Market Overview
The KSA Continuous Glucose Monitoring Systems market reached a value of USD ~ billion based on a recent historical assessment, supported by rising diabetes prevalence affecting more than ~ million adults according to international diabetes health databases. Market expansion is primarily driven by increased adoption of wearable glucose monitoring devices, improved reimbursement frameworks in private healthcare, and the expansion of digital health programs. Growth is further supported by hospital investments in remote patient monitoring infrastructure and increasing physician preference for real time glucose tracking technologies.
Riyadh and Jeddah dominate the KSA Continuous Glucose Monitoring Systems market due to strong hospital infrastructure, concentration of endocrinology centers, and higher healthcare spending capacity. These cities benefit from the presence of large tertiary hospitals and specialized diabetes treatment centers that actively deploy advanced monitoring technologies. Dammam and Al Khobar also show strong adoption due to industrial workforce health programs and insurance backed chronic disease management initiatives that support technology driven diabetes care solutions.
Market Segmentation
By System Type
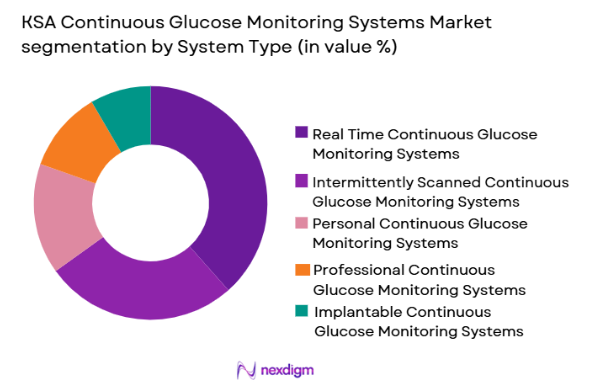
KSA Continuous Glucose Monitoring Systems market is segmented by system type into real-time CGM systems, intermittently scanned CGM systems, implantable CGM systems, professional CGM systems, and personal CGM systems. Recently, real-time CGM systems have a dominant market share due to factors such as strong patient preference for continuous alerts, wider physician recommendation, higher clinical accuracy, and integration with smartphone health ecosystems. The growth is also supported by increasing adoption among insulin-dependent diabetic populations and the availability of technologically advanced sensors offering automated alerts and predictive glucose trend analysis, making these systems more attractive in clinical and homecare environments.
By Platform Type
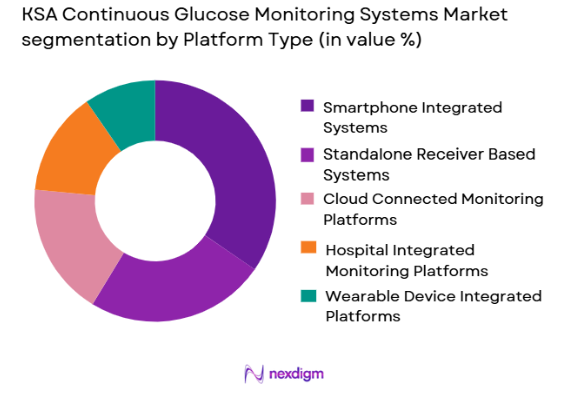
KSA Continuous Glucose Monitoring Systems market is segmented by platform type into standalone receiver based systems, smartphone integrated systems, cloud connected monitoring platforms, hospital integrated monitoring platforms, and wearable integrated platforms. Recently, smartphone integrated systems have a dominant market share due to factors such as high smartphone penetration, increasing use of mobile health apps, improved user convenience, and remote monitoring capabilities. These systems benefit from seamless connectivity with digital health records and telehealth services, which enhances physician oversight and patient adherence, making them particularly attractive for long-term diabetes management strategies.
Competitive Landscape
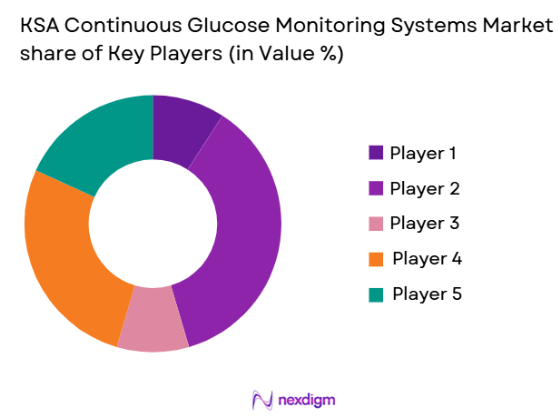
The KSA Continuous Glucose Monitoring Systems market shows moderate consolidation with a few multinational medical technology companies controlling a significant share through strong distribution partnerships and regulatory approvals. Leading players compete through sensor innovation, digital integration, and partnerships with healthcare providers. Market competition is also influenced by after sales service quality, sensor accuracy, and mobile application ecosystems. Regional distributors play a critical role in improving accessibility, while global companies maintain dominance through continuous product innovation and brand credibility.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Sensor Accuracy Range |
| Dexcom Inc | 1999 | United States | ~ | ~ | ~ | ~ | ~ |
| Abbott Laboratories | 1888 | United States | ~ | ~ | ~ | ~ | ~ |
| Medtronic plc | 1949 | Ireland | ~ | ~ | ~ | ~ | ~ |
| Senseonics Holdings | 1996 | United States | ~ | ~ | ~ | ~ | ~ |
| Roche Diagnostics | 1896 | Switzerland | ~ | ~ | ~ | ~ | ~ |
KSA Continuous Glucose Monitoring Systems Market Analysis
Growth Drivers
Rising prevalence of diabetes and prediabetes population in Saudi Arabia
Saudi Arabia continues to face a significant burden from diabetes and prediabetes, directly driving demand for continuous glucose monitoring solutions. According to the International Diabetes Federation, approximately 4,274,000 adults aged 20–79 were living with diabetes in Saudi Arabia in 2024, reflecting one of the highest absolute patient volumes in the Middle East. Additionally, data from the World Bank indicates that Saudi Arabia’s total population reached 36,947,000 in 2024, creating a large at-risk base for metabolic disorders. Urbanization levels exceeding 84,000,000 residents in cities across GCC-linked estimates and rising sedentary lifestyles further intensify the disease burden. The Saudi Ministry of Health reported over 2,100,000 outpatient diabetes-related visits in public healthcare facilities during 2023, indicating sustained healthcare system pressure. These figures collectively demonstrate a strong and expanding patient pool requiring long-term glucose monitoring, reinforcing demand for advanced monitoring devices across hospitals, clinics, and home-care settings.
Government healthcare modernization initiatives under Vision 2030
Saudi Arabia’s healthcare transformation under Vision 2030 is accelerating adoption of digital and connected medical technologies, including diabetes monitoring devices. The Saudi Ministry of Finance allocated SAR 214,000,000,000 to the health and social development sector in the 2024 national budget, emphasizing digital health infrastructure expansion. Additionally, the World Bank reports Saudi Arabia’s GDP reached USD 1,106,000,000,000 in 2024, supporting large-scale public investment capacity. The Saudi Data and AI Authority has also expanded national health data platforms, integrating over 32,000,000 digital health records to enable remote monitoring ecosystems. Furthermore, more than 500 hospitals and primary healthcare centers are undergoing privatization and digital upgrades under Vision 2030 programs. These developments create a strong institutional foundation for integrating continuous glucose monitoring devices into nationwide healthcare delivery systems.
Market Challenges
High cost of continuous glucose monitoring devices
The affordability of continuous glucose monitoring devices remains a key barrier despite rising healthcare spending. Although Saudi Arabia’s GDP per capita stood at USD 29,922 in 2024 (World Bank), income distribution disparities persist, limiting access to advanced monitoring technologies among middle-income and uninsured populations. The General Authority for Statistics reported that average monthly household expenditure reached SAR 16,125 in 2023, with healthcare accounting for a limited portion of spending capacity. Additionally, public healthcare utilization remains high, with over 140,000,000 patient visits recorded annually across government facilities (Saudi Ministry of Health), indicating reliance on subsidized services rather than out-of-pocket purchases of advanced devices. This financial dynamic restricts widespread adoption of premium monitoring systems, especially among patients requiring long-term continuous usage, thereby slowing penetration despite strong clinical need.
Limited reimbursement coverage for advanced monitoring devices
Reimbursement limitations continue to restrict adoption of advanced diabetes monitoring technologies across Saudi Arabia’s healthcare system. While the government provides extensive healthcare coverage, inclusion of high-end monitoring devices remains selective. According to the Council of Cooperative Health Insurance, over 11,500,000 individuals were covered under private health insurance schemes in 2024, yet device-specific reimbursements vary significantly across policies. Public healthcare still serves a majority of the population, with the Saudi Ministry of Health managing more than 60% of total hospital beds nationwide. Despite healthcare expenditure exceeding SAR 214,000,000,000 in 2024, allocation toward advanced device reimbursement remains limited compared to pharmaceuticals and primary care services. This gap reduces patient access to continuous monitoring technologies, particularly for lower-income and elderly populations who depend heavily on government-funded healthcare programs.
Market Opportunities
Expansion of telehealth integrated diabetes monitoring solutions
The rapid expansion of telehealth infrastructure in Saudi Arabia presents a strong opportunity for integrated diabetes monitoring solutions. The Saudi Ministry of Health reported over 30,000,000 telemedicine consultations conducted through national platforms such as Sehha and Mawid between 2022 and 2024. Additionally, internet penetration exceeded 36,000,000 users in 2024 (World Bank), enabling large-scale digital health adoption. The Communications, Space and Technology Commission also confirmed mobile subscription levels above 48,000,000 active connections, supporting real-time health data transmission. These digital infrastructure advancements allow continuous glucose monitoring devices to integrate with mobile applications and telehealth platforms, enabling remote patient management. This creates a scalable ecosystem for chronic disease monitoring, improving patient adherence while expanding device utilization across both urban and semi-urban populations.
Localization of device distribution through regional partnerships
Saudi Arabia’s push for localization of medical device supply chains is creating opportunities for regional partnerships and distribution expansion. Under Vision 2030, the Saudi Authority for Industrial Cities and Technology Zones has developed over 36 industrial cities, supporting domestic manufacturing and logistics infrastructure. The World Bank reported foreign direct investment inflows reaching USD 25,600,000,000 in 2023, indicating strong international participation in local industries. Additionally, the Saudi Food and Drug Authority has streamlined regulatory approvals for medical devices, reducing registration timelines and enabling faster market entry. With more than 500 licensed medical device distributors operating nationwide, localization efforts enhance supply chain efficiency and accessibility. These developments support partnerships between global manufacturers and local distributors, improving availability of diabetes monitoring devices across hospitals, pharmacies, and home-care channels.
Future Outlook
The KSA Continuous Glucose Monitoring Systems market is expected to witness strong growth over the next five years supported by rising diabetes awareness, government healthcare modernization, and growing adoption of connected health technologies. Advances in sensor miniaturization and artificial intelligence driven analytics are expected to improve monitoring accuracy. Regulatory support for digital health devices is likely to accelerate approvals. Demand from homecare patients and telehealth programs is expected to remain a major growth contributor.
Major Players
- Dexcom Inc
- Abbott Laboratories
- Medtronic plc
- Senseonics Holdings Inc
- Roche Diagnostics
- Ascensia Diabetes Care
- Ypsomed AG
- Novo Nordisk
- Terumo Corporation
- Nipro Corporation
- Sanofi
- LifeScan Inc
- B Braun Melsungen AG
- AgaMatrix Inc
- i-SENS Inc
Key Target Audience
- Medical device manufacturers
- Digital health technology providers
- Hospital procurement departments
- Private healthcare providers
- Diabetes specialty clinics
- Health insurance providers
- Investments and venture capitalist firms
- Government and regulatory bodies
Research Methodology
Step 1: Identification of Key Variables
Market variables such as diabetes prevalence, CGM adoption rates, pricing trends, regulatory environment, and healthcare investments were identified. Demand supply mapping and technology penetration factors were also considered to structure the research base.
Step 2: Market Analysis and Construction
Data triangulation was performed using healthcare statistics, company financials, device approvals, and procurement trends. Market sizing was constructed using bottom up device adoption analysis and validation through healthcare expenditure patterns.
Step 3: Hypothesis Validation and Expert Consultation
Industry assumptions were validated through consultations with healthcare professionals, distributors, and technology specialists. Expert insights helped validate adoption trends, regulatory developments, and competitive positioning.
Step 4: Research Synthesis and Final Output
All findings were consolidated through structured analytical frameworks. Quantitative and qualitative insights were combined to produce validated conclusions aligned with observed industry developments and technology adoption patterns.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Rising prevalence of diabetes and prediabetes population in Saudi Arabia
Government healthcare modernization initiatives under Vision 2030
Growing awareness of real time glucose monitoring benefits
Increasing adoption of remote patient monitoring technologies
Expansion of private diabetes specialty clinics - Market Challenges
High cost of continuous glucose monitoring devices
Limited reimbursement coverage for advanced monitoring devices
Patient discomfort related to sensor insertion
Data privacy concerns related to connected devices
Shortage of trained diabetes technology specialists - Market Opportunities
Expansion of telehealth integrated diabetes monitoring solutions
Localization of device distribution through regional partnerships
Growing demand for pediatric diabetes monitoring solutions - Trends
Integration of artificial intelligence in glucose trend prediction
Growth of minimally invasive and needle free sensors
Increasing use of mobile health applications for diabetes care
Partnerships between device makers and digital health firms
Shift toward long duration implantable glucose sensors - Government Regulations & Defense Policy
Saudi FDA regulatory approvals for wearable medical devices
National diabetes screening and monitoring initiatives
Digital health regulatory frameworks for connected monitoring devices - SWOT Analysis
- Stakeholder and Ecosystem Analysis
- Porter’s Five Forces Analysis
- Competition Intensity and Ecosystem Mapping
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Real Time Continuous Glucose Monitoring Systems
Intermittently Scanned Continuous Glucose Monitoring Systems
Implantable Continuous Glucose Monitoring Systems
Professional Continuous Glucose Monitoring Systems
Personal Continuous Glucose Monitoring Systems - By Platform Type (In Value%)
Standalone Receiver Based Systems
Smartphone Integrated Systems
Cloud Connected Monitoring Platforms
Hospital Integrated Monitoring Platforms
Wearable Device Integrated Platforms - By Fitment Type (In Value%)
Subcutaneous Sensor Based Systems
Implantable Long Duration Sensors
Disposable Sensor Based Systems
Reusable Transmitter Based Systems
Patch Based Wearable Monitoring Systems - By End User Segment (In Value%)
Hospitals and Specialty Diabetes Clinics
Homecare Patients
Ambulatory Surgical Centers
Diagnostic Centers
Research Institutions - By Procurement Channel (In Value%)
Direct Hospital Procurement
Retail Pharmacy Distribution
Online Medical Device Platforms
Government Healthcare Tenders
Private Insurance Provider Networks - By Material / Technology (in Value %)
Electrochemical Sensor Technology
Optical Fluorescence Based Sensors
Enzyme Based Glucose Detection Technology
Nanotechnology Enabled Sensors
Bluetooth Enabled Data Transmission Modules
- Market structure and competitive positioning
- Market share snapshot of major players
- Cross Comparison Parameters (Product Portfolio Breadth, Sensor Accuracy Levels, Device Connectivity Features, Regulatory Approvals, Distribution Network Strength, Pricing Strategy, R&D Investment, Digital Platform Integration, After Sales Support, Strategic Partnerships)
- SWOT Analysis of Key Players
- Pricing & Procurement Analysis
- Key Players
Dexcom Inc
Abbott Laboratories
Medtronic plc
Senseonics Holdings Inc
F Hoffmann La Roche Ltd
Ascensia Diabetes Care Holdings AG
Ypsomed AG
Novo Nordisk A/S
Terumo Corporation
Nipro Corporation
Sanofi SA
LifeScan Inc
B Braun Melsungen AG
AgaMatrix Inc
i-SENS Inc
- Hospitals adopting CGM systems for critical care glucose monitoring
- Homecare patients driving demand for easy to use wearable sensors
- Diagnostic centers using CGM data for metabolic profiling
- Insurance supported chronic disease management programs increasing adoption
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025