Market Overview
The KSA Prognostic Biomarkers Market is poised to witness significant growth, driven by advancements in diagnostic technologies and the increasing adoption of personalized medicine. Based on a recent historical assessment, the market size for prognostic biomarkers in KSA is valued at approximately USD ~ billion. The growth of this market is fueled by the expanding healthcare infrastructure, government investments in healthcare, and the increasing demand for early-stage disease diagnosis, particularly in oncology and cardiovascular diseases. These factors are expected to continue propelling the market’s expansion in the coming years.
Dominant regions in the KSA Prognostic Biomarkers Market include Riyadh, Jeddah, and Dammam. These cities are prominent due to their strong healthcare infrastructure, research capabilities, and medical tourism appeal. Riyadh leads in medical research and the development of advanced diagnostic tools, while Jeddah is a key player in private healthcare services. Dammam has seen a surge in demand due to growing investments in healthcare facilities. The presence of top-tier hospitals and research centers in these cities further strengthens their dominance in the market.
Market Segmentation
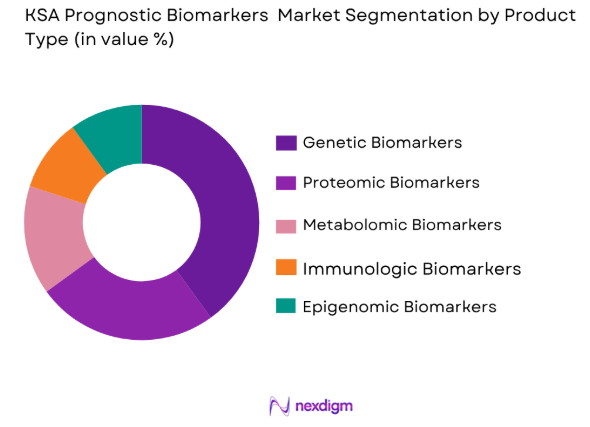
By Product Type
The KSA Prognostic Biomarkers Market is segmented by product type into genetic biomarkers, proteomic biomarkers, metabolomic biomarkers, immunologic biomarkers, and epigenomic biomarkers. Recently, genetic biomarkers have held the largest market share due to the increasing prevalence of genetic disorders and the growing adoption of genomics in personalized medicine. This sub-segment is driving growth due to advancements in genetic sequencing technologies and the increasing focus on genetic testing for early diagnosis and targeted treatment. The demand for genetic biomarkers has been further accelerated by the adoption of liquid biopsy techniques and the increasing prevalence of cancers, particularly breast, lung, and colorectal cancer, which are associated with genetic mutations.
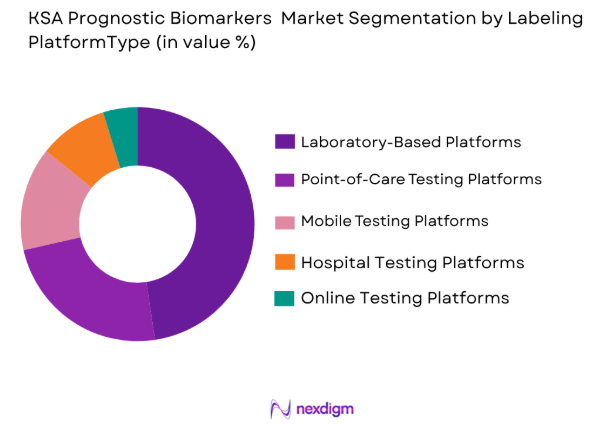
By Platform Type
The KSA Prognostic Biomarkers Market is segmented by platform type into laboratory-based platforms, point-of-care testing platforms, mobile testing platforms, hospital testing platforms, and online testing platforms. Laboratory-based platforms are the leading segment, driven by their high accuracy, reliability, and adoption in clinical settings. These platforms allow for complex testing and the generation of precise diagnostic results. The growing preference for personalized medicine and advancements in diagnostic technologies further enhance the demand for laboratory-based platforms. Point-of-care testing platforms are also gaining traction, offering quicker turnaround times and increasing patient accessibility, particularly for high-risk groups.
Competitive Landscape
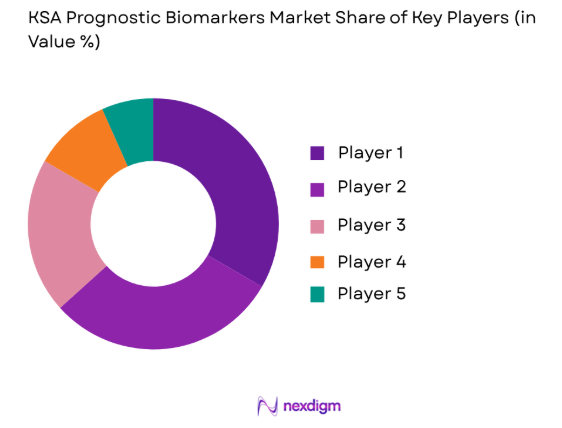
The competitive landscape in the KSA Prognostic Biomarkers Market is shaped by the presence of several leading players in the healthcare and diagnostic sectors. These companies are engaged in consolidating their market positions through strategic partnerships, mergers, and acquisitions. Major players are focusing on technological advancements, regulatory compliance, and expanding their product portfolios to cater to the growing demand for prognostic biomarker solutions. Their efforts are complemented by investments from both public and private sectors, ensuring a competitive and dynamic market environment.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Additional Parameter |
| Thermo Fisher Scientific | 1956 | Waltham, MA, USA | ~ | ~ | ~ | ~ | ~ |
| Roche Diagnostics | 1896 | Basel, Switzerland | ~ | ~ | ~ | ~ | ~ |
| Abbott Laboratories | 1888 | Chicago, IL, USA | ~ | ~ | ~ | ~ | ~ |
| Qiagen | 1984 | Hilden, Germany | ~ | ~ | ~ | ~ | ~ |
| PerkinElmer | 1937 | Waltham, MA, USA | ~ | ~ | ~ | ~ | ~ |
KSA Prognostic Biomarkers Market Analysis
Growth Drivers
Advancements in Genomic Technologies
The growth of the KSA Prognostic Biomarkers Market is primarily driven by significant advancements in genomic technologies. Breakthroughs in genetic sequencing, such as next-generation sequencing (NGS), have made genetic testing more accurate, affordable, and accessible. This has opened up new opportunities for early diagnosis and personalized treatment, particularly for cancer, cardiovascular diseases, and genetic disorders. Additionally, the growing emphasis on precision medicine has led to an increased demand for diagnostic tools that can analyze genetic variations, fueling the market’s expansion. As healthcare providers and researchers increasingly adopt genomic testing to identify biomarkers, the market for genomic-based prognostic biomarkers continues to grow. The ability to identify genetic mutations and predispositions allows for early intervention and tailored therapies, which significantly improves patient outcomes. Furthermore, the integration of artificial intelligence and machine learning in genomics is streamlining the analysis of genetic data, making it easier to identify biomarkers associated with disease progression and treatment response. This technological evolution is expected to accelerate the adoption of prognostic biomarkers in clinical practice, contributing to the overall growth of the market.
Increasing Healthcare Investments in KSA
The growing government and private sector investments in healthcare infrastructure in Saudi Arabia are significantly contributing to the market’s growth. The KSA Vision 2030 initiative, which aims to modernize the healthcare system and improve the quality of medical services, has led to substantial investments in healthcare facilities, medical research, and diagnostic technologies. The government has been actively promoting healthcare research and development, leading to increased demand for advanced diagnostic solutions, including prognostic biomarkers. This has resulted in the establishment of state-of-the-art diagnostic centers and the expansion of research institutes focused on biomarker discovery. Additionally, the rising focus on medical tourism in Saudi Arabia is further boosting the demand for advanced diagnostic services, including predictive and prognostic testing. The expansion of healthcare infrastructure, along with government incentives for innovation in diagnostics, is expected to create a conducive environment for the growth of the prognostic biomarkers market.
Market Challenges
High Costs of Diagnostic Testing
One of the major challenges facing the KSA Prognostic Biomarkers Market is the high cost associated with advanced diagnostic testing, particularly for genetic and molecular biomarkers. The process of developing and implementing prognostic biomarker tests involves significant research and development expenses, as well as the need for specialized laboratory equipment and highly trained personnel. These costs are often passed on to patients, making these tests less accessible, particularly in low-income segments. While the cost of diagnostic testing has decreased over the years, it remains a significant barrier to widespread adoption, especially in a market where the healthcare system is still developing. In addition, the reimbursement rates for biomarker-based tests are often low, further limiting the affordability of these services. The challenge of high costs is particularly prominent in the private healthcare sector, where patients are required to bear a larger portion of the expenses. Overcoming this challenge will require efforts to reduce the cost of testing through innovation, government intervention, and healthcare reforms that focus on improving accessibility and affordability.
Regulatory and Standardization Challenges
The KSA Prognostic Biomarkers Market also faces challenges related to regulatory hurdles and the lack of standardized protocols for the approval of prognostic biomarker tests. While Saudi Arabia has made significant progress in establishing regulatory frameworks for medical devices and diagnostic tests, the approval process for biomarker-based diagnostic solutions remains complex and time-consuming. The absence of standardized guidelines for biomarker validation and approval adds to the uncertainty and delays in bringing new diagnostic solutions to the market. Furthermore, there are varying levels of regulation and oversight across different regions within Saudi Arabia, which complicates the process for manufacturers looking to enter the market. These regulatory challenges can hinder the timely adoption of new technologies and limit the market’s growth potential. To overcome these obstacles, regulatory bodies in KSA will need to develop clearer guidelines for the validation and approval of biomarker tests, as well as streamline the approval process to support innovation and market entry.
Opportunities
Expansion of Personalized Medicine
One of the most significant opportunities for the KSA Prognostic Biomarkers Market is the expansion of personalized medicine, which relies heavily on the use of biomarkers for early detection and treatment planning. Personalized medicine aims to tailor healthcare treatments to individual patients based on their genetic makeup, lifestyle, and other factors. The increasing adoption of personalized treatment plans in oncology, cardiology, and genetic disorders presents a major opportunity for the growth of the biomarker market. As the healthcare system in Saudi Arabia shifts towards more patient-centric care, the demand for predictive biomarkers to identify disease risk, monitor treatment response, and guide therapy choices is expected to rise. Moreover, the integration of genomic data with electronic health records (EHR) systems will enable healthcare providers to offer more precise and individualized care. The growing focus on personalized medicine is expected to increase the demand for advanced biomarker testing, driving market growth.
Technological Innovations in Liquid Biopsy
Another significant opportunity lies in the development and adoption of liquid biopsy technologies, which offer a non-invasive method for detecting biomarkers in blood samples. Liquid biopsy is rapidly gaining popularity in the early detection and monitoring of various cancers, including lung, breast, and colorectal cancer. This technology is expected to revolutionize the way diagnostic tests are performed, offering several advantages over traditional biopsy methods, including reduced patient discomfort, lower costs, and faster results. Liquid biopsy also allows for the monitoring of disease progression and treatment response in real time, providing valuable insights for personalized treatment plans. As the technology continues to improve, liquid biopsy is expected to play a crucial role in the KSA Prognostic Biomarkers Market. With increased investment in research and development, Saudi Arabia is well-positioned to capitalize on this innovative technology, driving the growth of the biomarker market in the region.
Future Outlook
The KSA Prognostic Biomarkers Market is expected to experience steady growth over the next five years, driven by technological advancements, increased healthcare investments, and the growing demand for personalized medicine. Innovations in genetic testing and liquid biopsy technologies are expected to enhance diagnostic capabilities, while regulatory support for the adoption of these technologies will foster market expansion. Furthermore, government initiatives aimed at improving healthcare infrastructure will create favorable conditions for the continued growth of prognostic biomarkers, with a particular focus on cancer and genetic disorders.
Major Players
- ThermoFisher Scientific
- Roche Diagnostics
- Abbott Laboratories
- Qiagen
- PerkinElmer
- Illumina
- Bio-Rad Laboratories
- Siemens Healthineers
- GE Healthcare
- Agilent Technologies
- Danaher Corporation
- Abbott Laboratories
- Becton Dickinson
- Merck & Co.
- Johnson & Johnson
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Healthcare providers and hospitals
- Pharmaceutical companies
- Diagnostic laboratories
- Research and development institutions
- Biotech firms
- Medical device manufacturers
Research Methodology
Step 1: Identification of Key Variables
Identification of key variables such as biomarkers, market drivers, technology adoption, and healthcare infrastructure that influence market growth.
Step 2: Market Analysis and Construction
Analysis of market data from primary and secondary sources to develop market models, forecast trends, and segment the market based on technology, product types, and applications.
Step 3: Hypothesis Validation and Expert Consultation
Consultation with industry experts and key stakeholders to validate hypotheses, refine market models, and ensure accuracy in forecasts.
Step 4: Research Synthesis and Final Output
Synthesis of findings into a comprehensive report that includes detailed market insights, trends, and forecasts, ensuring that all findings are supported by credible data.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Rising prevalence of chronic diseases
Technological advancements in biomarker research
Growing demand for personalized medicine - Market Challenges
High costs of diagnostic tests
Regulatory hurdles in biomarker approval
Lack of standardized protocols for biomarker testing - Market Opportunities
Expansion of healthcare infrastructure in KSA
Growing healthcare investments from government
Advancements in genomic research - Trends
Integration of artificial intelligence in biomarker analysis
Increased use of liquid biopsy technologies
Growing adoption of wearable biomarker detection devices - Government Regulations
- SWOT Analysis of Key Competitors
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Genetic Biomarkers
Proteomic Biomarkers
Metabolomic Biomarkers
Immunologic Biomarkers
Epigenomic Biomarkers - By Platform Type (In Value%)
Laboratory-based Platforms
Point-of-Care Platforms
Microarray Platforms
Mass Spectrometry Platforms
Next-Generation Sequencing Platforms - By Fitment Type (In Value%)
On-premise Solutions
Cloud-based Solutions
Hybrid Solutions
Integrated Solutions
Modular Solutions - By End User Segment (In Value%)
Hospitals and Clinics
Research Institutes
Biopharmaceutical Companies
Diagnostics Laboratories
Contract Research Organizations (CROs) - By Procurement Channel (In Value%)
Direct Procurement
Government Tenders
Private Sector Procurement
Online Bidding Platforms
Third-party Distributors
- Market Share Analysis
- Cross Comparison Parameters (System Type, Platform Type, End User Segment, Procurement Channel, Fitment Type)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Illumina
Thermo Fisher Scientific
Roche Diagnostics
Abbott Laboratories
Qiagen
PerkinElmer
Agilent Technologies
Bio-Rad Laboratories
GE Healthcare
Siemens Healthineers
Medtronic
Johnson & Johnson
Becton Dickinson
Merck & Co.
Danaher Corporation
- Increasing adoption of personalized medicine in hospitals
- Rising demand for early disease detection in diagnostics laboratories
- Biopharmaceutical companies focusing on biomarker research
- Government initiatives to support medical research and development
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035