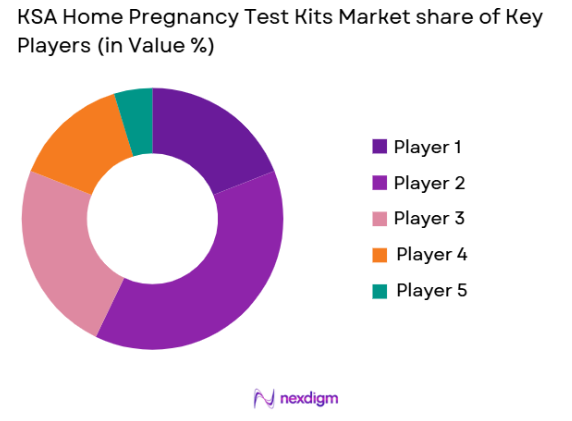
Market Overview
The KSA Home Pregnancy Test Kits market reached a value of USD ~ billion based on a recent historical assessment, supported by industry data from healthcare diagnostics market tracking databases. Growth is primarily driven by increasing awareness of early pregnancy detection, rising adoption of self-care diagnostic solutions, and improved retail pharmacy accessibility. The availability of affordable strip-based tests and discreet online purchasing channels has further strengthened demand, while expansion of women’s healthcare awareness programs continues to support consistent product adoption across the country.
Major demand is concentrated in cities such as Riyadh, Jeddah, and Dammam due to higher population density, stronger retail pharmacy infrastructure, and better access to women’s healthcare services. These metropolitan areas benefit from advanced healthcare retail networks, digital commerce adoption, and higher health awareness among working women. Urban purchasing patterns are also supported by higher disposable income levels and greater acceptance of self-testing healthcare products compared to smaller cities and rural regions where access remains comparatively limited.
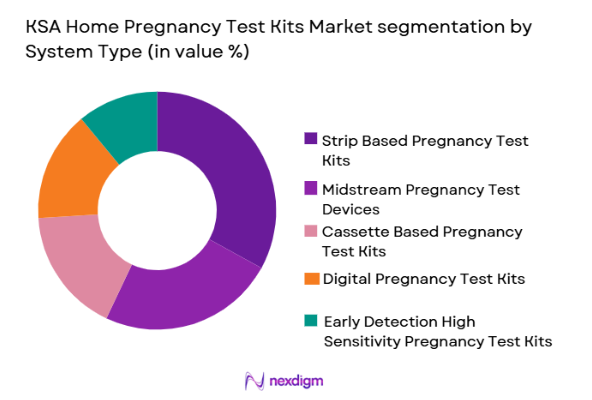
Market Segmentation
By System Type
KSA Home Pregnancy Test Kits market is segmented by system type into strip based pregnancy test kits, midstream pregnancy test devices, cassette based pregnancy test kits, digital pregnancy test kits, and early detection high sensitivity pregnancy test kits. Recently, strip based pregnancy test kits has a dominant market share due to factors such as affordability, easy availability, and strong consumer trust built over years of usage. These kits are widely preferred because they are inexpensive compared to digital alternatives and are easily accessible through pharmacies and online retailers. Their simple testing process and reliable accuracy have ensured repeat purchases among consumers. Additionally, manufacturers continue to offer these kits in multi-pack formats, which increases value for money. While digital and early detection kits are gaining traction in premium segments, strip based kits remain the preferred choice among price sensitive consumers, ensuring their leadership position in the market.
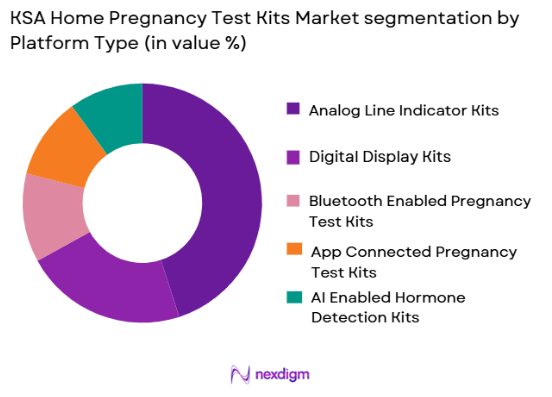
By Platform Type
KSA Home Pregnancy Test Kits market is segmented by platform type into analog line indicator kits, digital display kits, bluetooth enabled pregnancy test kits, app connected pregnancy test kits, and AI enabled hormone detection kits. Recently, analog line indicator kits has a dominant market share due to their low cost, high reliability, and strong consumer familiarity. These products are considered the standard option as they provide clear results without requiring electronic components, making them widely accepted across all income groups. Retailers also prefer stocking these products because of their steady demand and longer shelf stability. Digital display kits are increasingly being adopted in urban areas due to ease of result interpretation, but their higher price restricts mass adoption. Smart connected platforms are still emerging as niche offerings due to limited awareness and relatively higher costs, which keeps analog kits in the leading position.
Competitive Landscape
The KSA Home Pregnancy Test Kits market shows moderate consolidation with a mix of global diagnostic companies and regional pharmaceutical distributors. Large multinational brands dominate through strong distribution agreements with pharmacy chains and hospitals, while regional companies compete on pricing and localized distribution. Product innovation remains limited but branding, accuracy claims, and packaging privacy features influence competition. Market leaders maintain advantages through regulatory approvals, supply reliability, and strong retail pharmacy partnerships.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Distribution Strength |
| Abbott Laboratories | 1888 | USA | ~ | ~ | ~ | ~ | ~ |
| Procter & Gamble Health | 1837 | USA | ~ | ~ | ~ | ~ | ~ |
| Church & Dwight | 1846 | USA | ~ | ~ | ~ | ~ | ~ |
| QuidelOrtho Corporation | 1979 | USA | ~ | ~ | ~ | ~ | ~ |
| Swiss Precision Diagnostics | 2007 | Switzerland | ~ | ~ | ~ | ~ | ~ |
KSA Home Pregnancy Test Kits Market Analysis
Growth Drivers
Expansion of Retail Pharmacy and E-Commerce Distribution Channels
The growing penetration of organized pharmacy chains and online healthcare retail platforms has significantly improved the accessibility of home pregnancy test kits across Saudi Arabia. Large pharmacy retailers continue to expand their footprint across major cities, ensuring consistent product availability and brand visibility. Online healthcare platforms have enabled discreet purchases, which is an important factor influencing consumer behavior in conservative societies. The ability to order products confidentially with home delivery has reduced social barriers related to purchasing pregnancy tests. Increased smartphone usage and digital payment adoption have also supported the growth of online healthcare purchases. Retailers are also introducing private label products to improve margins and consumer loyalty. Promotional pricing strategies and bundle offers have further stimulated repeat purchases. Growing logistics efficiency has reduced product delivery timelines, enhancing customer satisfaction. Together these factors are strengthening distribution networks and expanding the addressable consumer base.
Rising Awareness of Women’s Preventive Health and Early Diagnosis
Increasing focus on women’s health awareness is driving demand for home pregnancy testing products in Saudi Arabia. Government healthcare transformation initiatives and private sector awareness campaigns have encouraged early pregnancy confirmation practices. Women are becoming more proactive in managing reproductive health through self-testing solutions. Healthcare providers are also recommending early confirmation through home tests before clinical consultations. Increased participation of women in the workforce has strengthened the need for convenient and time-saving healthcare solutions. Educational content through digital platforms has improved understanding of test accuracy and usage methods. Social media discussions around reproductive health have also reduced stigma associated with self-testing products. Growing demand for privacy and independence in health decisions further supports this market. These social and healthcare shifts continue to create favorable conditions for steady product adoption.
Market Challenges
Cultural Sensitivity and Social Purchase Barriers
Despite healthcare modernization, social norms still influence purchasing behavior for pregnancy testing kits in certain regions of Saudi Arabia. Some consumers remain hesitant to purchase these products in physical stores due to privacy concerns. This leads to delayed purchases or preference for online alternatives, limiting impulse buying opportunities. Retailers must ensure discreet packaging and confidential transactions to address this challenge. Lack of open discussion around reproductive health in some communities also affects awareness levels. Limited health education in certain demographics further slows adoption of self-diagnostic practices. Product marketing also remains conservative due to regulatory and cultural expectations. Pharmacies must balance visibility with discretion when merchandising such products. These factors collectively create a unique market environment requiring sensitive branding strategies. Addressing these social barriers remains essential for market expansion.
Price Sensitivity and Presence of Low Cost Alternatives
Price sensitivity remains a significant challenge as consumers often compare multiple brands before making purchase decisions. The availability of low-cost imported test kits increases pricing pressure on established brands. Some lower priced products may compromise on quality, creating trust concerns across the category. Premium digital kits face slower adoption due to their higher price points compared to traditional strip tests. Retailers also focus on fast-moving low-cost inventory to maintain turnover efficiency. Economic considerations among young consumers also affect willingness to experiment with higher priced variants. Promotional discounting by competitors further intensifies price competition. Manufacturers must balance affordability with quality assurance to maintain brand reputation. Maintaining competitive pricing while ensuring regulatory compliance remains a critical operational challenge.
Opportunities
Growth of Premium Digital and Early Detection Testing Products
There is increasing opportunity for premium pregnancy test kits that offer digital result displays and early detection capabilities. Urban consumers are showing growing interest in technologically advanced testing solutions that provide clearer results and improved sensitivity. Digital pregnancy tests reduce ambiguity in interpretation compared to traditional line indicator tests. Increasing disposable income among working women supports the adoption of higher value diagnostic products. Companies introducing multilingual digital interfaces can improve usability for diverse populations. Early detection capabilities also appeal to consumers seeking faster confirmation. Premium packaging and branding can further strengthen positioning in the high value segment. Companies investing in product differentiation through innovation may capture higher margins. As awareness improves, this premium category could experience accelerated adoption. This segment represents a strong future growth pathway for manufacturers.
Integration with Digital Health and Teleconsultation Services
Integration of home pregnancy testing with telehealth ecosystems offers strong future growth potential. Digital healthcare platforms are expanding rapidly, allowing consumers to consult doctors remotely after self-testing. Companies can develop bundled offerings combining test kits with virtual consultations. Mobile health applications can also integrate pregnancy tracking tools linked with diagnostic products. Subscription based reproductive health services could create recurring revenue models. Digital engagement can improve customer retention and brand loyalty. Healthcare providers may collaborate with diagnostic brands to promote early testing protocols. This approach also aligns with broader digital health transformation strategies in Saudi Arabia. Integration with digital health ecosystems could significantly enhance the value proposition of pregnancy test kits. Such partnerships could reshape the competitive landscape over the coming years.
Future Outlook
The KSA Home Pregnancy Test Kits market is expected to experience stable expansion driven by increasing acceptance of self-diagnostic healthcare solutions. Product innovation focusing on digital accuracy and early detection capabilities will shape future competition. Regulatory alignment with international diagnostic standards will support product quality improvements. Rising online pharmacy penetration and digital healthcare integration will further enhance accessibility. Demand is expected to remain strongest in urban regions with growing adoption gradually extending into secondary cities.
Major Players
- Abbott Laboratories
- Procter & Gamble Health
- Church & Dwight Co Inc
- QuidelOrtho Corporation
- Swiss Precision Diagnostics
- Geratherm Medical AG
- bioMérieux SA
- Cardinal Health
- ACON Laboratories
- Piramal Pharma Limited
- Mankind Pharma
- SPIMACO
- Jamjoom Pharma
- Wondfo Biotech
- Sugentech Inc
Key Target Audience
- Diagnostic device manufacturers
- Pharmaceutical distributors
- Retail pharmacy chains
- Online healthcare retailers
- Hospitals procurement departments
- Women healthcare product companies
- Investments and venture capitalist firms
- Government and regulatory bodies
Research Methodology
Step 1: Identification of Key Variables
The research process began with identification of key demand variables such as product adoption trends, pricing structures, distribution channels, and regulatory approvals. Healthcare consumption patterns and demographic indicators were also evaluated to define market boundaries. Product type classification and technology usage trends were mapped. Demand drivers were validated through industry publications and healthcare statistics. Macroeconomic indicators influencing healthcare spending were also considered.
Step 2: Market Analysis and Construction
Market size assessment was conducted through analysis of diagnostic industry databases and healthcare device reports. Segmentation analysis was constructed based on product types and platform technologies. Competitive benchmarking was conducted across manufacturers and distributors. Supply chain evaluation included distributors and pharmacy networks. Pricing patterns were analyzed across offline and online channels.
Step 3: Hypothesis Validation and Expert Consultation
Preliminary findings were validated through comparison with healthcare diagnostics reports and sector data. Industry assumptions were tested against regional healthcare development indicators. Product demand trends were evaluated against pharmacy sales patterns. Expert opinions from healthcare distribution professionals were considered. Data inconsistencies were corrected through triangulation methods.
Step 4: Research Synthesis and Final Output
Final insights were synthesized into a structured market framework combining quantitative and qualitative analysis. Market forecasts were aligned with healthcare technology adoption trends. Competitive analysis was structured using company positioning data. Strategic conclusions were developed through comparative analysis. The final report was structured to support strategic decision making.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Rising awareness of early pregnancy detection among women
Growing e commerce penetration for confidential purchases
Increasing female workforce participation in KSA
Improved affordability of rapid diagnostic kits
Expanding pharmacy retail networks across urban regions - Market Challenges
Cultural sensitivity around pregnancy testing purchases
Availability of low quality counterfeit products
Limited awareness in rural and semi urban regions
Price competition reducing manufacturer margins
Regulatory approval delays for advanced digital kits - Market Opportunities
Growth potential for discreet online subscription based kit delivery
Introduction of smart pregnancy kits integrated with fertility apps
Expansion into premium early detection product segments - Trends
Increasing demand for digital pregnancy test kits
Shift toward early detection high sensitivity kits
Growing preference for private home based diagnostics
Rise of eco friendly and biodegradable test materials
Brand differentiation through accuracy claims and digital features - Government Regulations & Defense Policy
Saudi FDA regulations governing OTC diagnostic devices
Product registration and import compliance requirements
Quality certification standards for rapid diagnostic kits - SWOT Analysis
- Stakeholder and Ecosystem Analysis
- Porter’s Five Forces Analysis
- Competition Intensity and Ecosystem Mapping
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Strip Based Pregnancy Test Kits
Midstream Pregnancy Test Devices
Cassette Based Pregnancy Test Kits
Digital Pregnancy Test Kits
Early Detection Pregnancy Test Kits - By Platform Type (In Value%)
Analog Line Indicator Kits
Digital Display Kits
Bluetooth Enabled Smart Test Kits
App Connected Fertility Tracking Kits
AI Enabled Hormone Detection Kits - By Fitment Type (In Value%)
Single Use Disposable Kits
Multi Pack Test Kits
Travel Friendly Compact Kits
High Sensitivity Early Detection Kits
Standard Sensitivity Kits - By EndUser Segment (In Value%)
Individual Home Users
Online Pharmacy Consumers
Retail Pharmacy Buyers
Women Health Clinics
Telehealth Diagnostic Service Providers - By Procurement Channel (In Value%)
Retail Pharmacy Chains
Online Marketplaces
Hospital Pharmacy Retail Counters
Direct to Consumer Brand Websites
Supermarkets and Hypermarkets - By Material / Technology (in Value %)
Lateral Flow Immunoassay Technology
hCG Hormone Detection Technology
Plastic Cassette Diagnostic Devices
Biodegradable Test Strip Materials
Digital Sensor Based Detection Technology
- Market structure and competitive positioning
- Market share snapshot of major players
- Cross Comparison Parameters (Product Accuracy Rate, Price Range Positioning, Distribution Network Strength, Product Portfolio Diversity, Digital Integration Capability, Regulatory Certifications, Brand Recognition Strength, Online Availability, Innovation Pipeline Strength, Packaging Discretion Features, Sensitivity Levels, Retail Partnerships, Manufacturing Capacity, Import Dependency Ratio, After Sales Support Strategy)
- SWOT Analysis of Key Players
- Pricing & Procurement Analysis
- Key Players
Abbott Laboratories
Procter & Gamble Health
Church & Dwight Co Inc
QuidelOrtho Corporation
bioMérieux SA
Becton Dickinson and Company
Cardinal Health
ACON Laboratories Inc
Geratherm Medical AG
Swiss Precision Diagnostics GmbH
Mankind Pharma
Piramal Pharma Limited
Al Nahdi Medical Company
Jamjoom Pharma
SPIMACO Addwaeih
- Home users prefer affordable multi pack test kits for routine confirmation
- Online buyers prioritize discreet packaging and fast delivery
- Clinics use home test kits for preliminary screening support
- Telehealth providers recommend home kits for remote consultations
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035