Market Overview
The KSA Biosimilar Recombinant Proteins Market is valued at approximately USD ~ billion based on a recent historical assessment, driven by the increasing demand for affordable biologics in the Kingdom of Saudi Arabia. The growth of this market is fueled by the rising prevalence of chronic diseases, an expanding healthcare infrastructure, and government incentives to boost the availability of biosimilars. Additionally, advancements in biotechnology and recombinant protein production technologies are significantly contributing to the market’s expansion.
The Saudi Arabian market for biosimilar recombinant proteins is concentrated in major cities like Riyadh, Jeddah, and Dammam. These regions benefit from well-established healthcare infrastructure and a growing patient base due to urbanization and increased healthcare investments. Government initiatives, including Vision 2030, which prioritizes the local manufacturing of biopharmaceuticals, further solidify these cities’ dominance in the sector. The Kingdom’s focus on improving healthcare accessibility and reducing costs for patients has played a key role in shaping this market.
Market Segmentation
By Product Type
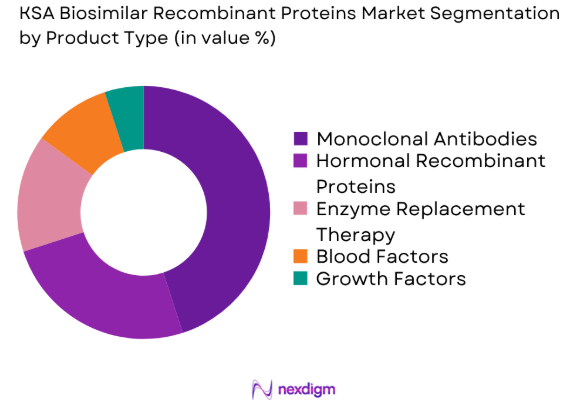
The KSA Biosimilar Recombinant Proteins market is segmented by product type into monoclonal antibodies, hormonal recombinant proteins, enzyme replacement therapy, blood factors, and growth factors. Recently, monoclonal antibodies have gained a dominant market share due to their high efficacy in treating chronic conditions such as cancer and autoimmune disorders. This sub-segment benefits from the increasing focus on immunotherapy, extensive research and development investments, and increasing patient demand for innovative biologic treatments.
By Platform Type
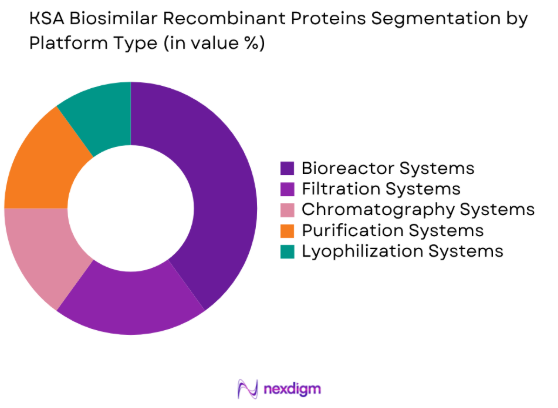
The KSA Biosimilar Recombinant Proteins market is also segmented by platform type into bioreactor systems, filtration systems, chromatography systems, purification systems, and lyophilization systems. Recently, bioreactor systems have become the dominant sub-segment, largely due to their crucial role in large-scale recombinant protein production. The demand for bioreactor systems is supported by the growth of the biotechnology industry and the increased adoption of biosimilars, which require efficient and scalable production platforms to meet market needs.
Competitive Landscape
The competitive landscape of the KSA Biosimilar Recombinant Proteins Market is dynamic, with a mix of global players and emerging local producers. The market is seeing consolidation, as large multinational pharmaceutical companies partner with regional players to strengthen their position. The influence of major players, combined with ongoing regulatory reforms, has led to increased competition and a focus on product differentiation, driving innovation in the sector.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Additional Parameter |
| Sandoz | 2009 | Holzkirchen, Germany | ~ | ~ | ~ | ~ | ~ |
| Amgen | 1980 | Thousand Oaks, USA | ~ | ~ | ~ | ~ | ~ |
| Celltrion Healthcare | 2002 | Incheon, South Korea | ~ | ~ | ~ | ~ | ~ |
| Mylan N.V. | 1961 | Canonsburg, USA | ~ | ~ | ~ | ~ | ~ |
| Pfizer | 1849 | New York, USA | ~ | ~ | ~ | ~ | ~ |
KSA Biosimilar Recombinant Proteins Market Analysis
Growth Drivers
Increased Prevalence of Chronic Diseases
The rising prevalence of chronic diseases such as diabetes, cancer, and autoimmune disorders in Saudi Arabia has significantly increased the demand for biologics. These conditions require long-term management, often relying on biologic treatments like biosimilars. As these diseases become more widespread, particularly among the aging population, the need for affordable and effective therapeutic options has grown, driving the recombinant protein market. Saudi Arabia’s healthcare initiatives, focusing on improving accessibility and reducing treatment costs, further boost the adoption of biosimilars. Additionally, advancements in biotechnology are enhancing the availability and affordability of biologic drugs, supporting market expansion and providing better treatment options for patients managing chronic conditions.
Government Support for Biosimilar Development
As part of its Vision 2030 initiative, the Saudi government has focused on developing the biosimilar market to reduce healthcare costs and promote local biologics manufacturing. The government has introduced regulatory incentives, allocated funding for research and development, and created a supportive policy environment to boost biosimilar production. These efforts aim to improve the affordability and accessibility of biosimilars, particularly recombinant proteins. By encouraging local manufacturing and innovation, the government is strengthening the market’s growth potential. With these measures in place, Saudi Arabia is positioned to meet the rising demand for cost-effective biologic treatments, contributing to long-term market expansion and improving patient access to essential therapies.
Market Challenges
Regulatory Hurdles in Biosimilar Approvals
A major challenge in the KSA Biosimilar Recombinant Proteins Market is the complex regulatory approval process. Although the government is working on improving biosimilar regulations, navigating this framework remains time-consuming and costly for companies. This delays the introduction of new biosimilar products and creates uncertainty for investors. As the regulatory environment evolves, companies must adapt to new requirements, demonstrating the safety and efficacy of their products. This ongoing adjustment process adds significant hurdles, potentially slowing market growth. The slow pace of regulatory changes can impede innovation and prevent timely market entry, limiting the overall expansion of the biosimilars market in Saudi Arabia.
High Production Costs
The production of biosimilars and recombinant proteins requires substantial upfront investments in both infrastructure and technology. For smaller and regional manufacturers, the high costs associated with recombinant protein synthesis create a significant barrier to entry. Specialized equipment and a skilled workforce are essential, further increasing production expenses. These factors make it difficult for companies to maintain profitability while offering competitive prices. As a result, many players struggle to scale their operations effectively, limiting their ability to compete in the market. The financial strain associated with these high production costs can slow innovation and hinder market expansion, especially for emerging or smaller biosimilar manufacturers in Saudi Arabia.
Opportunities
Rising Demand for Personalized Medicine
Personalized medicine, which customizes treatments based on genetic and molecular data, is gaining momentum in Saudi Arabia. The increasing focus on precision medicine is opening opportunities for biosimilar recombinant proteins designed to target specific diseases or conditions. With advancements in genomics and biotechnology, companies can use recombinant proteins to develop more effective and targeted therapies. This growing interest in personalized treatments is expected to drive the demand for biosimilars, offering significant growth potential for manufacturers. As the healthcare system shifts toward more tailored approaches, biosimilar recombinant proteins are well-positioned to meet the needs of patients requiring specific and advanced therapeutic solutions.
Increasing Investment in Biotechnology
There is a noticeable shift toward increased investment in biotechnology in Saudi Arabia, driven by government incentives and the potential for high returns in the biopharmaceutical industry. This trend is creating opportunities for companies to secure funding for the development of biosimilar recombinant proteins. Additionally, the rise of biotechnology startups and growing collaborations between international and local firms point to a promising future for the biosimilars market in the Kingdom. Investors are increasingly recognizing the untapped potential in biosimilars, positioning the sector as a key area for future growth. With continued investment, the market is expected to expand, fostering innovation and boosting the availability of affordable biologics.
Future Outlook
The future outlook for the KSA Biosimilar Recombinant Proteins Market is promising, with sustained growth expected over the next five years. Technological advancements in biomanufacturing, coupled with increasing government support for local production, will foster a competitive environment in the biosimilars sector. The demand for cost-effective therapeutic options, combined with the rising prevalence of chronic diseases, will continue to drive market expansion. Furthermore, the Kingdom’s regulatory reforms will likely make it easier for new players to enter the market, enhancing competition and innovation.
Major Players
- Sandoz
- Amgen
- Celltrion Healthcare
- Mylan N.V.
- Pfizer
- Biocon
- Roche
- Merck
- Novartis
- Teva Pharmaceuticals
- Samsung Biologics
- Eli Lilly
- AbbVie
- Regeneron Pharmaceuticals
- GSK
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Pharmaceutical manufacturers
- Biotech companies
- Healthcare providers
- Hospitals and clinics
- Contract research organizations
- Research institutions
Research Methodology
Step 1: Identification of Key Variables
The key variables impacting the market, including product types, technological advancements, and regulatory frameworks, were identified through extensive secondary research.
Step 2: Market Analysis and Construction
Market sizing and segmentation were conducted using reliable industry data from reputable sources, including government reports and company filings.
Step 3: Hypothesis Validation and Expert Consultation
The initial findings were validated through expert interviews and discussions with market leaders and industry specialists.
Step 4: Research Synthesis and Final Output
The synthesized data was analyzed to draw insights and produce a comprehensive report on the KSA Biosimilar Recombinant Proteins Market.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Increase in Chronic Disease Prevalence
Government Support for Biosimilar Development
Technological Advancements in Recombinant Protein Production - Market Challenges
Regulatory Hurdles in Biosimilar Approvals
High Production Costs
Lack of Market Awareness - Market Opportunities
Growth in Healthcare Infrastructure
Increasing Demand for Affordable Biopharmaceuticals
Emerging Markets Adoption - Trends
Personalized Medicine Integration
Shift Toward Sustainable Manufacturing
Increased Investment in Biotech Innovation - Government Regulations
- SWOT Analysis of Key Competitors
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Monoclonal Antibodies
Hormonal Recombinant Proteins
Enzyme Replacement Therapy
Blood Factors
Growth Factors - By Platform Type (In Value%)
Bioreactor Systems
Filtration Systems
Chromatography Systems
Purification Systems
Lyophilization Systems - By Fitment Type (In Value%)
On-Premise Solutions
Cloud-Based Solutions
Hybrid Solutions
Modular Solutions
Integrated Solutions - By End User Segment (In Value%)
Pharmaceutical Companies
Biotech Companies
Hospitals & Healthcare Providers
Contract Research Organizations
Research & Academic Institutions - By Procurement Channel (In Value%)
Direct Procurement
Third-Party Distributors
Online Platforms
B2B Marketplaces
Private Sector Procurement
- Market Share Analysis
- Cross Comparison Parameters (System Type, Platform Type, Procurement Channel, End User Segment, Fitment Type, Production Technology, Regulatory Framework, Market Adoption Rate, Pricing Models, Geographical Reach)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Sandoz
Amgen
Celltrion Healthcare
Mylan N.V.
Bayer AG
Roche
Pfizer
Samsung Biologics
Eli Lilly and Co.
Merck & Co.
AbbVie
Biogen
Teva Pharmaceuticals
UCB Pharma
Abbott Laboratories
- Pharmaceutical Companies’ Expanding R&D Focus
- Hospitals’ Role in Increasing Demand for Cost-effective Treatments
- Contract Research Organizations’ Growing Influence in Biosimilar Production
- Research Institutions’ Contribution to Market Innovation
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035