Market Overview
The market for ANA testing is growing steadily, driven by the increasing prevalence of autoimmune diseases and the rising demand for advanced diagnostic tools. Based on a recent historical assessment, the ANA testing market is valued USD ~ billion, with strong demand emerging from healthcare providers seeking better diagnostic methods. This growth is supported by expanding diagnostic capabilities in both urban and rural areas, providing more opportunities for market expansion.
The Philippines remains a dominant market due to advancements in healthcare infrastructure, government investments in the healthcare sector, and the growing awareness of autoimmune disorders. Major cities such as Metro Manila lead the adoption of ANA testing services, with healthcare providers, research institutions, and diagnostic centers increasingly implementing these technologies to meet growing diagnostic demand. The increasing number of healthcare facilities and specialized diagnostic laboratories has further fueled the growth in the region.
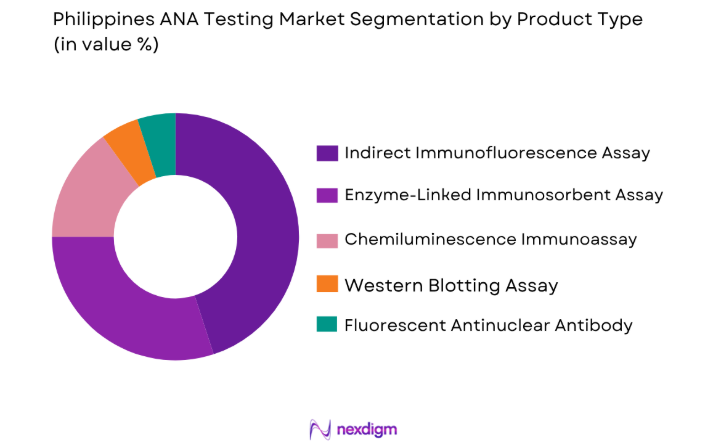
Market Segmentation
By Product Type
The ANA testing market is segmented by product type into various sub-segments, including Enzyme-Linked Immunosorbent Assay (ELISA), Indirect Immunofluorescence Assay (IFA), Chemiluminescence Immunoassay (CLIA), and others. The dominant sub-segment in this market is the Indirect Immunofluorescence Assay (IFA), driven by its reliability, high sensitivity, and widespread adoption in diagnostic laboratories. The high level of accuracy and the ability to detect a wide range of autoimmune diseases contribute to IFA’s market leadership, making it the preferred choice for both clinical and research applications.
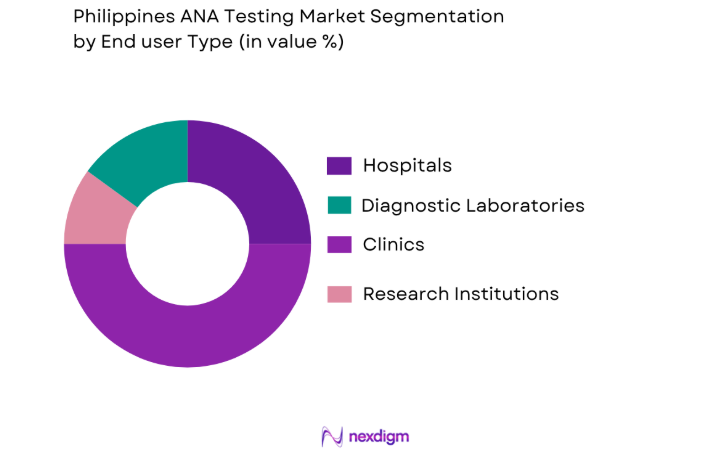
By End User Segment
The ANA testing market is further segmented by end-user into hospitals, diagnostic laboratories, research institutes, and clinics. Diagnostic laboratories lead the market due to their increasing role in offering specialized testing services and diagnostics for autoimmune diseases. The growing awareness of autoimmune disorders and the emphasis on early diagnosis have made diagnostic laboratories essential in the healthcare ecosystem, ensuring they remain a dominant end-user segment. Additionally, advancements in lab technologies and automation contribute to the sustained demand for ANA testing services.
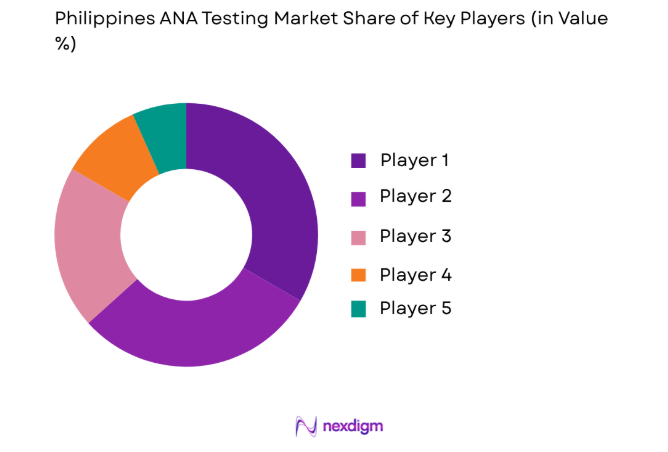
Competitive Landscape
The ANA testing market is highly competitive, with leading players focusing on innovation and partnerships to enhance their market presence. Major companies in the market are consolidating to expand their capabilities and increase product offerings. These players are using technological advancements and strategic mergers to strengthen their positions. Market competition is also driven by regional regulations and the increasing demand for accurate diagnostic tools.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Additional Parameter |
| Abbott Laboratories | 1888 | USA | ~ | ~ | ~ | ~ | ~ |
| Thermo Fisher Scientific | 1956 | USA | ~ | ~ | ~ | ~ | ~ |
| Roche Diagnostics | 1896 | Switzerland | ~ | ~ | ~ | ~ | ~ |
| Siemens Healthineers | 1847 | Germany | ~ | ~ | ~ | ~ | ~ |
| Beckman Coulter | 1935 | USA | ~ | ~ | ~ | ~ | ~ |
Philippines ANA Testing Market Analysis
Growth Drivers
Technological advancements in diagnostic testing
Technological innovation has become a key growth driver in the ANA testing market. New diagnostic platforms that offer increased sensitivity, higher throughput, and faster results have revolutionized the industry. Automated systems, which enhance workflow efficiency and reduce human error, are also contributing to market growth. The advent of artificial intelligence and machine learning in diagnostics is enabling more accurate interpretations of test results, which is enhancing the overall diagnostic process. This innovation is particularly crucial in the healthcare sector, where timely and accurate testing plays a vital role in patient outcomes. As healthcare providers increasingly adopt these new technologies, the ANA testing market is poised to experience significant growth.
Healthcare infrastructure expansion:
Another key driver of the ANA testing market is the ongoing expansion of healthcare infrastructure, particularly in emerging markets. Governments and private sectors are investing in healthcare facilities, medical research, and diagnostic laboratories to improve disease detection and management. This is especially prominent in regions like Southeast Asia, where healthcare systems are being rapidly modernized to cater to growing populations and rising healthcare demands. The proliferation of diagnostic centers, combined with the increasing availability of diagnostic tools, is driving the widespread adoption of ANA testing services. The enhanced accessibility and affordability of testing services are key to meeting the rising demand for accurate diagnostic tools for autoimmune diseases.
Market Challenges
High cost of diagnostic testing:
One of the significant challenges faced by the ANA testing market is the high cost associated with advanced diagnostic equipment and consumables. While the demand for accurate diagnostic testing is on the rise, the high capital investment required for setting up diagnostic laboratories and acquiring testing systems remains a barrier for smaller healthcare facilities. The pricing of ANA testing kits, reagents, and instruments also adds to the overall cost burden, limiting access in low-income regions. Furthermore, the need for continuous maintenance and technician training to operate these systems increases the overall operational costs. These factors present a significant challenge to the market’s expansion, particularly in developing countries where healthcare funding is constrained.
Regulatory hurdles and standardization issues:
The ANA testing market also faces challenges related to the regulatory environment. Different countries have varying regulations and certification requirements for diagnostic products, which can complicate market entry for manufacturers and disrupt the supply chain. The lack of standardized protocols for testing and reporting results also creates inconsistency in diagnosis, which affects the reliability of ANA testing. Moreover, approval processes for diagnostic equipment can be lengthy and costly, delaying the launch of new technologies and innovations in the market. Addressing these regulatory barriers and achieving standardization across global markets will be crucial for the market’s growth in the coming years.
Opportunities
Growing demand for personalized medicine:
The increasing shift toward personalized medicine presents significant opportunities for the ANA testing market. As the healthcare industry moves toward more tailored treatment options, the role of genetic and autoimmune disease testing becomes more pronounced. ANA testing plays a critical role in diagnosing autoimmune disorders and determining appropriate treatment plans. The rising awareness of autoimmune diseases, combined with the growing focus on personalized care, will continue to drive demand for ANA testing. Healthcare providers are increasingly turning to precision diagnostics to offer more effective treatments, and ANA testing is a key component of this shift. The market can capitalize on this trend by offering more advanced testing solutions that enable healthcare professionals to make more informed decisions about patient care.
Partnerships and collaborations for innovation:
Another significant opportunity lies in forming partnerships and collaborations with healthcare providers, research institutes, and biotechnology companies. These partnerships can foster the development of new and improved ANA testing platforms that incorporate the latest technologies, such as artificial intelligence and machine learning. Collaboration between diagnostic companies and healthcare providers can also facilitate the widespread adoption of advanced testing methods and drive innovation in diagnostic processes. With the growing demand for more accurate and efficient diagnostic solutions, ANA testing companies have the opportunity to leverage these collaborations to create cutting-edge products that meet market needs and expand their market reach.
Future Outlook
The ANA testing market is expected to continue its growth trajectory over the next five years, driven by ongoing technological advancements, increased healthcare spending, and expanding diagnostic infrastructure. The increasing prevalence of autoimmune diseases and the rising demand for more efficient and accurate diagnostic tools will contribute to this growth. As technology evolves, newer and more efficient diagnostic methods will likely become available, improving testing capabilities and driving market demand. Regulatory support, particularly in emerging markets, will also help to boost the adoption of ANA testing services, while the continued integration of personalized medicine into healthcare systems will further solidify the role of ANA testing in diagnosing autoimmune diseases.
Major Players
- Abbott Laboratories
- ThermoFisher Scientific
- Roche Diagnostics
- Siemens Healthineers
- Beckman Coulter
- Bio-Rad Laboratories
- Laboratory Corporation of America (LabCorp)
- Quidel Corporation
- DiaSorin
- Hitachi Chemical Diagnostics
- Sysmex Corporation
- Ortho Clinical Diagnostics
- Mayo Clinic Laboratories
- Becton Dickinson and Company
- Randox Laboratories
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Healthcare providers and clinics
- Diagnostic laboratories
- Pharmaceutical companies
- Biotechnology companies
- Research institutions
- Medical device distributors
Research Methodology
Step 1: Identification of Key Variables
Identification of key factors affecting the market is the first step, focusing on variables such as healthcare infrastructure, technology adoption, and disease prevalence.
Step 2: Market Analysis and Construction
This step involves analyzing historical data, trends, and patterns to construct a detailed market model that forecasts future performance and identifies growth opportunities.
Step 3: Hypothesis Validation and Expert Consultation
We validate our market hypotheses through expert consultations with key stakeholders, including industry leaders, healthcare professionals, and regulatory bodies.
Step 4: Research Synthesis and Final Output
The final research output synthesizes all findings, providing comprehensive market insights, actionable recommendations, and detailed market forecasts based on thorough analysis.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Growth Drivers
Increasing prevalence of autoimmune diseases
Rising healthcare awareness and diagnostics infrastructure
Government initiatives in disease management - Market Challenges
High cost of advanced testing equipment
Limited accessibility in rural areas
Shortage of trained professionals in specialized diagnostics - Market Opportunities
Expansion in healthcare infrastructure in rural regions
Advancements in test accuracy and speed
Increasing partnerships between healthcare providers and diagnostic companies - Trends
Rise in adoption of AI in diagnostic platforms
Shift towards personalized medicine and targeted therapies
Increasing demand for non-invasive diagnostic tests - Government Regulations
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Enzyme-Linked Immunosorbent Assay (ELISA)
Fluorescent Antinuclear Antibody (FANA)
Indirect Immunofluorescence Assay (IFA)
Chemiluminescence Immunoassay (CLIA)
Western Blotting Assay - By Platform Type (In Value%)
Laboratory-based Systems
Point-of-Care Systems
Mobile Testing Platforms
Automated Testing Platforms
Manual Testing Kits - By Fitment Type (In Value%)
Standalone Systems
Integrated Systems
Portable Systems
Bench-top Systems
Microfluidic-based Systems - By EndUser Segment (In Value%)
Hospitals
Diagnostic Laboratories
Research Institutes
Clinics
Pharmaceutical Companies
- Market Share Analysis
- CrossComparison Parameters (System Type, Platform Type, Procurement Channel, EndUser Segment, Fitment Type)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Porter’s Five Forces
- Key Players
Abbott Laboratories
Thermo Fisher Scientific
Roche Diagnostics
Siemens Healthineers
Beckman Coulter
Bio-Rad Laboratories
Laboratory Corporation of America (LabCorp)
Quidel Corporation
DiaSorin
Hitachi Chemical Diagnostics
Sysmex Corporation
Ortho Clinical Diagnostics
Mayo Clinic Laboratories
Becton Dickinson and Company
Randox Laboratories
- Growth in demand from diagnostic laboratories
- Hospitals adopting advanced diagnostic systems
- Research institutes focusing on autoimmune disorder studies
- Clinics enhancing diagnostic offerings for autoimmune diseases
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035