Market Overview
The KSA Biosimilar Insulin market is expected to see substantial growth, driven by the increasing prevalence of diabetes and the growing demand for cost-effective insulin alternatives. Based on a recent historical assessment, the market size is forecasted to reach USD ~ billion, driven by rising healthcare needs and government support for biosimilar products. The expansion of the biosimilar insulin segment is also supported by the increasing pressure to reduce healthcare expenditures in Saudi Arabia, with biosimilars offering a cost-effective solution to branded insulins.
Saudi Arabia, particularly in cities like Riyadh, Jeddah, and Dammam, leads the demand for biosimilar insulin due to its advanced healthcare infrastructure and the growing number of diabetes cases. These cities are central to the government’s push for more affordable healthcare solutions, with healthcare reforms making biosimilars more accessible to the general population. The ongoing initiatives to tackle chronic diseases like diabetes through affordable therapies have also accelerated the adoption of biosimilar insulin across the country.
Market Segmentation
By Product Type
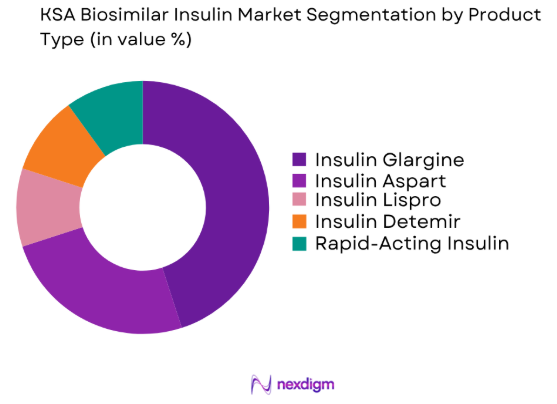
The KSA Biosimilar Insulin market is segmented by product type into Insulin Glargine, Insulin Aspart, Insulin Lispro, Insulin Detemir, and Rapid-Acting Insulin. Insulin Glargine holds a dominant market share due to its use as a long-acting insulin treatment that provides steady blood glucose control, which is a key requirement for managing diabetes effectively. The demand for cost-effective alternatives to branded Insulin Glargine has been bolstered by the growing number of diabetes patients and government incentives promoting affordable biosimilars.
By Platform Type
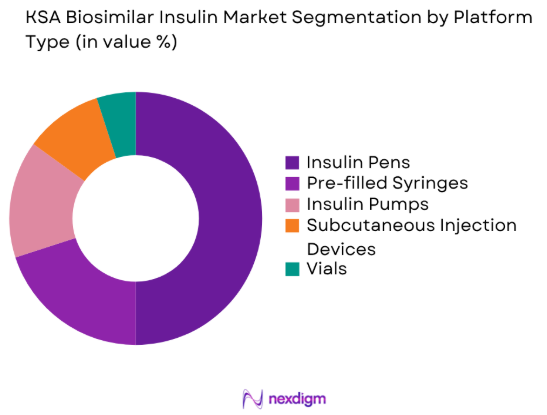
The KSA Biosimilar Insulin market is also segmented by platform type into Insulin Pens, Pre-filled Syringes, Insulin Pumps, Subcutaneous Injection Devices, and Vials. Insulin Pens dominate the market due to their convenience and ease of use for both healthcare professionals and patients. These pens are increasingly popular as they enable patients to manage their insulin dosage effectively and with greater ease, particularly in homecare settings where self-management is a priority. The widespread adoption of insulin pens has further driven their dominance in the market.
Competitive Landscape
The KSA Biosimilar Insulin market is characterized by strong competition among both global pharmaceutical companies and local players. Leading multinational firms, alongside emerging local manufacturers, are working to expand their production capabilities to meet the increasing demand for biosimilars. Government support for biosimilar insulin adoption further intensifies the competition, encouraging both established and new players to innovate and capture a larger share of the market. The regulatory environment continues to evolve in favor of biosimilar products, giving major players the opportunity to scale their market presence.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Market-Specific Parameter |
| Sandoz | 2002 | Switzerland | ~ | ~ | ~ | ~ | ~ |
| Boehringer Ingelheim | 1885 | Germany | ~ | ~ | ~ | ~ | ~ |
| Mylan | 1961 | USA | ~ | ~ | ~ | ~ | ~ |
| Sanofi | 2004 | France | ~ | ~ | ~ | ~ | ~ |
| Novo Nordisk | 1923 | Denmark | ~ | ~ | ~ | ~ | ~ |
KSA Biosimilar Insulin Market Analysis
Growth Drivers
Government support for biosimilars
One of the key growth drivers for the KSA Biosimilar Insulin market is the strong support from the Saudi Arabian government for the adoption of biosimilar products. The government’s healthcare policies aim to reduce the overall cost of treatment for chronic conditions like diabetes, and biosimilars are seen as a cost-effective alternative to branded products. By implementing reforms, including price reductions, subsidies, and facilitating access to affordable medicines, the government has played a crucial role in expanding the use of biosimilars. This support has encouraged healthcare providers to increasingly adopt biosimilars, especially in government-funded healthcare institutions, ensuring greater accessibility for patients across the country.
Technological advancements in insulin delivery systems
Another significant growth driver is the continuous innovation in insulin delivery technologies. Developments such as smart insulin pens, insulin pumps, and automated insulin delivery systems have enhanced the accuracy and ease of insulin administration for patients. These technological advancements are driving the demand for biosimilar insulin, as patients seek more convenient and effective ways to manage diabetes. The rise in personalized diabetes care, which these technologies support, further accelerates the need for biosimilars. With their cost-effectiveness and efficiency, biosimilars are positioned to meet the growing demand for reliable and accessible diabetes treatments, offering patients more options for managing their condition effectively.
Market Challenges
High production costs of biosimilars
Despite the cost-saving benefits for patients, biosimilar production remains expensive due to high development, testing, and manufacturing costs. Meeting stringent regulatory requirements adds further complexity and expense, which acts as a barrier for many companies. This limits the number of players in the market, reducing competition and potentially leading to higher prices for consumers. Smaller biosimilar manufacturers, in particular, face difficulties scaling up production, further hindering their ability to offer affordable insulin options. The high initial investment needed for production facilities and the ongoing manufacturing costs create significant challenges for the broader adoption of biosimilars. As a result, the market faces slower growth and limited availability of low-cost insulin alternatives.
Regulatory challenges and approval delays
The regulatory environment in Saudi Arabia, although supportive of biosimilars, presents challenges with complex and lengthy approval processes for new products. These regulations are designed to ensure the safety and effectiveness of biosimilars, but they can delay the introduction of new treatments to the market. As a result, patients may experience delays in accessing the latest biosimilar options, while manufacturers face slow market penetration. Additionally, the cost and complexity of navigating the regulatory approval process can discourage smaller companies from entering the market, limiting competition and the diversity of available biosimilars. This situation slows the overall growth of the biosimilar insulin market and affects its accessibility to consumers.
Opportunities
Expansion into emerging markets
Saudi Arabia’s biosimilar insulin market offers significant opportunities for expansion into other emerging markets within the MENA region. Neighboring countries face similar challenges in managing diabetes, creating a growing demand for affordable insulin alternatives. Saudi Arabia’s market, supported by favorable government regulations and increasing patient needs, can serve as a model for surrounding nations. Companies manufacturing biosimilars in Saudi Arabia have the potential to expand into these markets, establishing a strong presence and capturing new revenue streams. This regional expansion can increase the availability of biosimilar insulin products, contributing to improved diabetes care across the MENA region and positioning Saudi Arabia as a leader in affordable insulin solutions.
Increasing use of homecare solutions
The increasing adoption of homecare solutions for diabetes management creates a significant opportunity for the KSA Biosimilar Insulin market. As more patients prefer to manage their diabetes independently, the demand for user-friendly insulin delivery systems, such as insulin pens and pumps, is growing. These devices offer patients flexibility and convenience for managing their condition at home. The rise of home-based care solutions, fueled by advancements in healthcare technology, provides biosimilar insulin manufacturers with a chance to tap into this expanding market. By addressing the needs of patients seeking greater autonomy in diabetes management, manufacturers can meet growing demand while contributing to improved patient outcomes.
Future Outlook
The KSA Biosimilar Insulin market is poised for continued growth, with increasing demand driven by government support, technological advancements in insulin delivery systems, and the rise in diabetes cases. The next five years will see further regulatory support for biosimilars, enhancing the market environment. The growing emphasis on affordable healthcare solutions, combined with innovations in treatment delivery, will ensure that biosimilar insulin continues to play a central role in diabetes care, positioning Saudi Arabia as a leader in the MENA region for biosimilar insulin adoption.
Major Players
- Sandoz
- Boehringer Ingelheim
- Mylan
- Sanofi
- Novo Nordisk
- Eli Lilly
- Baxter
- Fresenius Kabi
- Teva Pharmaceuticals
- Wockhardt
- GSK
- Bristol-Myers Squibb
- Amgen
- Pfizer
- Roche
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Healthcare providers and institutions
- Pharmaceutical distributors
- Insulin delivery system manufacturers
- Biosimilar producers
- Retail pharmacy chains
- Private healthcare investors
Research Methodology
Step 1: Identification of Key Variables
Identification of key market variables that affect the demand for biosimilars, such as product types, platform types, and procurement channels.
Step 2: Market Analysis and Construction
Analysis of market trends, consumer behavior, and regulatory frameworks to build a reliable market model.
Step 3: Hypothesis Validation and Expert Consultation
Consultation with industry experts and stakeholders to validate market assumptions and projections.
Step 4: Research Synthesis and Final Output
Consolidation of collected data and insights into a comprehensive report, providing actionable recommendations for stakeholders.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Growing prevalence of diabetes
Government initiatives to reduce healthcare costs
Increasing awareness of biosimilars among healthcare professionals - Market Challenges
High initial investment for biosimilar production
Regulatory hurdles in product approval
Resistance from branded insulin manufacturers - Market Opportunities
Expansion of biosimilar market in emerging markets
Technological advancements in insulin delivery systems
Strategic collaborations between biosimilar manufacturers and healthcare providers - Trends
Shift toward personalized medicine in diabetes care
Increased investment in diabetes management technologies
Rising adoption of self-administration insulin devices - Government Regulations
- SWOT Analysis of Key Competitors
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Insulin Glargine
Insulin Aspart
Insulin Lispro
Insulin Detemir
Rapid-Acting Insulin - By Platform Type (In Value%)
Subcutaneous Injection Devices
Pre-filled Syringes
Insulin Pens
Vials
Insulin Pumps - By Fitment Type (In Value%)
Patient-Specific Solutions
Clinician-Administered Solutions
Automated Delivery Systems
Disposable Solutions
Re-usable Solutions - By End User Segment (In Value%)
Hospitals
Clinics
Home Care Settings
Pharmacies
Specialty Diabetes Centers - By Procurement Channel (In Value%)
Direct Procurement
Government Tenders
Private Sector Procurement
Hospital Networks
Pharmaceutical Distributors
- Market Share Analysis
- Cross Comparison Parameters (Product Type, Platform Type, End User Segment, Procurement Channel, Fitment Type, Delivery Mechanism, Regulatory Compliance, Market Penetration)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Sandoz
Boehringer Ingelheim
Mylan
Sanofi
Novo Nordisk
Eli Lilly
Baxter
Fresenius Kabi
Teva Pharmaceuticals
Wockhardt
GSK
Bristol-Myers Squibb
Amgen
Pfizer
Roche
- Increasing demand from homecare settings
- Growth in clinic-based treatments
- Rising diabetes management programs in hospitals
- Pharmacy-driven insulin distribution model
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035