Market Overview
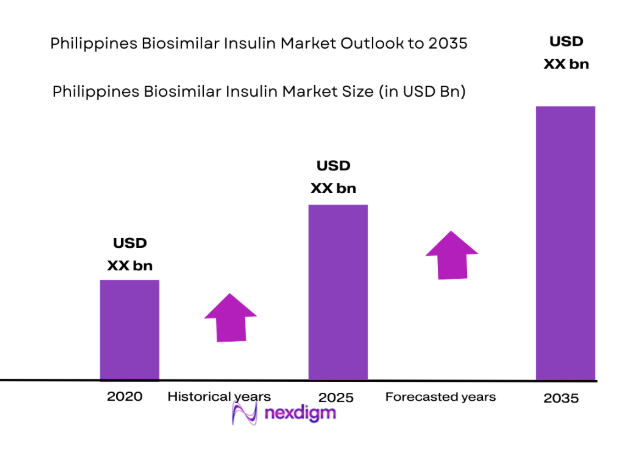
The Philippines biosimilar insulin market is poised for growth, with an increasing demand for affordable insulin alternatives. Based on a recent historical assessment, the market size is expected to reach USD ~ billion, driven by rising diabetes prevalence, government initiatives, and the demand for cost-effective healthcare solutions. The adoption of biosimilars is further supported by regulatory frameworks that encourage competition and affordability in the healthcare sector, enabling broader access to insulin therapies.
The market is dominated by key regions like Metro Manila and Cebu, where healthcare infrastructure and the concentration of healthcare providers are more advanced. These areas account for the majority of insulin demand due to a high urban population, increased healthcare access, and government-driven health initiatives that promote the use of biosimilar drugs. The growing adoption of biosimilars is encouraged by the efforts to ensure better management of chronic diseases like diabetes.
Market Segmentation
By Product Type
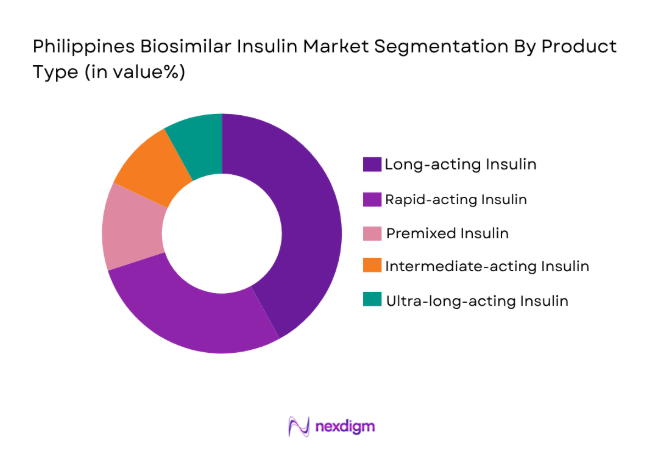
The Philippines biosimilar insulin market is segmented by product type into long-acting insulin, rapid-acting insulin, premixed insulin, intermediate-acting insulin, and ultra-long-acting insulin. Recently, the long-acting insulin sub-segment has dominated the market share due to its high demand for diabetes management, as it provides a consistent blood sugar control over a 24-hour period. The availability of biosimilar versions has further encouraged adoption among patients looking for affordable alternatives to branded insulin products. Additionally, long-acting insulin is preferred by patients with Type 1 diabetes and insulin-dependent Type 2 diabetes due to its convenience and efficacy.
By Platform Type
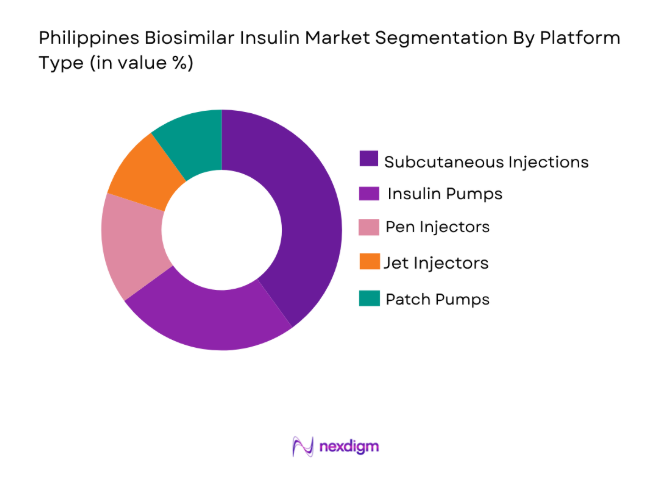
The Philippines biosimilar insulin market is segmented by platform type into subcutaneous injections, insulin pumps, pen injectors, jet injectors, and patch pumps. Recently, the subcutaneous injections sub-segment has dominated the market share due to its widespread adoption for diabetes management. These injections are convenient, cost-effective, and easy to administer at home, making them the preferred choice for many patients. The growing availability of affordable biosimilars in injectable form, along with increased patient education about their benefits, has driven the adoption of subcutaneous insulin delivery. As a result, this platform type continues to capture a significant portion of the market, with both patients and healthcare providers favoring it for routine diabetes treatment.
Competitive Landscape
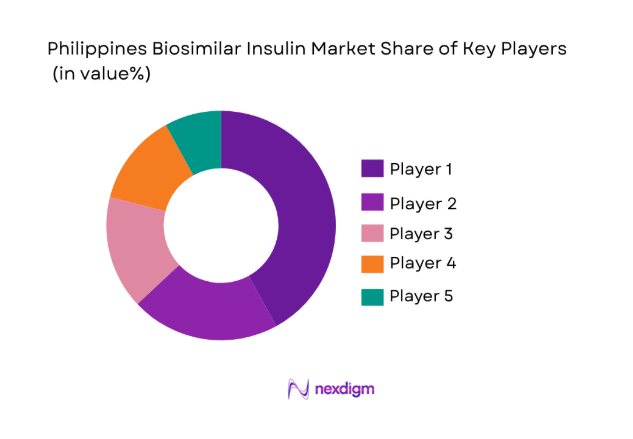
The competitive landscape of the Philippines biosimilar insulin market is highly influenced by both local and international players. Major players dominate the market, with significant consolidation seen through partnerships and acquisitions. These companies leverage advanced biotechnological solutions to improve the efficacy and availability of biosimilar insulin, which is crucial for the growing diabetic population. Market leaders are focusing on expanding distribution networks and improving healthcare accessibility across the nation.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Market-specific Parameter |
| Sanofi | 2004 | Paris, France | ~ | ~ | ~ | ~ | ~ |
| Novo Nordisk | 1923 | Denmark | ~ | ~ | ~ | ~ | ~ |
| Mylan N.V. | 1961 | Netherlands | ~ | ~ | ~ | ~ | ~ |
| Biocon | 1978 | Bangalore, India | ~ | ~ | ~ | ~ | ~ |
| Eli Lilly and Company | 1876 | Indianapolis, USA | ~ | ~ | ~ | ~ | ~ |
Philippines Biosimilar Insulin Market Analysis
Growth Drivers
Increasing Diabetes Prevalence
The rising number of diabetes cases in the Philippines has significantly boosted the demand for affordable insulin therapies. The country has witnessed a steady rise in the diabetic population, driven by a combination of factors, including urbanization, dietary changes, and increasingly sedentary lifestyles. As the prevalence of diabetes continues to grow, so does the urgent need for cost-effective insulin alternatives. Traditional insulin therapies, which often come with high costs, have become increasingly inaccessible to a large portion of the population, creating an opening for more affordable options such as biosimilars. Additionally, the increasing burden of diabetes on the healthcare system has spurred a greater focus on managing chronic diseases more effectively. As a result, there has been a significant push from both public and private sectors to make diabetes management more affordable and accessible, which is driving the growth of the biosimilar insulin market. Government efforts to expand healthcare coverage, especially for chronic conditions like diabetes, have ensured that patients who may not have access to branded insulin products can now rely on biosimilars. Furthermore, many insurance providers have started to include biosimilar insulin in their coverage plans, further making these products more affordable and widely accessible to patients across the country. This shift in healthcare policy, coupled with a growing understanding of the therapeutic efficacy of biosimilars, has made them a key solution in addressing the increasing diabetes burden. In turn, the demand for biosimilar insulin has seen an upward trajectory as more patients opt for these cost-effective alternatives to manage their condition.
Government Support for Affordable Healthcare
Government initiatives aimed at enhancing access to affordable healthcare are accelerating the growth of the biosimilar insulin market in the Philippines. The Philippine government has recognized the growing prevalence of diabetes as a national healthcare challenge and has responded by making significant investments in improving healthcare access, particularly for chronic diseases. This has included increasing the availability of essential medicines such as insulin, which has historically been costly and out of reach for many patients. The government’s policies have been designed to not only improve the quality of healthcare services but also make those services more affordable, particularly for marginalized populations. The introduction of favorable regulations, including a streamlined approval process for biosimilars and price controls on essential medicines, has made biosimilars more attractive to both consumers and healthcare providers. These policies have allowed biosimilar insulin to become a viable alternative to more expensive branded insulin products, ensuring that more patients can access the treatment they need. Furthermore, public health campaigns led by the government have helped raise awareness about diabetes and the benefits of biosimilars, encouraging more patients and healthcare providers to consider them as an effective and affordable option. The government’s commitment to improving healthcare access, coupled with the regulatory support for biosimilars, has created a robust environment for the growth of the biosimilar insulin market. This proactive stance in tackling chronic diseases such as diabetes has played a pivotal role in driving the demand for more affordable treatment options, ensuring that patients can manage their condition without financial hardship.
Market Challenges
Regulatory Barriers to Market Entry
Despite the Philippine government’s efforts to streamline healthcare policies, the regulatory framework for approving biosimilars remains complex and can delay market entry for new players. Biosimilars, though scientifically equivalent to their reference products, must undergo rigorous clinical trials and extensive documentation processes to gain approval from regulatory bodies like the Philippine Food and Drug Administration (FDA). These regulatory processes can be time-consuming, costly, and complicated, posing a significant barrier for smaller manufacturers who may not have the resources to navigate this demanding landscape. The complexity of the regulatory framework increases the overall time to market, which can delay the availability of affordable biosimilars, including insulin, to patients who desperately need them. Furthermore, the regulatory bodies’ cautious approach to approving new biosimilars—stemming from concerns about their safety and efficacy—can hinder faster adoption and availability of new products. Although biosimilars are designed to be highly similar to branded versions, the perception that they are less proven can result in reluctance from both healthcare professionals and patients. This hesitation further slows market penetration and restricts the full potential of biosimilar insulin to address the growing demand in the Philippines. To overcome these regulatory barriers, biosimilar manufacturers must invest significant resources in meeting the stringent requirements set by the Philippine FDA, adding to the overall cost of production and potentially limiting the number of players in the market.
Public Perception and Physician Hesitancy
Another major challenge facing the market is the hesitancy of some healthcare professionals to prescribe biosimilars due to concerns about their safety and efficacy. While biosimilars are scientifically equivalent to their reference products, there remains a perception among some doctors that they may not deliver the same therapeutic outcomes, particularly for complex conditions such as advanced diabetes. Physicians may worry about potential variations in therapeutic efficacy, which can influence their decisions to prescribe biosimilars to their patients. This hesitancy is compounded by a lack of widespread awareness about the safety and effectiveness of biosimilars, both among healthcare professionals and patients. Some doctors may feel more comfortable prescribing branded insulin, especially if they have established relationships with pharmaceutical companies and have seen the long-term success of branded products. This resistance can also stem from a limited understanding of the regulatory rigor that goes into the approval of biosimilars, which can perpetuate skepticism regarding their use. For patients, the perception that biosimilars are “inferior” to branded options can be a major obstacle to adoption, even when these alternatives are more affordable. Overcoming this challenge requires ongoing education and awareness campaigns targeted at both healthcare professionals and patients, emphasizing the proven safety, efficacy, and cost-effectiveness of biosimilars. As more data becomes available and as healthcare providers gain experience with biosimilars, the acceptance of these alternatives is likely to increase, ultimately leading to higher adoption rates across the Philippines.
Opportunities
Expansion of Biosimilar Insulin in Rural Areas
A significant opportunity exists in expanding the availability of biosimilar insulin in rural areas where access to insulin therapies has traditionally been limited. In many rural regions of the Philippines, access to healthcare services, including insulin, has been historically inadequate due to factors such as a lack of infrastructure, limited healthcare professionals, and higher costs associated with obtaining essential medications. However, the Philippine government has made substantial efforts in improving healthcare infrastructure in these areas, providing an opportunity for biosimilar insulin manufacturers to expand their reach. By leveraging government initiatives to build healthcare facilities and increase the availability of essential medicines, biosimilar insulin can be made more accessible to rural populations who have previously struggled to afford or obtain branded insulin products. This expansion presents an opportunity for manufacturers to target untapped markets, especially as rural populations are increasingly diagnosed with diabetes. As healthcare services improve in these regions, the demand for affordable, accessible diabetes management solutions will continue to rise, driving further growth in the biosimilar insulin market. By establishing distribution networks, partnering with local healthcare providers, and offering education on the benefits of biosimilars, manufacturers can help address the growing demand in rural areas, ultimately improving the health outcomes of diabetic patients who may not otherwise have access to the treatment they need.
Technological Advancements in Insulin Delivery Systems
Advances in insulin delivery systems, such as the development of insulin pens, pumps, and smart devices, present a valuable opportunity for the biosimilar insulin market. These technologies enhance the convenience and effectiveness of insulin administration, making it easier for patients to manage their diabetes. The increasing adoption of digital health solutions, including smartphone apps and smart insulin pens, has the potential to revolutionize the way patients monitor their condition and administer insulin. These devices provide real-time data and allow for more personalized treatment plans, improving patient outcomes and encouraging adherence to treatment regimens. As these insulin delivery systems become more widespread and affordable, they create a larger market for biosimilar insulin, especially among younger and tech-savvy patients who are more likely to embrace these advancements. The integration of digital solutions with biosimilar insulin can also enhance patient monitoring, facilitating better decision-making and more effective diabetes management. This shift towards smart insulin delivery systems, coupled with the affordability and accessibility of biosimilars, creates a unique opportunity for manufacturers to position their products as part of a comprehensive, modern approach to diabetes care. By aligning with technological advancements in insulin delivery, biosimilar insulin can gain increased acceptance and usage, further driving the demand for these products in the market.
Future Outlook
The future of the Philippines biosimilar insulin market looks promising, with expected steady growth over the next five years. Technological advancements in insulin delivery systems, along with government policies supporting biosimilar adoption, will fuel market expansion. The demand for cost-effective insulin alternatives will continue to rise due to the increasing prevalence of diabetes and the push for affordable healthcare. Additionally, the market will likely see innovations in drug formulations and delivery mechanisms, which will contribute to the continued adoption of biosimilar insulin in the country.
Major Players
- Sanofi
- Novo Nordisk
- Mylan N.V.
- Biocon
- Eli Lilly and Company
- Merck & Co.
- Amgen
- Pfizer
- Boehringer Ingelheim
- Sandoz
- Wockhardt
- Lupin Pharmaceuticals
- Intas Pharmaceuticals
- Teva Pharmaceutical Industries
- Glenmark Pharmaceuticals
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Pharmaceutical manufacturers
- Healthcare providers
- Diabetes care organizations
- Hospital networks
- Pharmaceutical distributors
Research Methodology
Step 1: Identification of Key Variables
We identify critical market variables and metrics to form the foundation of our analysis, ensuring the relevant factors are addressed in all stages.
Step 2: Market Analysis and Construction
We conduct in-depth data analysis, leveraging secondary research to construct a robust market model based on historical and current trends.
Step 3: Hypothesis Validation and Expert Consultation
We validate assumptions and projections through consultations with industry experts and stakeholders, ensuring the model’s accuracy and reliability.
Step 4: Research Synthesis and Final Output
We synthesize the research findings into actionable insights, presenting them in a comprehensive, clear, and accessible format for decision-making.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Rising Prevalence of Diabetes
Government Initiatives for Affordable Healthcare
Increase in Biosimilar Acceptance - Market Challenges
Regulatory Hurdles for Biosimilars
Limited Local Production Capabilities
High Cost of Advanced Biosimilar Insulin - Market Opportunities
Expanding Healthcare Access in Rural Areas
Partnerships with Private Sector for Market Growth
Technological Advancements in Insulin Delivery Systems - Trends
Increase in Biosimilar Insulin Adoption
Advances in Injectable Insulin Technologies - Government Regulations
Regulations on Biosimilar Approval
Healthcare Reimbursement Policies
Importation and Taxation Policies - SWOT Analysis
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Long-acting Insulin
Rapid-acting Insulin
Premixed Insulin
Intermediate-acting Insulin
Ultra-long-acting Insulin - By Platform Type (In Value%)
Subcutaneous Injections
Insulin Pumps
Pen Injectors
Jet Injectors
Patch Pumps - By Fitment Type (In Value%)
Self-administered Solutions
Healthcare Provider-administered Solutions
In-hospital Solutions
At-home Solutions - By End User Segment (In Value%)
Hospitals & Clinics
Homecare Patients
Healthcare Providers
- Market Share Analysis
- Cross Comparison Parameters (System Type, Platform Type, Fitment Type, End User Segment, Cost Efficiency, Delivery Mechanism, Technology Integration)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Sanofi
Novo Nordisk
Bayer AG
Eli Lilly and Company
Mylan N.V.
Boehringer Ingelheim
Sandoz (Novartis)
Baxter International
Wockhardt Limited
Intas Pharmaceuticals
Lupin Pharmaceuticals
Zydus Cadila
Hikma Pharmaceuticals
AbbVie
Biocon
- Homecare Patients’ Growing Need for Convenience
- Hospitals’ Rising Demand for Cost-effective Treatments
- Government-Backed Healthcare Programs
- Diabetes-focused Clinics’ Expanding Role
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035