Market Overview
The KSA Predictive Biomarkers market is valued at USD ~ billion, experiencing steady growth, largely driven by advancements in genomics, proteomics, and biotechnology. These fields have fueled demand for predictive biomarkers used in personalized medicine and diagnostics. As technology continues to improve, healthcare providers are increasingly adopting predictive biomarkers to enable earlier disease detection and better patient outcomes. The Saudi government’s investments in healthcare infrastructure and research also contribute to this growth, bolstering the market’s potential for expansion in the coming years.
Key regions such as Riyadh, Jeddah, and Dhahran are pivotal in the KSA Predictive Biomarkers market due to their robust healthcare infrastructure and the presence of leading medical research centers. These cities dominate the market owing to their proximity to government-backed healthcare initiatives and the growing adoption of advanced medical technologies. Moreover, the strategic focus on healthcare modernization under Saudi Arabia’s Vision 2030 has made these cities significant hubs for medical innovation, fostering an environment conducive to the rapid adoption of predictive biomarkers.
Market Segmentation
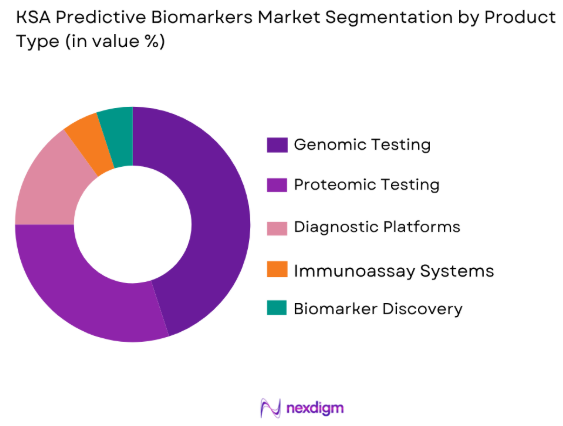
By Product Type
The KSA Predictive Biomarkers market is segmented by product type into genomic testing, proteomic testing, and diagnostic platforms. Recently, genomic testing has captured a dominant market share due to factors such as the increasing prevalence of genetic disorders, the growing demand for precision medicine, and the advancement of sequencing technologies. Genomic testing offers personalized insights into genetic predispositions and is extensively used for cancer screening, infectious diseases, and rare genetic conditions. The growing availability of genetic data, improved testing accuracy, and supportive government policies have fueled the growth of this sub-segment in Saudi Arabia.
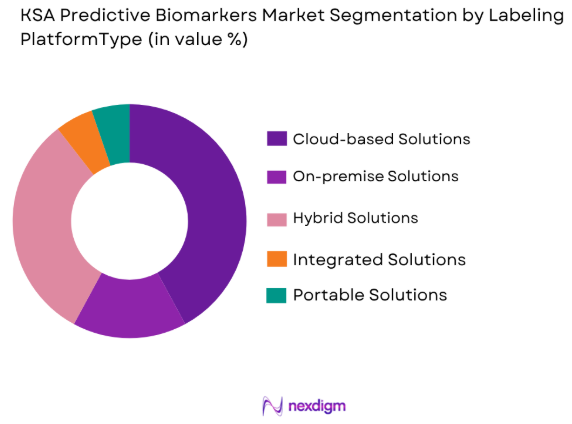
By Platform Type
The market is also segmented by platform type into on-premise solutions, cloud-based solutions, and hybrid systems. Cloud-based solutions have recently dominated the market share due to their cost-effectiveness, scalability, and flexibility. These solutions are increasingly popular among healthcare providers due to their ability to store large datasets securely and offer real-time access to predictive biomarker analysis. The growing adoption of electronic health records and the need for data-sharing across institutions are key drivers of the cloud-based sub-segment’s growth in KSA.
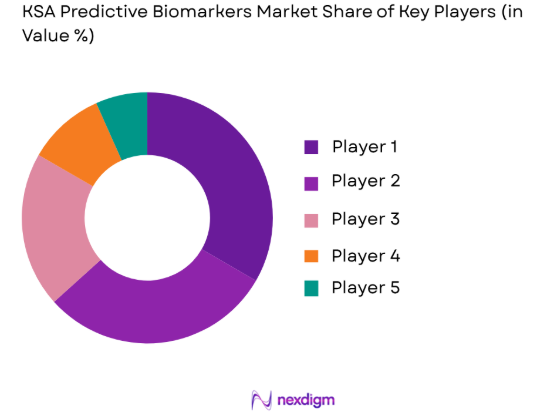
Competitive Landscape
The KSA Predictive Biomarkers market is competitive, with a mix of local and international players striving for a stake in this rapidly evolving sector. Consolidation trends are observed as leading healthcare companies integrate predictive biomarker technologies into their service offerings. Major players in the market are significantly influencing growth, investing in R&D, and forming strategic partnerships with local healthcare providers and research institutions.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD) | Additional Market Parameter |
| Illumina | 1998 | San Diego, USA | ~ | ~ | ~ | ~ | ~ |
| Thermo Fisher | 1956 | Waltham, USA | ~ | ~ | ~ | ~ | ~ |
| Qiagen | 1984 | Hilden, Germany | ~ | ~ | ~ | ~ | ~ |
| Abbott Laboratories | 1888 | Abbott Park, USA | ~ | ~ | ~ | ~ | ~ |
| Merck & Co. | 1891 | Kenilworth, USA | ~ | ~ | ~ | ~ | ~ |
KSA Predictive Biomarkers Market Analysis
Growth Drivers
Technological Advancements in Predictive Biomarkers
Technological advancements in predictive biomarkers are driving the market’s expansion in KSA. With rapid developments in genomic and proteomic testing technologies, biomarkers are increasingly being used to predict disease risk, assess therapeutic efficacy, and monitor patient responses to treatments. The rising integration of artificial intelligence (AI) and machine learning (ML) into these technologies is further enhancing the accuracy and speed of biomarker analysis. These technological improvements enable healthcare professionals to offer more personalized, efficient treatments, thus improving patient outcomes. In Saudi Arabia, investments in medical research, along with growing interest in precision medicine, have created a conducive environment for the growth of predictive biomarkers. As the availability of these advanced technologies increases, healthcare providers are increasingly adopting them to support early disease detection, particularly in oncology and rare genetic conditions. Furthermore, collaborations between local hospitals and global biotech companies are accelerating the development of new biomarkers, further supporting the technological advancement of this market segment.
Government Support and Healthcare Investment
Government initiatives play a crucial role in the growth of the KSA Predictive Biomarkers market. Saudi Arabia’s Vision 2030 aims to transform the healthcare system by enhancing the quality and accessibility of medical services. Increased government funding for healthcare infrastructure and research is expected to drive the development and adoption of advanced diagnostic tools, including predictive biomarkers. The government has been actively promoting partnerships between the public and private sectors to encourage innovation in medical technologies, thus improving the availability of cutting-edge healthcare solutions. Additionally, the establishment of specialized medical centers and research facilities has strengthened the country’s position as a hub for medical advancements. These investments in healthcare modernization are essential for enabling the widespread use of predictive biomarkers across various disease areas, particularly in the diagnosis of cancer, cardiovascular diseases, and neurological disorders. The supportive regulatory framework and continuous government investments ensure that Saudi Arabia remains at the forefront of healthcare technology development in the region.
Market Challenges
High Cost of Implementation
The high cost associated with the adoption of predictive biomarker technologies remains a significant challenge in the KSA market. Advanced genomic and proteomic testing platforms require substantial capital investment, which can be a barrier for smaller healthcare providers, especially those in less urbanized regions. Moreover, the operational costs for maintaining these high-tech systems, including training staff and ensuring data security, can further increase the financial burden on healthcare facilities. Despite the long-term cost savings and improved patient outcomes that predictive biomarkers offer, the initial investment required for adopting such technologies may deter smaller institutions from integrating them into their services. As a result, there is a need for government-backed funding and more cost-effective solutions to support the adoption of predictive biomarkers in Saudi Arabia. The lack of financial resources, especially in public healthcare institutions, limits the market’s potential to expand at a faster pace.
Regulatory and Certification Barriers
Regulatory and certification processes present another challenge to the widespread use of predictive biomarkers in Saudi Arabia. As new diagnostic technologies and platforms emerge, they must undergo rigorous testing and certification procedures to meet local healthcare standards. These processes can be time-consuming and costly, delaying the market entry of innovative biomarker testing technologies. The complexity of regulatory frameworks can also create uncertainty for companies looking to enter the market, particularly international firms unfamiliar with local requirements. Additionally, compliance with international standards, such as ISO certifications, is mandatory for medical devices and diagnostic equipment, further adding to the regulatory burden. The lengthy approval process can slow down the introduction of new predictive biomarkers, hindering market growth and innovation in the country.
Opportunities
Rising Demand for Personalized Medicine
One of the most significant opportunities in the KSA Predictive Biomarkers market lies in the rising demand for personalized medicine. As healthcare becomes increasingly focused on individualized treatment plans, predictive biomarkers are playing a crucial role in identifying the most effective therapies for patients. Personalized medicine allows for more targeted treatments, improving patient outcomes and reducing the likelihood of adverse reactions. The growing prevalence of chronic diseases, such as cancer, diabetes, and cardiovascular conditions, has fueled the demand for more tailored healthcare solutions. In Saudi Arabia, the government’s focus on developing a personalized healthcare system aligns with this trend, offering a substantial opportunity for the predictive biomarkers market. By integrating advanced biomarker testing into clinical practices, Saudi healthcare providers can improve diagnosis accuracy and offer customized treatment plans to patients, further driving market growth.
Expansion of Research Partnerships
The expansion of research partnerships presents another significant opportunity for the KSA Predictive Biomarkers market. Collaborations between local healthcare providers, academic institutions, and international biotech companies are accelerating the development of new and more accurate predictive biomarkers. These partnerships allow for the sharing of expertise, research data, and financial resources, facilitating faster innovation in biomarker technologies. Additionally, government-backed research initiatives are enabling the growth of a supportive ecosystem for medical advancements. As the demand for predictive biomarkers in disease diagnosis and treatment monitoring continues to rise, these research collaborations are vital in pushing the boundaries of what is possible in the field. By fostering a collaborative environment, Saudi Arabia can strengthen its position as a leader in the global predictive biomarkers market.
Future Outlook
The future of the KSA Predictive Biomarkers market looks promising, with strong growth expected over the next five years. Key drivers such as technological advancements in genomic and proteomic testing, government support for healthcare modernization, and the rising demand for personalized medicine will continue to propel the market forward. Additionally, the increasing prevalence of chronic diseases, along with the shift toward more efficient and accurate diagnostic tools, will further fuel demand for predictive biomarkers. With the continued evolution of medical research and innovation, the KSA market is well-positioned to embrace new breakthroughs, further expanding its potential.
Major Players
- Illumina
- ThermoFisher
- Qiagen
- Abbott Laboratories
- Merck & Co.
- Roche Diagnostics
- Bio-Rad Laboratories
- Siemens Healthineers
- Agilent Technologies
- Beckman Coulter
- PerkinElmer
- GE Healthcare
- Becton Dickinson
- Luminex Corporation
- Cepheid
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Pharmaceutical companies
- Biotechnology firms
- Healthcare providers
- Diagnostic laboratories
- Research institutions
- Medical device manufacturers
Research Methodology
Step 1: Identification of Key Variables
Identifying the critical factors influencing market dynamics, such as technological developments, regulatory trends, and industry challenges.
Step 2: Market Analysis and Construction
Assessing the market’s current state and constructing a comprehensive model based on primary and secondary data sources.
Step 3: Hypothesis Validation and Expert Consultation
Validating initial hypotheses through consultation with industry experts, stakeholders, and subject-matter specialists.
Step 4: Research Synthesis and Final Output
Compiling findings into a final output, ensuring the accuracy and validity of all market insights.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Government Support and Healthcare Investment
Technological Advancements in Biomarker Testing - Market Challenges
High Cost of Diagnostic Systems
Regulatory Hurdles and Compliance Issues - Market Opportunities
Expansion of Genomic and Proteomic Research
Rising Demand for Personalized Medicine - Trends
Shift towards multi-omics and integrated diagnostic approaches
Adoption of liquid biopsy technologies for non-invasive testing - Government Regulations
- SWOT Analysis
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By Product Type (In Value%)
Genomic Testing
Proteomic Testing
Diagnostic Platforms
Immunoassay Systems
Biomarker Discovery - By System Type (In Value%)
Biomarker Discovery Systems
Genomic Testing Systems
Proteomic Testing Systems
Diagnostic Platforms Immunoassay Systems - By Platform Type (In Value%)
On-premise Solutions
Cloud-based Solutions
Hybrid Solutions
Portable Solutions
Integrated Solutions - By End User Segment (In Value%)
Hospitals
Clinics
Research Institutions
Pharmaceutical Companies
Biotechnology Firms - By Procurement Channel (In Value%)
Direct Procurement
Government Tenders
Private Sector Procurement
Online Bidding Platforms
Third-party Distributors
- Market Share Analysis
- Cross Comparison Parameters (Biomarker Discovery, Genomic Testing, Proteomic Testing, Diagnostic Platforms, Immunoassay Systems, Technology Innovation, Product Accuracy, Regulatory Compliance, Turnaround Time, Cost Competitiveness, Market Presence, Partnerships & Collaborations, Automation Capacity, Data Analytics Support, After-sales Service)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Illumina
Thermo Fisher Scientific
Bio-Rad Laboratories
Roche Diagnostics
Qiagen
Abbott Laboratories
Merck & Co.
Agilent Technologies
Beckman Coulter
GE Healthcare
PerkinElmer
Siemens Healthineers
BD Biosciences
Luminex Corporation
Cepheid
- Hospitals Increasing Adoption of Predictive Testing
- Research Institutions Focusing on Disease Biomarkers
- Pharmaceutical Companies Integrating Biomarkers for Drug Development
- Biotechnology Firms Advancing Personalized Medicine Solutions
- By Market Value, 2026-2035
- By Installed Units, 2026-2035
- By Average System Price, 2026-2035
- By System Complexity Tier, 2026-2035