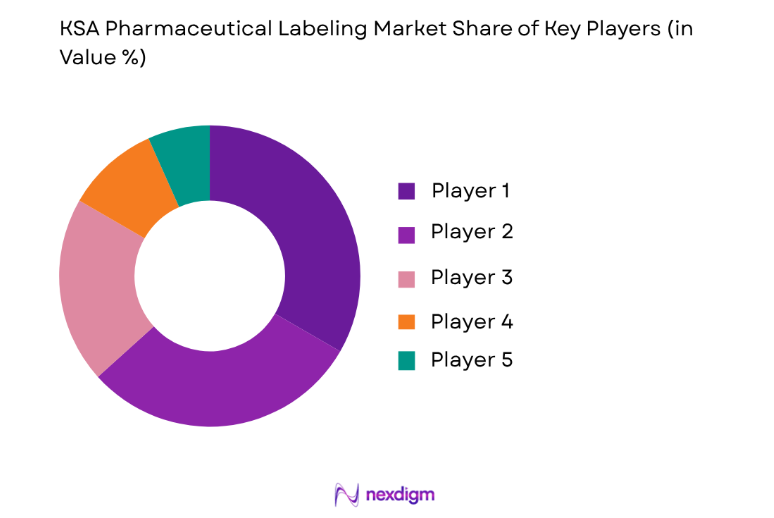
Market Overview
The KSA Pharmaceutical Labeling Market is witnessing significant growth and is estimated to reach USD ~ million, driven by increased demand for pharmaceutical products and rising regulatory requirements in the Kingdom. The market is poised to expand due to technological advancements in labeling systems, such as barcode and RFID solutions, which help enhance product tracking and safety. These advancements, combined with an increase in the adoption of eco-friendly labeling, are supporting market growth. Additionally, pharmaceutical companies are focusing on compliance with local regulations to meet industry standards, driving the need for more efficient labeling systems. This growth is further supported by the rising healthcare needs and pharmaceutical distribution in the region.
Saudi Arabia is the dominant player in the pharmaceutical labeling market in the Middle East, owing to its well-established healthcare infrastructure and strong regulatory environment. The country benefits from being a hub for pharmaceutical production and distribution, attracting multinational companies to establish their operations in the region. The Saudi Food and Drug Authority (SFDA) has also strengthened its regulations, enhancing the demand for compliant labeling solutions. Additionally, Saudi Arabia’s Vision 2030, which emphasizes healthcare and industrial growth, is expected to drive further investments in pharmaceutical manufacturing and labeling technologies, thereby boosting the market.
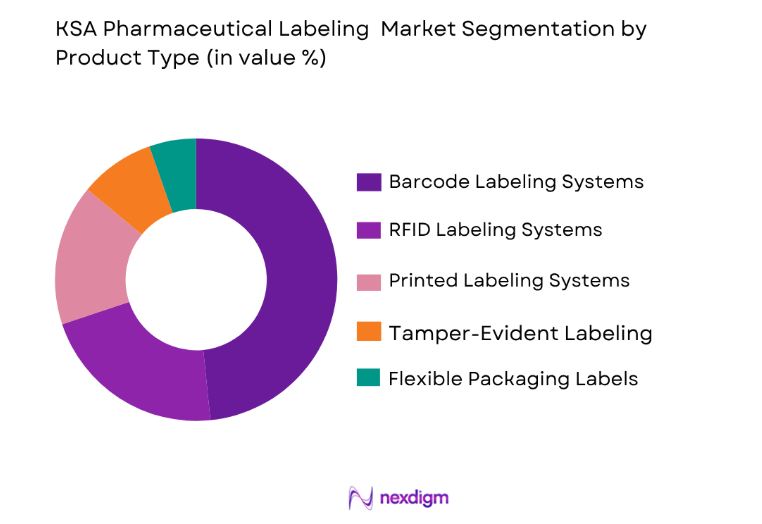
Market Segmentation
By Product Type:
The KSA Pharmaceutical Labeling Market is segmented by product type into several sub-segments. Recently, barcode labeling systems dominate the market due to their efficiency, ease of use, and cost-effectiveness. Barcode labeling is widely used across pharmaceutical manufacturers for tracking and identifying products, ensuring compliance with regulations. The growth in automated labeling solutions is also contributing to the dominance of this segment, as automation improves production efficiency. Additionally, barcode labels can integrate seamlessly with existing pharmaceutical production systems, providing a higher level of accuracy and reducing the risk of human error. With the ongoing advancement in labeling technology, barcode systems continue to play a crucial role in pharmaceutical labeling.
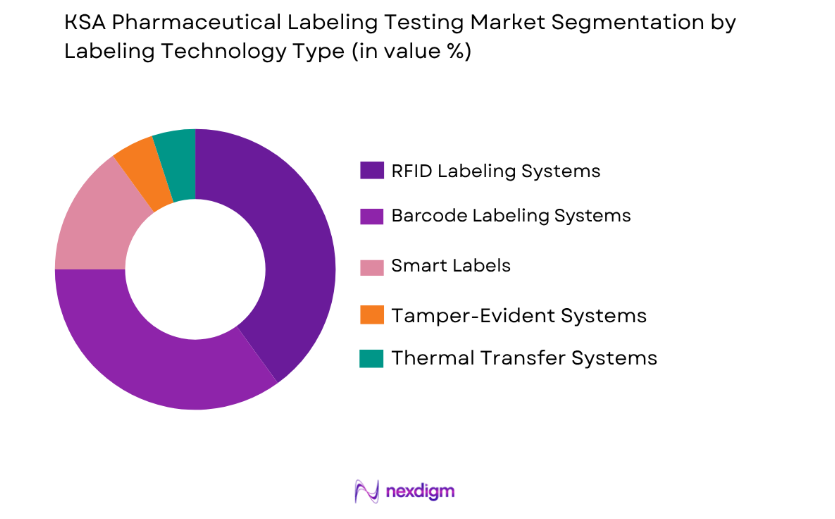
By Labeling Technology:
The KSA Pharmaceutical Labeling Market is also segmented by labeling technology, with RFID labeling systems currently leading the market. RFID technology offers significant advantages, such as real-time tracking and improved inventory management, making it highly beneficial for the pharmaceutical sector. RFID labels are crucial in ensuring the traceability of pharmaceutical products throughout the supply chain. The growing focus on combating counterfeit drugs and enhancing product security is driving the adoption of RFID systems. Furthermore, RFID tags provide additional features, such as temperature monitoring and authentication, which are essential for ensuring the safety and integrity of pharmaceuticals.
Competitive Landscape
The competitive landscape of the KSA Pharmaceutical Labeling Market is marked by consolidation and the dominance of several key players. Major international companies, along with regional manufacturers, are competing to meet the growing demand for advanced labeling solutions. These companies are focusing on expanding their market presence through strategic partnerships, acquisitions, and technological advancements. As regulatory requirements continue to evolve, these players are also investing in compliance-oriented labeling technologies to ensure their products meet the highest standards. The increasing emphasis on digital labeling technologies and eco-friendly solutions is likely to fuel further competition in the market.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Compliance Certifications |
| Zebra Technologies | 1969 | USA | ~ | ~ | ~ | ~ | ~ |
| Sato Holdings | 1940 | Japan | ~ | ~ | ~ | ~ | ~ |
| Avery Dennison Corporation | 1908 | USA | ~ | ~ | ~ | ~ | ~ |
| Videojet Technologies | 1986 | USA | ~ | ~ | ~ | ~ | ~ |
| Markem-Imaje | 2007 | France | ~ | ~ | ~ | ~ | ~ |
KSA Pharmaceutical Labeling Market Analysis
Growth Drivers
Technological Advancements in Labeling:
The KSA pharmaceutical labeling market has experienced substantial growth due to technological innovations in labeling systems, such as barcode and RFID solutions. These advancements have revolutionized the way pharmaceutical companies manage their supply chains, ensuring better traceability, security, and compliance with stringent regulatory requirements. RFID, in particular, has become increasingly popular due to its ability to provide real-time tracking and enhance product security. As pharmaceutical companies focus on improving their operational efficiency, the adoption of advanced labeling technologies that integrate with existing systems and provide higher levels of automation has been critical. Automation not only reduces human error but also increases the speed of production, which is vital for meeting the growing demand for pharmaceutical products. In addition, the rising adoption of digital and smart labeling solutions has opened up new opportunities for providing enhanced safety and customer engagement features. These innovations contribute significantly to the growth of the market, allowing companies to meet evolving regulatory standards while increasing operational efficiency.
Regulatory Compliance Requirements:
The increasing number of stringent regulations imposed by governments and international bodies is driving the growth of the KSA pharmaceutical labeling market. Saudi Arabia, through the Saudi Food and Drug Authority (SFDA), has implemented regulations that mandate specific labeling standards for pharmaceutical products, ensuring their safety and authenticity. This includes guidelines on labeling content, packaging, and traceability. These regulatory requirements have fueled the demand for accurate and secure labeling solutions, such as tamper-evident labels and anti-counterfeiting technologies, that help prevent the sale of counterfeit drugs in the market. As the regulatory environment becomes more complex, pharmaceutical companies must adopt advanced labeling systems that comply with these standards. The need for compliance-driven labeling solutions has led to a rise in demand for labeling technologies that offer greater accuracy and efficiency, such as RFID and digital printing systems.
Market Challenges
High Initial Investment Costs:
One of the key challenges in the KSA pharmaceutical labeling market is the high initial investment required for advanced labeling technologies. Although systems like RFID and smart labels offer numerous benefits, including enhanced product tracking and security, the upfront costs of implementing these technologies can be prohibitive for some companies. Additionally, the maintenance costs of these systems can also be significant. Smaller pharmaceutical manufacturers or distributors, particularly those in the early stages of development, may find it difficult to justify the expense of upgrading their labeling systems. This can hinder the adoption of these advanced technologies, particularly in a market where cost-efficiency is a critical factor. Despite the long-term benefits of automated and smart labeling systems, the initial cost remains a barrier for many companies, potentially limiting the growth of the market. Overcoming this challenge will require collaboration between technology providers and pharmaceutical companies to reduce costs and make advanced labeling technologies more accessible.
Counterfeit Drugs and Security Concerns:
Counterfeit drugs continue to pose a significant challenge in the pharmaceutical industry, and the KSA market is no exception. Counterfeit drugs not only undermine the reputation of pharmaceutical companies but also pose serious health risks to patients. As a result, there is increasing pressure on pharmaceutical companies to adopt more robust anti-counterfeit labeling solutions, such as tamper-evident and RFID-based systems. However, the effectiveness of these solutions is often dependent on the ability of companies to integrate new technologies with their existing production systems, which can be challenging. In addition, the growing complexity of global supply chains has made it more difficult to monitor and ensure the authenticity of pharmaceutical products. As regulatory bodies continue to strengthen requirements related to anti-counterfeit measures, the need for more secure labeling systems will increase. This challenge underscores the need for continuous innovation and investment in advanced labeling solutions to stay ahead of counterfeiters.
Opportunities
Eco-friendly Labeling Solutions:
As environmental concerns continue to grow, there is a significant opportunity in the KSA pharmaceutical labeling market for the development of eco-friendly labeling solutions. With increasing awareness of sustainability, consumers and regulatory bodies are pushing for greener alternatives in packaging and labeling. Pharmaceutical companies are under pressure to reduce their environmental footprint, leading to a rise in demand for biodegradable, recyclable, and eco-friendly labels. Additionally, the use of environmentally friendly materials in labeling systems, such as paper-based labels or plant-based inks, offers a competitive advantage in a market where corporate social responsibility is becoming a key consideration. Pharmaceutical companies that adopt sustainable labeling solutions will not only comply with growing environmental regulations but also improve their brand image among environmentally conscious consumers. This presents a major opportunity for labeling solution providers to innovate and offer more sustainable products that align with the industry’s shifting priorities.
Growth of E-commerce and Direct-to-Consumer Sales:
The rise of e-commerce in the pharmaceutical industry presents a new opportunity for labeling companies in Saudi Arabia. As more pharmaceutical products are sold online, the need for effective labeling solutions that can withstand the challenges of direct-to-consumer sales increases. E-commerce platforms require packaging that ensures the integrity and safety of pharmaceutical products during transit, making tamper-evident and traceable labeling solutions essential. The growing trend of online pharmaceutical sales has spurred demand for packaging and labeling that not only ensures product safety but also enhances customer experience through features like personalized labels and digital interactions. Companies that can offer innovative, user-friendly labeling solutions that cater to the growing e-commerce sector will benefit from increased demand and market growth. This trend presents a significant opportunity for labeling providers to expand their customer base and diversify their product offerings.
Future Outlook
The KSA pharmaceutical labeling market is poised for substantial growth over the next five years, driven by the increasing demand for advanced labeling solutions that meet regulatory requirements and consumer expectations. As the Saudi pharmaceutical sector expands, there will be a strong emphasis on automation and digital technologies, with RFID, barcode, and smart labels becoming the standard. The regulatory environment will continue to evolve, demanding higher levels of accuracy and traceability in pharmaceutical labeling. Technological advancements, such as eco-friendly labels and anti-counterfeit solutions, will play a key role in shaping the future of the market. With government support for the healthcare sector and Vision 2030 driving industrial growth, the market will witness significant investments in innovative labeling solutions. This progress is expected to be accompanied by increased competition, making the market more dynamic and accessible.
Major Players
- Zebra Technologies
- Sato Holdings
- Avery Dennison Corporation
- Videojet Technologies
- Markem-Imaje
- Label Tech
- TSC Auto ID Technology
- Brother Industries
- UPM Raflatac
- Amcor Limited
- Seiko Instruments Inc.
- ScanSource
- Toshiba TEC Corporation
- PTC Inc.
- Systech International
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Pharmaceutical manufacturers
- Contract packaging companies
- Healthcare institutions
- Pharmaceutical distributors
- E-commerce platforms selling pharmaceutical products
- Retailers and wholesalers
Research Methodology
Step 1: Identification of Key Variables
This step involves defining the scope of the market, identifying key parameters, and determining the research variables that affect the market’s dynamics.
Step 2: Market Analysis and Construction
In this phase, primary and secondary data sources are analyzed, and a comprehensive market model is developed to evaluate market trends, growth drivers, and segmentation.
Step 3: Hypothesis Validation and Expert Consultation
Consultation with industry experts and key stakeholders is conducted to validate assumptions, ensuring that the market model reflects current market conditions and future projections.
Step 4: Research Synthesis and Final Output
All collected data is synthesized to create a detailed market report, including actionable insights, market forecasts, and strategic recommendations.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Growth Drivers
Increasing Government Regulations on Labeling Standards
Rising Demand for Pharmaceutical Products
Technological Advancements in Labeling Solutions - Market Challenges
Stringent Regulatory Compliance Requirements
High Initial Investment Costs
Counterfeit Products and Labeling Fraud - Market Opportunities
Rising Demand for Eco-Friendly Labels
Adoption of Smart Labels and Digital Solutions
Growing Adoption of Pharmaceutical Packaging Automation - Trends
Shift Towards Eco-friendly Labeling Solutions
Increase in Adoption of QR Codes and Smart Labels
Technological Integration in Pharmaceutical Labeling - Government Regulations
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Barcode Labeling Systems
RFID Labeling Systems
Printed Labeling Systems
Tamper-Evident Labeling Systems
Flexible Packaging Labeling Systems - By Platform Type (In Value%)
Manual Labeling Systems
Automated Labeling Systems
Semi-Automated Labeling Systems
Integrated Labeling Systems
Digital Labeling Systems - By Fitment Type (In Value%)
Adhesive Labels
Shrink Sleeve Labels
Pressure Sensitive Labels
In-Mold Labels
Stretch Sleeves Labels - By EndUser Segment (In Value%)
Pharmaceutical Manufacturers
Contract Packaging Companies
Pharmaceutical Distributors
Hospitals & Healthcare Institutions
Retailers & Wholesalers
- Market Share Analysis
- CrossComparison Parameters (System Type, Platform Type, Procurement Channel, EndUser Segment, Fitment Type)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Porter’s Five Forces
- Key Players
Sato Holdings Corporation
Zebra Technologies Corporation
Toshiba TEC Corporation
Avery Dennison Corporation
Honeywell International Inc.
Markem-Imaje
Videojet Technologies Inc.
Jet 3M
Quad/Graphics, Inc.
Herma GmbH
The Mérieux Group
Laetus GmbH
Cognex Corporation
Krones AG
Dai Nippon Printing Co., Ltd.
- Demand from Pharmaceutical Manufacturers for Innovative Labeling Solutions
- Growth in Contract Packaging Services in KSA
- Increased Focus on Regulatory Compliance among Healthcare Providers
- Retailers Demanding Consumer-Friendly Pharmaceutical Labels
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035