Market Overview
The KSA lancet devices market is valued at SAR ~ billion in 2024, with a steady growth trajectory driven by the increasing prevalence of diabetes in the region and the rising adoption of home healthcare solutions. With the Saudi Arabian population experiencing a significant rise in chronic diseases such as diabetes, demand for self-monitoring devices like lancets has escalated. The government’s initiatives to encourage healthcare accessibility and affordability have also positively impacted market expansion. The market’s growth is propelled by the need for advanced, pain-free testing devices that cater to diabetic patients’ requirements.
The market is predominantly dominated by cities with large urban populations like Riyadh, Jeddah, and Dammam. These regions benefit from a more advanced healthcare infrastructure and are home to numerous private and public hospitals and clinics that serve a significant portion of the diabetic population. Additionally, the demand for lancets is driven by these cities’ high rates of diabetes diagnoses and healthcare expenditure. The eastern and central regions are also key contributors, supported by government initiatives aimed at providing healthcare to underserved areas.
Market Segmentation
By Product Type
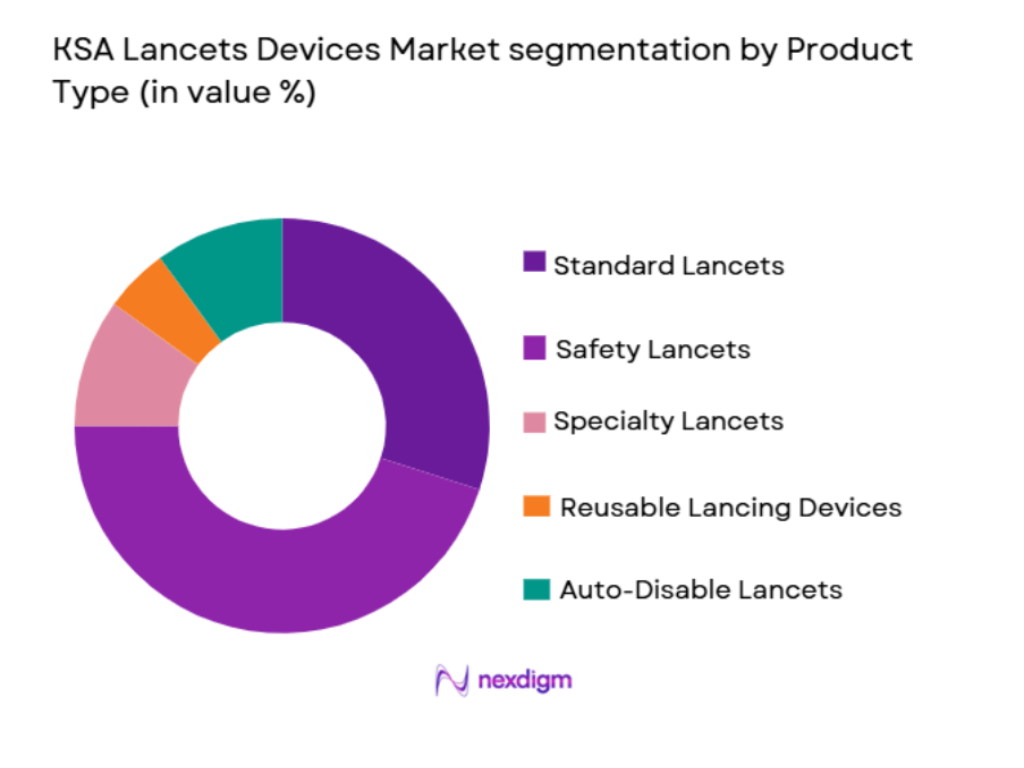
The KSA lancets devices market is segmented by product type into standard lancets, safety lancets, specialty lancets, reusable lancing devices, and auto-disable lancets. Among these, safety lancets have been dominating the market due to growing consumer demand for safety features, especially in light of increasing awareness about infection risks associated with traditional lancets. These lancets, equipped with automatic retractable needles, ensure that patients do not come into contact with the needle after use, significantly reducing the risk of needle-stick injuries. Furthermore, the preference for painless and easy-to-use safety lancets has grown as a result of their appeal to diabetic patients who require frequent blood sampling. The evolving consumer preference for convenience and safety is thus driving the market for safety lancets in KSA.
By End-User
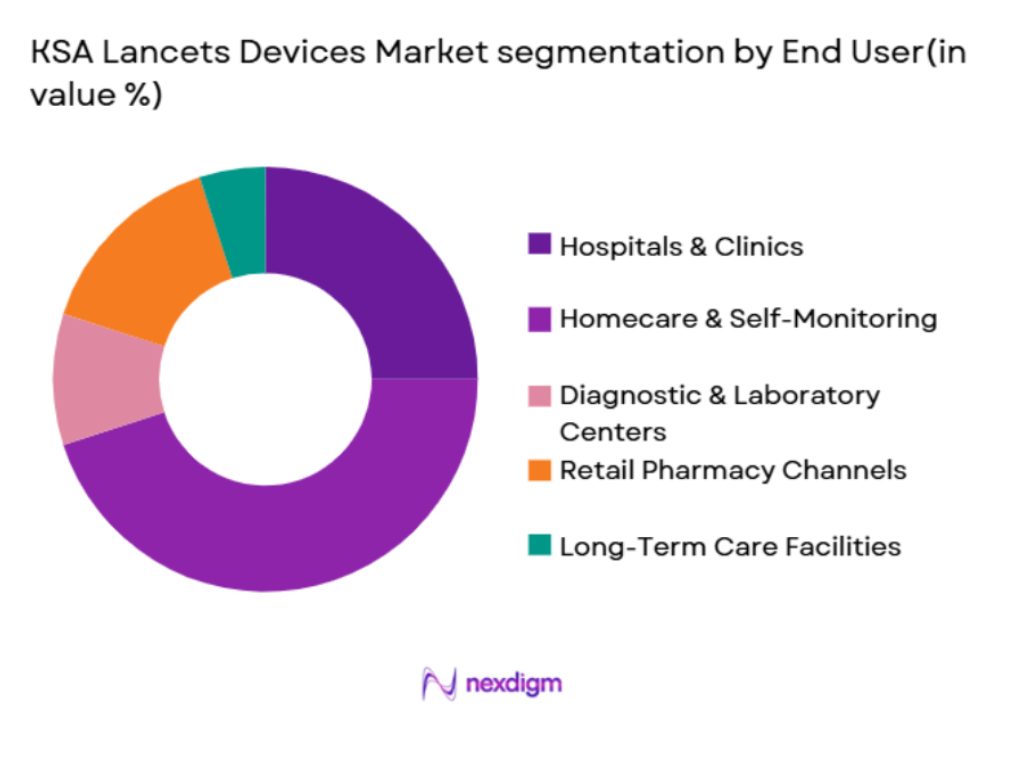
The market segmentation by end-user includes hospitals & clinics, homecare & self-monitoring, diagnostic & laboratory centers, retail pharmacy channels, and long-term care facilities. Homecare & self-monitoring segments account for the largest share, driven by the increasing trend of patients managing their own diabetes. With the rising awareness and affordability of home testing devices, many diabetic patients in Saudi Arabia are preferring self-monitoring methods. This shift is further reinforced by the ongoing advancements in lancet technology, making it easier and more comfortable for individuals to monitor their blood glucose levels independently. The trend toward more personalized healthcare is a key driver for this market segment’s dominance.
 Competitive Landscape
Competitive Landscape
The KSA lancets devices market is highly competitive, with both global and regional players dominating the sector. Companies like Becton Dickinson & Co, F. Hoffmann-La Roche Ltd, and B. Braun SE are prominent due to their strong brand presence and advanced product offerings in safety lancets and self-testing devices. The local players also compete with international brands by providing cost-effective solutions tailored to local needs. Additionally, there is a rising number of smaller players who offer specialized lancets with innovative features to cater to specific patient needs.
| Company | Establishment Year | Headquarters | Product Portfolio | Technology Focus | Key Distribution Channels | Revenue Sources |
| Becton Dickinson & Co | 1897 | Franklin Lakes, NJ, USA | ~ | ~ | ~ | ~ |
| F. Hoffmann-La Roche Ltd | 1896 | Basel, Switzerland | ~ | ~ | ~ | ~ |
| B. Braun SE | 1839 | Melsungen, Germany | ~ | ~ | ~ | ~ |
| Abbott Laboratories | 1888 | Abbott Park, IL, USA | ~ | ~ | ~ | ~ |
| Owen Mumford Ltd | 1952 | Oxford, UK | ~ | ~ | ~ | ~ |
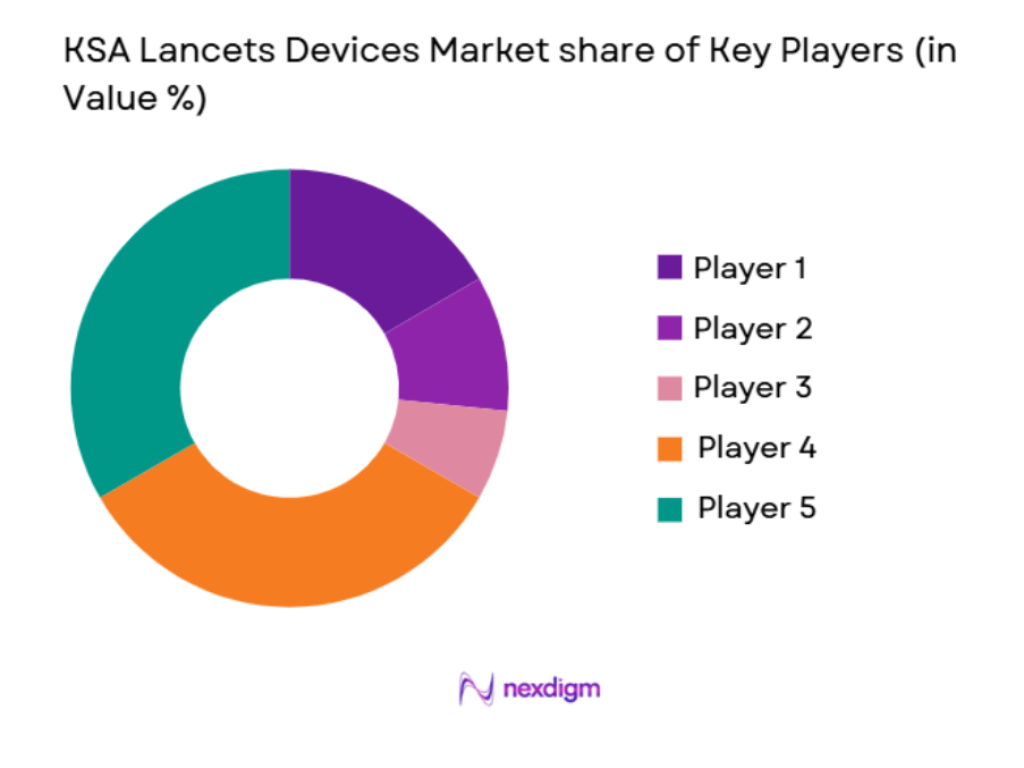 KSA Lancets Devices Market Analysis
KSA Lancets Devices Market Analysis
Growth Drivers
Rising diabetes prevalence and testing frequency
The rising prevalence of diabetes in Saudi Arabia is a key driver for the lancets devices market. In 2023, the International Diabetes Federation (IDF) reported that approximately 3.8 million people in Saudi Arabia are living with diabetes, accounting for 17.6% of the adult population. This significant number is expected to increase in the coming years, pushing demand for regular glucose testing. Saudi Arabia has also seen a rise in diabetes-related complications, further increasing the need for frequent blood sugar monitoring. As a result, the consumption of glucose test consumables, including lancets, is expected to grow alongside the disease burden.
Self-care adoption and home healthcare trend
The trend toward self-care and home healthcare has significantly influenced the demand for lancet devices in Saudi Arabia. The Saudi government has actively promoted home-based healthcare solutions, especially in the context of chronic disease management. According to the Saudi Ministry of Health, there has been an increase in the number of home healthcare visits, reaching over 1.5 million cases annually as of 2023. This trend is closely tied to the rising number of patients managing chronic conditions like diabetes at home, further boosting demand for devices such as lancets that facilitate daily monitoring.
Market Challenges
Price sensitivity and cost constraints in procurement
The issue of price sensitivity continues to be a significant challenge for the lancets devices market in Saudi Arabia. Despite the growth of the healthcare sector, cost remains a crucial factor in the procurement process for both healthcare institutions and patients. The government, which plays a central role in healthcare procurement, faces rising costs in healthcare provision. In 2023, the Saudi Arabian government allocated approximately SAR 12.6 billion for healthcare funding, yet healthcare costs, including medical devices like lancets, continue to exert financial pressure on the system. Lower-income groups are particularly impacted by these rising costs, affecting the overall accessibility of self-testing devices.
Limited standardized device compatibility frameworks
A notable challenge in the lancet devices market is the lack of standardized compatibility frameworks across devices and brands. While the demand for self-testing solutions is increasing, a significant gap exists in device compatibility, particularly between lancets and glucose meters. This issue has led to consumer frustration and contributed to a lack of seamless integration between blood glucose meters and lancets. Despite advancements, the absence of regulatory standards that ensure universal compatibility among devices has hindered market growth. This lack of integration is acknowledged by both manufacturers and healthcare providers, who are pushing for more standardized solutions.
Market Opportunities
Safety lancets expansion
There is a notable opportunity for expansion in the safety lancets segment, driven by increasing consumer demand for safer and more user-friendly products. With the rise in diabetes cases, particularly among the elderly and children, the demand for lancets with advanced safety features such as automatic needle retraction and secure disposal is on the rise. According to the Saudi Ministry of Health, there is a growing emphasis on safety in medical devices due to rising concerns about infections and needle-stick injuries. As part of ongoing healthcare reforms, the government is encouraging the adoption of safety lancets to enhance patient safety. This trend reflects a broader shift toward improved healthcare standards in Saudi Arabia.
E-commerce growth for consumable devices
E-commerce is rapidly becoming a critical distribution channel for consumable devices like lancets in Saudi Arabia. The e-commerce market in the country has seen significant growth, with the total value of e-commerce transactions in Saudi Arabia reaching SAR 100 billion in 2023. The convenience of online shopping, combined with the increasing penetration of digital platforms and mobile health applications, is making it easier for consumers to purchase lancets and other diabetic care products. E-commerce offers competitive pricing and greater accessibility, especially in rural and underserved areas, providing a substantial growth opportunity for lancet manufacturers and distributors.
Future Outlook
Over the next 5 years, the KSA lancet devices market is expected to continue growing at a robust pace driven by factors such as an increasing diabetic population, advancements in lancet technologies, and a growing demand for home healthcare solutions. The government’s emphasis on healthcare improvements and expanding access to healthcare services, especially in rural areas, will also contribute to the market’s expansion. Additionally, the continuous innovation in lancet technology, particularly in terms of safety and ease of use, is expected to attract more consumers, further solidifying the market’s growth potential.
Major Players in the KSA Lancets Devices Market
- Becton Dickinson & Co
- F. Hoffmann-La Roche Ltd
- Braun SE
- Abbott Laboratories
- Owen Mumford Ltd
- Arkray Inc
- LifeScan Inc
- Ypsomed Holding AG
- Novo Nordisk A/S
- Sanofi
- Omron Healthcare
- Sinocare
- HemoCue AB
- Menarini Diagnostics
- Philips Healthcare
Key Target Audience
- Investments and Venture Capitalist Firms
- Government and Regulatory Bodies,(Saudi Food and Drug Authority, Ministry of Health)
- Healthcare Providers
- Hospitals and Clinics
- Retail Pharmacy Chains
- Self-Monitoring Patients/End Users
- Medical Device Distributors
- E-commerce Platforms (medical supplies)
Research Methodology
Step 1: Identification of Key Variables
The first phase involves mapping the key stakeholders involved in the KSA lancet devices market, including manufacturers, distributors, healthcare providers, and regulatory bodies. This phase involves collecting secondary data through industry reports, government publications, and online databases to identify the key market variables.
Step 2: Market Analysis and Construction
This phase will include compiling and analyzing historical data about lancet device penetration across different healthcare sectors. In-depth analysis of market dynamics, including supply-demand, competitive landscape, and consumer behavior, will be conducted.
Step 3: Hypothesis Validation and Expert Consultation
Hypotheses regarding market growth, segmentation trends, and technological shifts will be validated through consultations with key industry experts, including device manufacturers, healthcare providers, and distributors. These insights will further enhance the accuracy of our analysis.
Step 4: Research Synthesis and Final Output
Final market estimates will be developed by synthesizing data from primary and secondary research and validating them through industry feedback. This final output will provide a comprehensive overview of the market, backed by the most accurate and relevant data.
- Executive Summary
- Research Methodology (Market definitions and operational scope, lancets, lancing devices, safety lancets, disposable vs reusable, Data sources, primary interviews, industry panels, SFDA filings, Market sizing approach, bottom‑up by consumables volume and value, Forecasting model assumptions, demographic prevalence, device replacement cycles)
- Market definition and product taxonomy, (standard, safety, specialty, device compatibility)
- Disease burden context, (diabetes prevalence and testing frequency impact)
- Regional healthcare infrastructure and diagnostic penetration
- Distribution ecosystem, (hospital procurement, retail pharmacies, online retail)
- Supply chain flows and import dependencies
- Growth Drivers
Rising diabetes prevalence and testing frequency (glucose test consumables)
Self‑care adoption and home healthcare trend
Technology shifts (painless design/safety features) - Market Challenges
Price sensitivity and cost constraints in procurement
Limited standardized device compatibility frameworks
Counterfeit/lower‑quality imports concerns - Market Opportunities
Safety lancets expansion
E‑commerce growth for consumable devices
High‑end diabetic care programs - Market Trends
Shift to safety and low‑pain lancets
Product bundling with glucometers
Connected lancets and digital health integration - Regulatory and Standards Framework
SFDA regulatory pathways and compliance benchmarks
Quality certification and import licensing
Local manufacturing incentives
Competitive Environment
Porter’s Five Forces analysis (supplier power, buyer power, competitive rivalry)
SWOT Analysis (market‑wide)
- By Market Value (SAR/USD) by device category (standard, safety, specialty), 2020-2025
- By Market Volume (units) by lancet type and device compatibility, 2020-2025
- By Average Selling Price (ASP) analysis by channel and product category, 2020-2025
- By Consumables vs Device Bundling contribution, 2020-2025
- By Product Type (in value %)
Standard Lancets (single‑use)
Safety Lancets (built‑in safety guard)
Specialty Lancets (pediatric/low‑pain)
Reusable Lancing Devices (multi‑use holders)
Auto‑disable Lancets - By End‑User (in value %)
Hospitals & Clinics
Homecare & Self‑Monitoring
Diagnostic & Laboratory Centers
Retail Pharmacy Channel
Long‑Term Care Facilities - By Distribution Channel (in value %)
Medical Distributors/Wholesalers
Retail Pharmacy Chains
E‑commerce/Direct‑to‑Consumer
Hospital Group Procurement
Government Tenders - By Lancing Depth/Size (in value %)
Ultra‑Fine (31–33G)
Fine (28–30G)
Standard (25–27G)
Large (24G and below) - By Price Tier (in value %)
Economy/Generic
Mid‑Tier Branded
Premium/Advanced Technology
- Market share by value and volume – Top players
- Cross‑comparison parameters (Company overview, Business strategies and growth orientation, Product portfolio breadth (lancets & device compatibility), Safety/technology differentiation, Pricing and ASP benchmarks, Distribution reach and channel strength, Regulatory approvals and SFDA registrations, Manufacturing footprint and capacity)
- SWOT profiles of major competitors
- Pricing SKU structure and channel price dynamics
- Major Company Profiles
Becton Dickinson & Co (lancets & safety systems)
F. Hoffmann‑La Roche Ltd (glucose testing consumables integration)
B. Braun SE (lancing devices and consumables)
Abbott Laboratories (integrated diabetes devices)
Owen Mumford Ltd (lancet technology)
Arkray Inc (safety lancets)
LifeScan Inc (diabetes consumables)
AgaMatrix (monitor & lancet combos)
Ypsomed Holding AG (pen and lancet devices)
Novo Nordisk A/S (related consumables)
Sanofi (diabetes care portfolio)
Omron Healthcare (homecare devices)
Sinocare (cost‑competitive segment)
HemoCue AB (diagnostic interfaces)
Menarini Diagnostics (lab device synergy)
- Demand drivers by end‑user segment
- Purchase decision factors (price, quality, safety features)
- Adoption barriers and compliance requirements
- End‑user budget allocation patterns
- By Market Value, 2026-2035
- By Installed Units, 2026-2325
- By Average System Price, 2026-2035
- By System Complexity Tier, 2026-2035