Market Overview
The KSA Biosimilar Vaccines market size for biosimilar vaccines is poised to experience significant growth, driven by rising demand for affordable and effective vaccine alternatives. Based on a recent historical assessment, the market size for biosimilar vaccines in the KSA region has reached USD ~ billion, with growth propelled by increased government investments in healthcare infrastructure and initiatives to expand vaccination coverage. Technological advancements in vaccine production, along with a favorable regulatory environment, have further contributed to this growth trajectory.
The Kingdom of Saudi Arabia has emerged as a dominant player in the biosimilar vaccine market, fueled by its strategic healthcare investments and a rapidly growing population. Dominant cities such as Riyadh and Jeddah are central to vaccine distribution, owing to their role as key healthcare hubs with robust medical infrastructure. Additionally, Saudi Arabia’s Vision 2030, which focuses on improving healthcare access and quality, supports the market’s future growth by creating a conducive environment for both domestic and international pharmaceutical players.
Market Segmentation
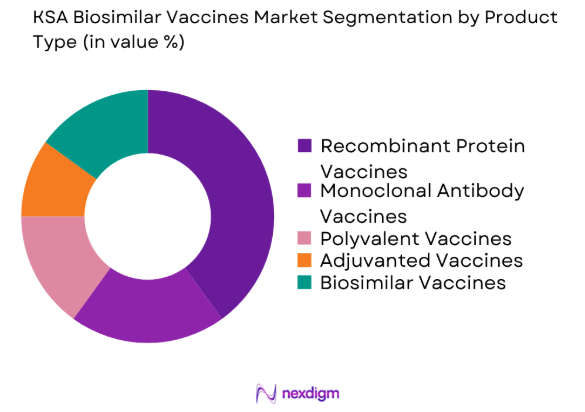
By Product Type
The KSA Biosimilar Vaccines market is segmented by product type into recombinant protein vaccines, monoclonal antibody vaccines, polyvalent vaccines, adjuvanted vaccines, and biosimilar vaccines. Recently, recombinant protein vaccines have dominated the market share, largely due to their established safety profiles, widespread applications in infectious disease prevention, and regulatory acceptance. Their proven effectiveness and significant clinical data availability contribute to their leadership in the market, especially in key therapeutic areas such as hepatitis, flu, and HPV.
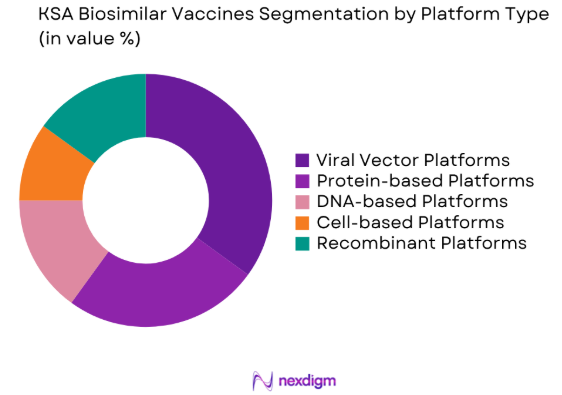
By Platform Type
The KSA Biosimilar Vaccines market is also segmented by platform type into viral vector platforms, protein-based platforms, DNA-based platforms, cell-based platforms, and recombinant platforms. Recently, viral vector platforms have gained a dominant share due to their ability to efficiently deliver genes that code for vaccine antigens, thus facilitating the production of highly effective vaccines. The growing use of viral vectors in COVID-19 vaccines and the increasing number of vaccines being developed using this platform have positioned it as a leader in the segment.
Competitive Landscape
The competitive landscape of the biosimilar vaccines market is marked by increasing consolidation, with major players leveraging their extensive research and manufacturing capabilities to capture a larger share of the market. The presence of both established pharmaceutical giants and specialized biosimilar manufacturers has intensified competition, resulting in rapid advancements in vaccine development and delivery technologies. As a result, market players are focusing on strategic collaborations and acquisitions to expand their product portfolios and gain a competitive edge.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD) | Market-specific Parameter |
| Sandoz | 2006 | Holzkirchen, Germany | ~ | ~ | ~ | ~ | ~ |
| Biocon | 1978 | Bangalore, India | ~ | ~ | ~ | ~ | ~ |
| Mylan | 1961 | Canonsburg, USA | ~ | ~ | ~ | ~ | ~ |
| Pfizer | 1849 | New York, USA | ~ | ~ | ~ | ~ | ~ |
| Merck & Co | 1891 | Kenilworth, USA | ~ | ~ | ~ | ~ | ~ |
KSA Biosimilar Vaccines Market Analysis
Growth Drivers
Increasing Government Investment in Healthcare Infrastructure
The Saudi government’s growing support to enhance healthcare access is a significant driver for the biosimilar vaccines market. The Vision 2030 initiative focuses on improving healthcare by increasing access to affordable medical services and expanding vaccination coverage. This includes substantial investments in healthcare infrastructure, research, and initiatives aimed at lowering financial barriers to healthcare. Government-backed policies and programs are fostering the adoption of biosimilar vaccines by healthcare providers, hospitals, and clinics, making vaccines more accessible and affordable. These initiatives are playing a crucial role in driving the growth of the biosimilar vaccines market in Saudi Arabia, ensuring better public health outcomes.
Rising Prevalence of Infectious Diseases
The increasing prevalence of infectious diseases in Saudi Arabia, such as influenza, hepatitis, and human papillomavirus (HPV), has led to a stronger emphasis on vaccination programs. This trend has boosted the demand for biosimilar vaccines as a more affordable alternative to branded vaccines. With growing awareness of preventive healthcare, there is a significant push for cost-effective vaccination solutions to curb the spread of infectious diseases. Public health campaigns and vaccination drives are central to expanding vaccine access, further driving the adoption of biosimilar vaccines. This increased focus on vaccination and affordability is accelerating the growth of the biosimilar vaccines market in the region.
Market Challenges
High Production Costs of Biosimilars
A major challenge in the biosimilar vaccines market is the high production cost associated with developing and manufacturing these vaccines. Although biosimilars are typically more affordable than their reference counterparts, the complexity of their production process and the stringent regulatory requirements make them expensive to produce. Manufacturers struggle to balance affordable pricing with maintaining high-quality standards. The substantial initial costs, including clinical trials, regulatory approvals, and the required infrastructure, present significant obstacles. These high production costs hinder the widespread adoption of biosimilar vaccines, particularly in markets with limited healthcare funding or where cost efficiency is a critical factor in vaccine deployment.
Regulatory Hurdles for Biosimilar Approval
A significant challenge in the biosimilar vaccines market is the stringent regulatory framework for approval. Agencies like the Saudi Food and Drug Authority (SFDA) demand extensive clinical trial data to prove the safety and efficacy of biosimilar vaccines. The approval process is often lengthy and complex, leading to delays in bringing new vaccines to market. This regulatory burden makes the entry of new players into the market challenging, as it requires substantial time, effort, and financial investment. The cost and time required to navigate these regulatory requirements can be particularly difficult for new entrants, further limiting the market’s growth and accessibility.
Opportunities
Expansion in Emerging Market Access
A major opportunity in the biosimilar vaccines market lies in expanding access to vaccines in emerging markets. Developing countries face significant challenges in providing affordable healthcare, driving the growing demand for cost-effective biosimilars. By expanding access to these vaccines in regions with high disease burdens, such as the Middle East, Asia, and Africa, biosimilar manufacturers can tap into a substantial growth opportunity. In these regions, improvements in healthcare infrastructure, alongside government-led initiatives aimed at increasing vaccination coverage, are creating a favorable environment for biosimilars. The push for affordable and accessible healthcare solutions, supported by both public and private sector efforts, presents a promising avenue for market growth and further adoption of biosimilar vaccines.
Technological Advancements in Vaccine Production
The adoption of new vaccine production technologies, including gene-editing techniques and AI-driven vaccine development, offers significant growth opportunities for the biosimilar vaccines market. These innovations enhance production efficiency, reduce manufacturing costs, and accelerate development timelines. As a result, manufacturers can lower production expenses, improve vaccine availability, and respond more swiftly to emerging diseases. With the continuous advancement of production technologies, the biosimilar vaccine market is poised for expansion. These technological advancements not only support cost-effective production but also enable the development of more diverse vaccine options, addressing various infectious diseases. The ongoing evolution of these technologies is expected to unlock further opportunities for the widespread adoption of biosimilar vaccines globally.
Future Outlook
The biosimilar vaccines market is expected to continue its upward trajectory over the next five years, driven by technological innovations, regulatory support, and growing demand for affordable vaccines. The ongoing development of new biosimilars, particularly in the oncology and infectious disease segments, along with increasing investment in healthcare infrastructure, will contribute to market growth. Additionally, favorable government policies promoting the use of biosimilars and the expansion of healthcare access in emerging markets will play a key role in shaping the market’s future.
Major Players
- Sandoz
- Biocon
- Mylan
- Pfizer
- Merck & Co
- Amgen
- Johnson & Johnson
- Sanofi
- AbbVie
- Roche
- BMS
- GSK
- Eli Lilly
- Bayer
- AstraZeneca
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Pharmaceutical companies
- Vaccine manufacturers
- Healthcare providers
- Hospitals
- Research institutions
- Public health organizations
Research Methodology
Step 1: Identification of Key Variables
Identification of key market drivers, challenges, opportunities, and growth potential in the biosimilar vaccines market, followed by comprehensive market research.
Step 2: Market Analysis and Construction
Detailed analysis of historical data and trends to build a robust market model, including segmentation and forecasting.
Step 3: Hypothesis Validation and Expert Consultation
Engagement with industry experts, stakeholders, and key players to validate market insights and refine hypotheses.
Step 4: Research Synthesis and Final Output
Synthesizing findings from primary and secondary research, along with expert insights, to produce a comprehensive report with actionable recommendations.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Increasing Government Investment in Vaccination Programs
Rising Prevalence of Infectious Diseases
Growing Adoption of Biosimilars in Healthcare - Market Challenges
High Production Costs of Biosimilars
Regulatory Barriers for Biosimilar Approval
Limited Awareness Among Healthcare Providers - Market Opportunities
Expansion in Emerging Market Access
Advancements in Vaccine Development Technology
Strategic Partnerships Between Global Pharma Players - Trends
Rise in Demand for Preventive Healthcare Solutions
Technological Innovation in Vaccine Development
Government Initiatives to Reduce Healthcare Costs - Government Regulations
- SWOT Analysis of Key Competitors
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Biosimilar Vaccines
Recombinant Protein Vaccines
Monoclonal Antibody Vaccines
Polyvalent Vaccines
Adjuvanted Vaccines - By Platform Type (In Value%)
Viral Vector Platforms
Protein-based Platforms
DNA-based Platforms
Cell-based Platforms
Recombinant Platforms - By Fitment Type (In Value%)
Single-dose Vaccines
Multi-dose Vaccines
Pre-filled Syringes
Vial-based Vaccines
Auto-injector Vaccines - By End User Segment (In Value%)
Hospitals
Healthcare Providers
Pharmacies
Government Health Programs
Private Sector Healthcare - By Procurement Channel (In Value%)
Direct Procurement
Government Tenders
Pharmaceutical Distributors
Healthcare Institutions
Online Procurement
- Market Share Analysis
- Cross Comparison Parameters (System Type, Platform Type, Procurement Channel, End User Segment, Fitment Type, Market Reach, Vaccine Efficacy, Regulatory Approval Process, Cost of Production, Distribution Channels)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Sandoz
Biocon
Mylan
Pfizer
Novartis
Amgen
Merck & Co
Sanofi
Bayer
AbbVie
Roche
Johnson & Johnson
Eli Lilly
GlaxoSmithKline
Boehringer Ingelheim
- Government Health Programs Increasing Vaccine Distribution
- Growing Demand from Private Sector Healthcare Providers
- Hospitals Expanding Biosimilar Vaccine Offerings
- Pharmacies Offering Broader Access to Vaccines
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035