Market Overview
The KSA Gene Therapy Market is projected to experience significant growth due to technological advancements and increasing investment in the healthcare sector. With a market size in USD ~ billion, the growth is driven by the rising demand for genetic disorder treatments, government support for gene therapy initiatives, and advancements in CRISPR technology. Key factors such as improved healthcare infrastructure and regulatory approvals further bolster the market’s expansion.
The market dominance is expected to be concentrated in major urban centers like Riyadh and Jeddah. These cities are experiencing increasing healthcare investments and the establishment of cutting-edge research facilities. The government’s focus on modernizing healthcare infrastructure and the rising awareness about genetic therapies will make these areas pivotal in driving market growth.
Market Segmentation
By Product Type:
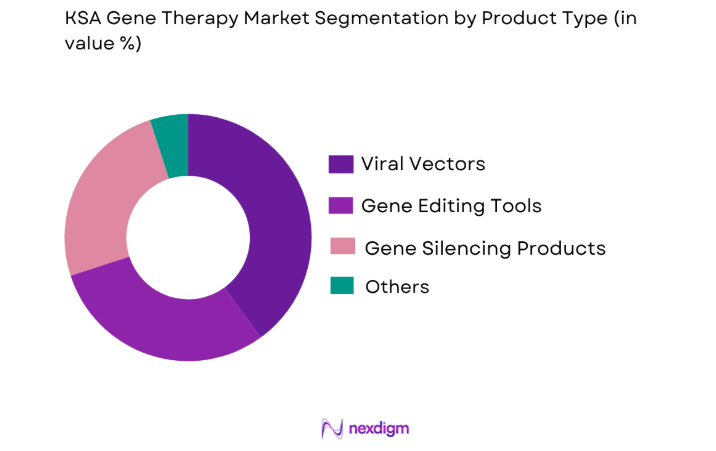
The KSA Gene Therapy Market is segmented by product type into viral vectors, gene editing tools, and gene silencing products. The viral vectors sub-segment currently dominates the market due to the wide application of viral vectors in gene delivery systems. This growth is propelled by the increasing use of adenoviral vectors and lentiviral vectors in the treatment of genetic disorders and cancers. Additionally, the high efficiency of viral vectors in gene transfer makes them a preferred choice for many gene therapy applications.
By End-User Type:
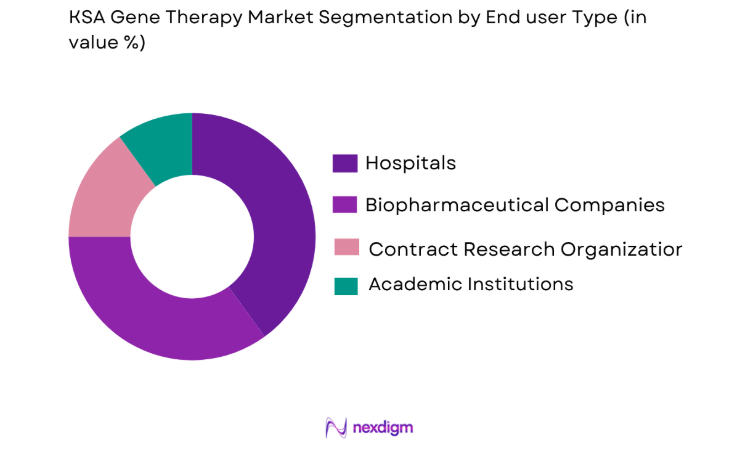
The end-user segment of the KSA Gene Therapy Market is crucial in determining the adoption rates of gene therapies. Hospitals and biopharmaceutical companies dominate this segment due to their pivotal role in the research, development, and application of gene therapy solutions. Hospitals provide direct patient care and access to gene therapies, while biopharmaceutical companies contribute to the development and distribution of these therapies. Additionally, contract research organizations (CROs) and academic institutions play a key role in conducting clinical trials and research on new gene therapies, further supporting market growth.
Competitive Landscape
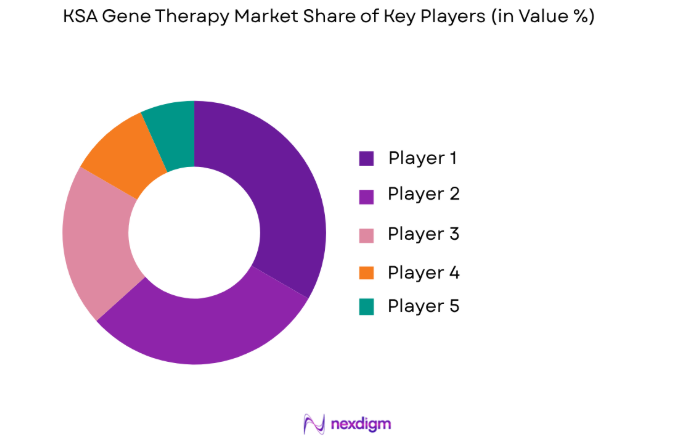
The competitive landscape of the KSA Gene Therapy Market is shaped by a few large multinational players and emerging biotech companies. These companies are collaborating with local organizations and government bodies to enhance their presence in the region. Consolidation is evident as larger firms seek partnerships and acquisitions to expand their product offerings and R&D capabilities in the field of gene therapy.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD) | Clinical Trials |
| Novartis | 1996 | Switzerland | ~ | ~ | ~ | ~ | ~ |
| Gilead Sciences | 1987 | USA | ~ | ~ | ~ | ~ | ~ |
| Bluebird Bio | 1992 | USA | ~ | ~ | ~ | ~ | ~ |
| Sangamo Therapeutics | 1995 | USA | ~ | ~ | ~ | ~ | ~ |
| CRISPR Therapeutics | 2013 | Switzerland | ~ | ~ | ~ | ~ | ~ |
KSA Gene Therapy Market Analysis
Growth Drivers
Rising Prevalence of Genetic Disorders:
The demand for gene therapy solutions in KSA is increasingly driven by the rising prevalence of genetic disorders such as hemophilia, sickle cell anemia, and various types of cancer. These diseases are becoming more prominent, pushing the healthcare system to find more effective and sustainable treatment options. Gene therapy presents a promising alternative to conventional treatments, with the ability to address the root cause of genetic disorders by targeting specific genes. As more genetic disorders are identified, the potential for gene therapy to offer long-term solutions grows, leading to further investment in research and clinical trials. The government’s focus on advancing genetic medicine, along with increasing funding for research in the field, has created a conducive environment for the development and application of gene therapy in the Kingdom. This growing recognition of gene therapy’s potential in treating genetic disorders, paired with technological advancements in gene delivery systems, is expected to drive the market’s growth in the coming years.
Technological Advancements in Gene Editing:
The rapid evolution of gene editing technologies, particularly CRISPR/Cas9, is playing a significant role in advancing gene therapy. With the development of more precise and efficient gene editing techniques, the potential to treat a broader range of genetic diseases has increased dramatically. The high accuracy of gene editing methods allows for targeted treatments, reducing the risks associated with traditional methods. Furthermore, innovations in gene editing are improving the delivery mechanisms of gene therapies, ensuring better outcomes for patients. As these technologies continue to evolve and become more accessible, the cost of gene therapies is expected to decrease, making it more affordable for both patients and healthcare providers. This reduction in cost, combined with more effective treatments, will likely result in greater adoption of gene therapy solutions in KSA, especially as healthcare infrastructure continues to modernize.
Market Challenges
High Cost of Gene Therapies:
One of the primary challenges hindering the growth of the KSA Gene Therapy Market is the high cost of gene therapies. Despite the promising results that gene therapies offer for treating genetic disorders, the development, production, and administration of these treatments come with significant costs. The advanced technologies and specialized equipment required for gene editing, along with the need for personalized therapies, contribute to the high price points. This cost is often prohibitive for many patients and healthcare systems, limiting the accessibility of these treatments in certain regions of KSA. While government investment in healthcare infrastructure is increasing, it remains a significant challenge for the market. Without substantial price reductions or government support to offset these costs, widespread adoption of gene therapy may be limited.
Regulatory Hurdles in Approval Process:
Another significant challenge for the KSA Gene Therapy Market is the regulatory approval process for new gene therapies. Regulatory bodies must ensure that gene therapies meet rigorous safety and efficacy standards before they can be made available to the public. However, the process for approval can be lengthy and complex, particularly for novel treatments such as gene therapies. These delays can impede the speed at which new treatments reach the market, thus slowing overall growth. Furthermore, regulatory frameworks in KSA are still evolving, and there is a lack of standardized guidelines for gene therapy. This uncertainty can create barriers for companies looking to enter the market, as they may be unsure of how their products will be regulated. Regulatory bottlenecks can delay the commercialization of innovative gene therapies, limiting market expansion.
Opportunities
Expanding Healthcare Infrastructure:
KSA’s ongoing efforts to enhance healthcare infrastructure present significant opportunities for the gene therapy market. The government is heavily investing in modernizing healthcare facilities and establishing specialized centers for advanced treatments. As part of the Vision 2030 initiative, there is an emphasis on increasing the availability of high-quality medical services to the population. This includes the expansion of research centers focused on gene therapy and the adoption of advanced healthcare technologies. The improvement of healthcare facilities provides a favorable environment for the introduction of new treatments, including gene therapies. As more specialized centers emerge across KSA, the accessibility of gene therapy solutions will increase, creating a broader patient base and supporting the market’s growth.
Collaborations and Partnerships with Global Players:
Collaborations between KSA healthcare institutions and global biopharmaceutical companies present a lucrative opportunity for the gene therapy market. Partnerships with established players in the gene therapy industry will facilitate the transfer of knowledge, technology, and resources, helping local players enhance their capabilities. Moreover, these collaborations can lead to joint research initiatives, accelerating the development of new treatments for genetic disorders. Global players bring experience, advanced technologies, and regulatory expertise, which can help navigate the complex market dynamics in KSA. These partnerships are expected to foster innovation in gene therapy and improve the overall treatment landscape in the Kingdom, positioning KSA as a regional leader in gene therapy development.
Future Outlook
The KSA Gene Therapy Market is poised for steady growth, driven by technological advancements, government investment in healthcare, and the increasing prevalence of genetic disorders. Over the next five years, the market is expected to expand significantly, particularly in the areas of gene editing and viral vector-based therapies. With continued support from both the public and private sectors, technological developments will help reduce the costs of gene therapy treatments, making them more accessible to a broader patient base. As regulatory frameworks stabilize and healthcare infrastructure improves, the Kingdom will become a hub for gene therapy innovation in the Middle East.
Major Players
- Novartis
- Gilead Sciences
- Bluebird Bio
- Sangamo Therapeutics
- CRISPR Therapeutics
- Roche
- Bayer
- Celgene
- Bristol-Myers Squibb
- ThermoFisher Scientific
- Takeda Pharmaceutical
- Intellia Therapeutics
- Biogen
- Pfizer
- Merck
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Biopharmaceutical companies
- Hospitals and healthcare providers
- Biotechnology firms
- Research institutions
- Contract research organizations
- Healthcare technology providers
Research Methodology
Step 1: Identification of Key Variables
This step involves defining the critical factors driving the market and establishing boundaries for research, focusing on genetic disorders, healthcare infrastructure, and technological advancements in gene therapy.
Step 2: Market Analysis and Construction
Market data is gathered through primary and secondary sources, including interviews with industry experts, public reports, and clinical trial data. The market is then segmented based on product types and end users.
Step 3: Hypothesis Validation and Expert Consultation
The collected data is analyzed to formulate hypotheses on market growth, technological trends, and regulatory developments. Expert consultations help validate assumptions and refine projections.
Step 4: Research Synthesis and Final Output
The research is synthesized to produce a comprehensive market report, detailing trends, growth drivers, challenges, and opportunities in the KSA Gene Therapy Market. The final output includes validated market forecasts and strategic recommendations.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Growth Drivers
Rising Prevalence of Genetic Disorders
Government Investment in Healthcare
Technological Advancements in Gene Editing - Market Challenges
High Cost of Gene Therapies
Regulatory and Compliance Barriers
Lack of Skilled Professionals - Market Opportunities
Increasing Demand for Personalized Medicine
Expansion of Healthcare Infrastructure
Collaboration Between Biopharma and Research Institutions - Trends
Adoption of CRISPR Technology
Growth of Gene Therapy Clinical Trials
Advances in Viral Vectors - Government Regulations
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Gene Editing Systems
Viral Vector Systems
Non-Viral Vector Systems
Gene Silencing Systems
Gene Delivery Platforms - By Platform Type (In Value%)
Ex Vivo Platforms
In Vivo Platforms
Viral Vectors Platforms
CRISPR-based Platforms
Other Gene Therapy Platforms - By Fitment Type (In Value%)
Therapeutic Solutions
Research Solutions
Diagnostic Solutions
Clinical Trial Solutions
Manufacturing Solutions - By EndUser Segment (In Value%)
Hospitals and Clinics
Biopharmaceutical Companies
Contract Research Organizations
Academic and Research Institutes
- Market Share Analysis
- CrossComparison Parameters (System Type, Platform Type, Procurement Channel, EndUser Segment, Fitment Type)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Porter’s Five Forces
- Key Players
Novartis
Gilead Sciences
Bluebird Bio
Sangamo Therapeutics
Spark Therapeutics
CRISPR Therapeutics
Regeneron Pharmaceuticals
Roche
Bayer
Celgene
Bristol-Myers Squibb
Thermo Fisher Scientific
Takeda Pharmaceutical
Intellia Therapeutics
Biogen
- Hospitals’ Growing Role in Gene Therapy
- Biopharmaceutical Companies Increasing Investment
- Emerging Role of Academic Institutions in Research
- The Rise of Contract Research Organizations
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035