Equipment Costs
Lack of Skilled Technicians in Remote Areas - Market Opportunities
Growing Demand for Home Healthcare Devices
Rising Popularity of Wearable Medical Devices
Advances in Diagnostic Technologies - Trends
Increased Focus on Personalized Medicine
Integration of AI in Medical Devices - Government Regulations
- SWOT Analysis
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Diagnostic Devices
Therapeutic Devices
Surgical Instruments
Monitoring Devices
Imaging Equipment - By Platform Type (In Value%)
Hospital-Based Platforms
Point-of-Care Platforms
Mobile Platforms
Diagnostic Lab Platforms - By Fitment Type (In Value%)
Fixed Devices
Portable Devices
Wearable Devices
Disposable Devices - By End User Segment (In Value%)
Hospitals
Clinics
Research Institutes
Home Care Providers
- Market Share Analysis
- Cross Comparison Parameters (System Type, Platform Type, Procurement Channel, End User Segment, Fitment Type, Technological Integration, Service Availability, Market Reach, Compliance with Standards, Data Security)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Siemens Healthineers
GE Healthcare
Medtronic
Philips Healthcare
Stryker Corporation
Johnson & Johnson
Abbott Laboratories
Becton Dickinson
Thermo Fisher Scientific
Zimmer Biomet
Roche Diagnostics
Boston Scientific
Smith & Nephew
Fujifilm Healthcare
Stryker Corporation
- Hospitals Expanding Medical Device Adoption
- Research Institutes Innovating with New Technologies
- Clinics Integrating Advanced Diagnostic Equipment
- Home Care Providers Using Remote Monitoring Devices
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035