Market Overview
The KSA diagnostic biomarkers market is growing significantly due to the increasing prevalence of chronic diseases and the rise in awareness about personalized medicine. Based on a recent historical assessment, the market size is expected to reach USD ~ billion, driven by advancements in genomics, biotechnology, and the demand for precision healthcare. This growth is also facilitated by continuous government initiatives and healthcare infrastructure development, which are enhancing biomarker-based diagnostics.
The dominant countries in this market include the United States, Germany, and Japan, with major cities such as New York, Berlin, and Tokyo driving significant demand. The dominance of these regions can be attributed to their advanced healthcare systems, higher investment in research and development, and the presence of leading biotechnology firms. These countries are also seeing rising demand for early disease detection and precision medicine, fostering the growth of the diagnostic biomarkers market.
Market Segmentation
By Product Type
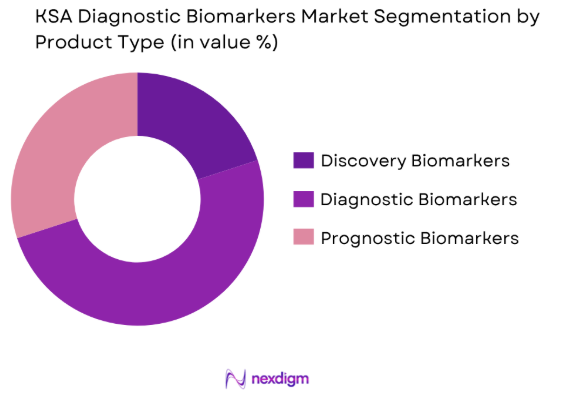
The KSA diagnostic biomarkers market is segmented by product type into discovery biomarkers, diagnostics biomarkers, and prognostic biomarkers. Recently, diagnostic biomarkers have dominated the market share, driven by increasing adoption in clinical diagnostics, particularly for early-stage cancer detection, genetic disorders, and other chronic diseases. The expanding research base and advancements in molecular technologies, alongside growing demand for non-invasive diagnostic tools, are key factors contributing to the dominance of this segment.
By Platform Type
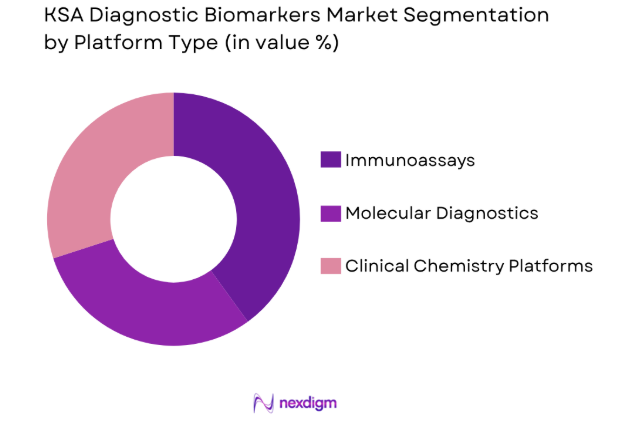
The KSA diagnostic biomarkers market is segmented by platform type into immunoassays, molecular diagnostics, and clinical chemistry platforms. Immunoassays currently hold the largest market share due to their widespread use in various disease diagnostics, particularly in immuno-oncology and infectious diseases. The established infrastructure, ease of use, and the ability to perform high-throughput testing have cemented the dominance of immunoassays in diagnostic settings.
Competitive Landscape
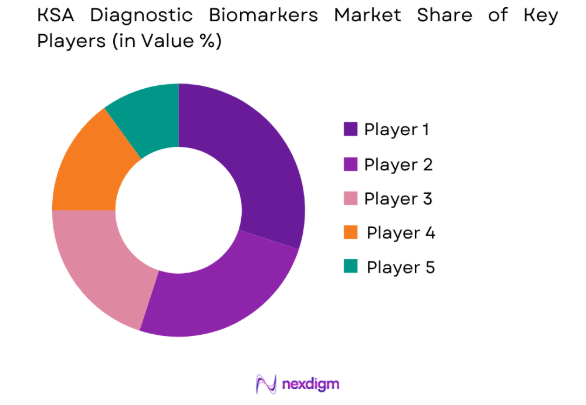
The diagnostic biomarkers market is highly competitive, with both established players and new entrants competing for market share. Major players are focusing on innovation, mergers, and acquisitions to expand their offerings and global reach. Consolidation in the market is expected as companies strive to enhance their product portfolios and enter emerging markets. Key players are investing heavily in R&D to develop new diagnostic technologies, thus driving market consolidation.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD) | Additional Parameter |
| Siemens Healthineers | 1847 | Germany | ~ | ~ | ~ | ~ | ~ |
| Abbott Laboratories | 1888 | USA | ~ | ~ | ~ | ~ | ~ |
| Roche Diagnostics | 1896 | Switzerland | ~ | ~ | ~ | ~ | ~ |
| Thermo Fisher Scientific | 1956 | USA | ~ | ~ | ~ | ~ | ~ |
| Illumina | 1998 | USA | ~ | ~ | ~ | ~ | ~ |
KSA Diagnostic Biomarkers Market Analysis
Growth Drivers
Technological Advancements in Molecular Diagnostics
The adoption of advanced molecular diagnostic tools has significantly accelerated the growth of the diagnostic biomarkers market. Technologies such as next-generation sequencing (NGS), polymerase chain reaction (PCR), and microarrays have enabled the identification of biomarkers with unprecedented precision and sensitivity. These innovations allow for early detection of diseases, particularly cancer and genetic disorders, and are becoming integral in personalized medicine. The increasing demand for non-invasive diagnostic methods further fuels the growth of these technologies, as they offer quicker results with minimal patient discomfort. As the demand for personalized healthcare grows, the market for molecular diagnostics and associated biomarkers is expected to expand rapidly, driven by technological improvements in sequencing technologies, data analytics, and biomarker discovery.
Rising Chronic Disease Prevalence
The increasing prevalence of chronic diseases, particularly cancer, diabetes, and cardiovascular diseases, is another key growth driver for the diagnostic biomarkers market. With chronic diseases becoming a leading cause of death globally, early detection and personalized treatment have gained prominence. Diagnostic biomarkers are playing a vital role in identifying these diseases at an early stage, improving patient outcomes and reducing healthcare costs. As the global population ages and lifestyle-related diseases become more common, the need for advanced diagnostic biomarkers will continue to rise. Government funding for healthcare infrastructure and biomarker-based research also supports the widespread adoption of diagnostic biomarkers in clinical settings.
Market Challenges
High Costs of Biomarker Testing
Despite the technological advancements and growing demand for diagnostic biomarkers, one of the significant challenges facing the market is the high cost of biomarker testing. The complexity of the tests, the need for sophisticated equipment, and the expertise required for data analysis contribute to the high costs. This is particularly challenging for healthcare systems in low-income countries, where the cost of advanced diagnostic procedures can be prohibitive. While costs are expected to decrease with technological improvements and economies of scale, the price of diagnostic biomarker testing remains a barrier to widespread adoption. As healthcare systems continue to prioritize cost-effectiveness, lowering the cost of these tests will be crucial for market growth.
Regulatory and Compliance Barriers
The regulatory landscape for diagnostic biomarkers is intricate, with differing requirements across regions. In several countries, biomarker-based diagnostics face lengthy approval processes, requiring extensive clinical trials and evidence-based validation, which can delay market entry. These regulatory barriers are especially challenging in emerging markets, where frameworks may be less structured. Moreover, concerns regarding data privacy and security add complexity, as biomarker testing often involves handling sensitive genetic and health data. The evolving nature of regulations and the need for robust data protection laws further complicate the market’s growth. Addressing these regulatory hurdles will be crucial for unlocking market potential, especially in developing regions with less mature healthcare infrastructures.
Opportunities
Expansion of Personalized Medicine
The shift towards personalized medicine presents a key opportunity for the diagnostic biomarkers market. As healthcare becomes more individualized, diagnostic biomarkers are essential in tailoring therapies to specific patient profiles. The growing use of genomic data to guide treatment decisions is driving the adoption of biomarkers, particularly in oncology, cardiology, and rare diseases. Personalized medicine enhances treatment efficacy, reduces side effects, and improves patient outcomes. As advancements in genomics, bioinformatics, and data analysis continue, the demand for diagnostic biomarkers is expected to rise. This growing trend provides significant growth opportunities for market players, enabling them to innovate and expand their offerings in personalized healthcare.
Government Initiatives and Healthcare Investments
Government initiatives aimed at enhancing healthcare systems and expanding access to diagnostics in developing countries offer a significant opportunity for the diagnostic biomarkers market. Increased government spending on healthcare infrastructure and support for biomarker-based research are expected to drive market growth. Public-private partnerships and collaborations with biotechnology firms are fostering innovation in the sector. Furthermore, the growing emphasis on preventive healthcare and early diagnosis in both developed and emerging markets is set to increase the demand for diagnostic biomarkers. These factors create a favorable environment for market expansion, providing a platform for growth and further adoption of diagnostic biomarker technologies worldwide.
Future Outlook
The future of the diagnostic biomarkers market looks promising, with expected growth driven by technological innovations, an increasing focus on personalized healthcare, and favorable government policies. Over the next five years, advancements in genomics, artificial intelligence, and data analytics are expected to enhance the accuracy and efficiency of diagnostic biomarkers. Increased adoption of precision medicine and government funding for healthcare initiatives are also key factors that will drive the market forward. As the demand for early-stage disease detection and personalized treatment continues to rise, the market is poised for substantial growth.
Major Players
- Siemens Healthineers
- Abbott Laboratories
- Roche Diagnostics
- ThermoFisher Scientific
- Illumina
- Bio-Rad Laboratories
- Beckman Coulter
- PerkinElmer
- Qiagen
- GE Healthcare
- Hologic
- Sysmex Corporation
- Agilent Technologies
- Abbott Diagnostics
- Danaher Corporation
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Healthcare providers
- Diagnostic laboratories
- Pharmaceutical companies
- Biotechnology firms
- Research institutions
- Medical device manufacturers
Research Methodology
Step 1: Identification of Key Variables
Identification of key factors influencing the diagnostic biomarkers market, such as technological advancements, regulatory frameworks, and disease prevalence.
Step 2: Market Analysis and Construction
Comprehensive market analysis using primary and secondary research, assessing market size, growth drivers, and competitive landscape.
Step 3: Hypothesis Validation and Expert Consultation
Validation of hypotheses through expert interviews and consultations with industry leaders and market stakeholders.
Step 4: Research Synthesis and Final Output
Synthesis of data into actionable insights, including market forecasts, trends, and recommendations for stakeholders.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Increased Healthcare Awareness
Government Funding for Healthcare Innovations
Rising Prevalence of Chronic Diseases - Market Challenges
High Cost of Diagnostic Equipment
Lack of Skilled Healthcare Personnel
Regulatory Barriers for Market Entry - Market Opportunities
Advancements in Biomarker Discovery Technology
Growing Adoption of Personalized Medicine
Expansion of Private and Government Healthcare Funding - Trends
Emergence of AI-Driven Diagnostics
Integration of Biomarkers in Disease Prevention
Growth in Genetic Biomarker Testing - Government Regulations
- SWOT Analysis of Key Competitors
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Biomarker Discovery Systems
Biomarker Diagnostics Systems
Biomarker Validation Systems
Point-of-Care Testing Systems
Research Use Only Systems - By Platform Type (In Value%)
Immunoassays
Molecular Diagnostics
Clinical Chemistry
Next-Generation Sequencing
Mass Spectrometry - By Fitment Type (In Value%)
Lab-Based Solutions
Point-of-Care Solutions
Wearable Devices
Mobile Health Solutions
Home Testing Kits - By End User Segment (In Value%)
Hospitals
Diagnostic Laboratories
Research Institutions
Pharmaceutical and Biotechnology Companies
Clinics - By Procurement Channel (In Value%)
Direct Procurement
Healthcare Tenders
Online Purchasing Platforms
Third-Party Distributors
Hospital Supply Chains
- Market Share Analysis
- Cross Comparison Parameters (Biomarker Discovery, Diagnostic Technology, Procurement Channel, System Complexity, End User Segment)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Siemens Healthineers
Roche Diagnostics
Abbott Laboratories
Thermo Fisher Scientific
BD Biosciences
Medtronic
GE Healthcare
PerkinElmer
Illumina
Bio-Rad Laboratories
Qiagen
Agilent Technologies
Hologic
Sysmex Corporation
Beckman Coulter
- Increasing Demand from Public and Private Healthcare
- Shift Towards Home-Based Diagnostics
- Growing Collaboration Between Private and Public Healthcare Sectors
- Expanding Role of Research Institutions
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035