Market Overview
The Philippines biosimilar monoclonal antibodies market has experienced significant growth driven by increasing demand for affordable biologics, particularly in oncology and autoimmune treatments. The market size is primarily fueled by the rising prevalence of chronic diseases and the increasing adoption of biosimilars as a cost-effective alternative to originator monoclonal antibodies. Based on a recent historical assessment, the market size is valued at USD ~ billion, with growth expected to continue as regulatory frameworks evolve to support biosimilar approvals.
The market is dominated by urban centers such as Manila, which serve as key hubs for the healthcare industry. These regions are characterized by robust healthcare infrastructure, increased patient access to medical treatments, and growing investments in healthcare technologies. The Philippines’ government and healthcare programs are actively promoting the use of biosimilars, contributing to the country’s role as a leader in the biosimilar monoclonal antibodies market in Southeast Asia.
Market Segmentation
By Product Type:
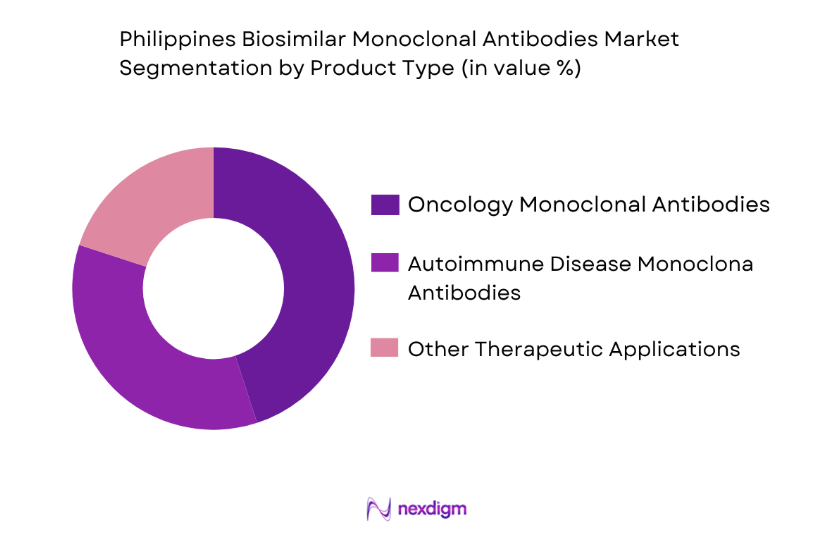
The biosimilar monoclonal antibodies market is segmented by product type into oncology monoclonal antibodies, autoimmune disease monoclonal antibodies, and other therapeutic applications. Recently, oncology monoclonal antibodies have a dominant market share due to factors such as the growing incidence of cancer and increasing healthcare infrastructure investments. The rise in cancer awareness and the demand for targeted therapies has significantly boosted the adoption of these biosimilars, making oncology the leading sub-segment.
By Platform Type:
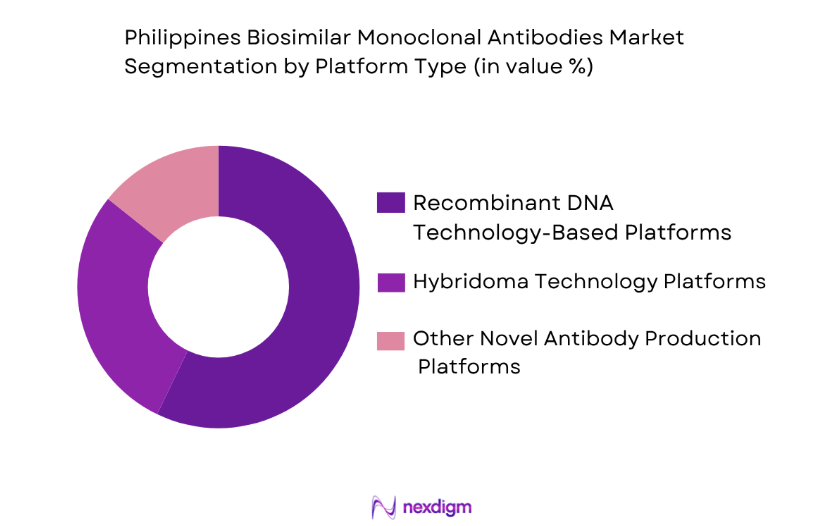
The market is also segmented by platform type into recombinant DNA technology-based platforms, hybridoma technology platforms, and other novel antibody production platforms. Recombinant DNA technology-based platforms dominate the market share due to their advanced capabilities in producing high-quality monoclonal antibodies. This platform offers scalability, cost-efficiency, and precision, which are critical factors for the widespread adoption of biosimilars, especially in oncology and autoimmune diseases.
Competitive Landscape
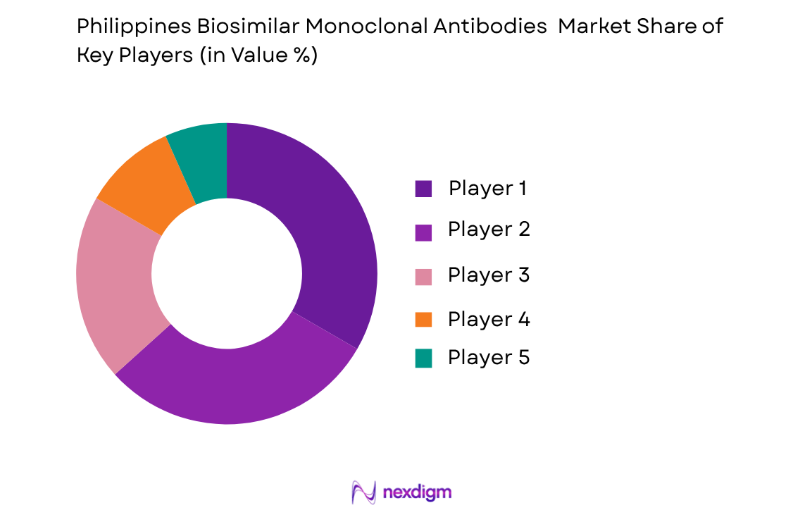
The competitive landscape of the Philippines biosimilar monoclonal antibodies market is characterized by a mix of global pharmaceutical companies and regional players. Major players are leveraging partnerships and regulatory advancements to gain a strong foothold in the market. There has been notable consolidation in the sector, with leading firms acquiring smaller biotech companies to enhance their biosimilar portfolios.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD Billion) | Regulatory Approval Rate |
| Pfizer | 1849 | USA | ~ | ~ | ~ | ~ | ~ |
| Amgen | 1980 | USA | ~ | ~ | ~ | ~ | ~ |
| Celltrion | 2002 | South Korea | ~ | ~ | ~ | ~ | ~ |
| Sandoz | 2003 | Switzerland | ~ | ~ | ~ | ~ | ~ |
| Biocon | 1978 | India | ~ | ~ | ~ | ~ | ~ |
Philippines Biosimilar Monoclonal Antibodies Market Analysis
Growth Drivers
Government Support for Biosimilars:
The Philippine government has introduced several initiatives to encourage the adoption of biosimilars, including regulatory incentives and policies that reduce the cost of biologics. The Department of Health has been actively supporting biosimilars by implementing frameworks to streamline the approval process and ensure the safety of these drugs. This government-backed support is crucial for increasing access to biosimilars, particularly in the context of rising healthcare costs. Furthermore, public health initiatives aim to reduce the economic burden of expensive monoclonal antibodies by encouraging their substitution with biosimilars. In turn, this creates opportunities for biosimilar producers to increase their market penetration, thereby accelerating market growth.
Increase in Chronic Diseases and Cancer Incidence:
Another key growth driver for the biosimilar monoclonal antibodies market is the increasing prevalence of chronic diseases and cancer. As the population ages and lifestyle-related diseases become more common, the demand for affordable treatments rises. Cancer, in particular, is a leading cause of death in the Philippines, and monoclonal antibodies have become a staple in cancer treatment regimens. Biosimilars offer a cost-effective alternative to branded monoclonal antibodies, making them accessible to a wider range of patients. This is particularly significant in the Philippines, where healthcare affordability remains a primary concern. As the government continues to prioritize cancer care, the adoption of biosimilars is set to increase significantly.
Market Challenges
Regulatory Barriers to Market Entry:
Despite supportive government policies, regulatory challenges remain a significant hurdle for new biosimilar entrants. The approval process for biosimilars in the Philippines requires a rigorous demonstration of biosimilarity to the reference biologic, which can be time-consuming and costly. Additionally, the regulatory framework is still evolving, with new requirements that could further complicate the market entry for emerging biosimilar manufacturers. These regulatory barriers create delays in getting biosimilars to market, which in turn limits the potential growth of the sector. Companies looking to enter the Philippine market must navigate a complex regulatory environment, which can slow the adoption and availability of new biosimilar products.
High Development Costs and Infrastructure Limitations:
The production of biosimilars is highly capital-intensive, and the Philippines faces limitations in terms of manufacturing capabilities and infrastructure. The need for state-of-the-art facilities, specialized equipment, and skilled labor adds to the cost of production, making it difficult for smaller companies to compete with larger multinational corporations. While the demand for biosimilars is growing, the high cost of production and lack of advanced infrastructure are barriers to entry for many local players. In addition, the Philippines’ reliance on imported biosimilars adds to the overall cost, making it challenging to offer products at competitive prices. This market challenge poses a significant constraint on the growth potential of the biosimilar monoclonal antibodies market.
Opportunities
Expansion of Biosimilar Portfolio in Oncology:
One of the biggest opportunities in the biosimilar monoclonal antibodies market lies in the expansion of biosimilar portfolios in oncology. With the increasing incidence of cancer, particularly in Southeast Asia, biosimilars offer an affordable alternative to expensive oncology biologics. The government and healthcare providers are focused on reducing the financial burden of cancer treatments, and biosimilars are emerging as a solution to this problem. Companies can seize the opportunity to expand their offerings by developing a wide range of oncology biosimilars, particularly for high-demand treatments such as HER2-targeted therapies. By focusing on oncology biosimilars, manufacturers can tap into a growing market and drive significant revenue growth.
Collaborations with Global Biopharmaceutical Companies:
The biosimilar monoclonal antibodies market in the Philippines also presents an opportunity for local players to collaborate with global biopharmaceutical companies. These partnerships can help local manufacturers gain access to advanced technologies, expertise, and distribution networks, accelerating the development and commercialization of biosimilars. Through strategic alliances, local players can improve their manufacturing capabilities, scale production, and improve their ability to meet the growing demand for biosimilars. As the global demand for biosimilars increases, these collaborations will become an essential avenue for expanding market reach and enhancing product portfolios in the Philippines.
Future Outlook
The future of the Philippines biosimilar monoclonal antibodies market looks promising, with continued growth expected over the next five years. This growth will be driven by increasing government support, a rise in chronic disease incidences, and advancements in biosimilar manufacturing technologies. Additionally, the Philippines’ regulatory framework is expected to further evolve, making it easier for new biosimilars to enter the market. As healthcare systems continue to prioritize affordability and accessibility, biosimilars will play an increasingly significant role in reducing the financial burden on patients and healthcare providers. Technological developments in biologic drug manufacturing will also help lower production costs, facilitating greater market penetration.
Major Players
- Pfizer
- Amgen
- Celltrion
- Sandoz
- Biocon
- Mylan
- SamsungBioepis
- Eli Lilly
- Roche
- Novartis
- Teva Pharmaceutical
- AbbVie
- Merck & Co.
- GSK
- Boehringer Ingelheim
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Pharmaceutical manufacturers
- Oncology treatment providers
- Biotechnology companies
- Hospitals and healthcare providers
- Cancer research institutions
- Public health organizations
Research Methodology
Step 1: Identification of Key Variables
This step involves identifying and defining the key variables affecting the biosimilar monoclonal antibodies market, including technological factors, regulatory policies, and consumer demand drivers.
Step 2: Market Analysis and Construction
The market analysis combines qualitative and quantitative data from primary and secondary sources to build a comprehensive model that represents the current market scenario.
Step 3: Hypothesis Validation and Expert Consultation
In this step, the developed hypotheses are validated through consultations with industry experts and stakeholders to ensure the accuracy and reliability of the market model.
Step 4: Research Synthesis and Final Output
The final output synthesizes all gathered data, providing insights and forecasts on market trends, opportunities, challenges, and competitive landscape, which form the basis of the market report.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Growth Drivers
Rising Prevalence of Chronic Diseases
Increasing Adoption of Cost-effective Biosimilars
Expanding Healthcare Infrastructure in the Philippines - Market Challenges
Regulatory Hurdles in Biosimilar Approval
High Production Costs of Biosimilar Monoclonal Antibodies
Limited Patient Awareness of Biosimilars - Market Opportunities
Expansion in Cancer Treatment Biosimilars
Partnerships with Global Pharmaceutical Companies
Government Initiatives to Support Biosimilar Adoption - Trends
Growing Preference for Biologic Drugs in Cancer Treatments
Technological Advancements in Monoclonal Antibody Production
Increased Focus on Patient Access and Affordability - Government Regulations
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Therapeutic Monoclonal Antibodies
Preventive Monoclonal Antibodies
Combination Monoclonal Antibodies
Biosimilar Monoclonal Antibodies for Oncology
Biosimilar Monoclonal Antibodies for Autoimmune Diseases - By Platform Type (In Value%)
Cell Culture-Based Systems
Recombinant DNA Technology-Based Platforms
Hybridoma Technology Platforms
Phage Display Platforms
Antibody Humanization Platforms - By Fitment Type (In Value%)
Injectable Solutions
Oral Solutions
Subcutaneous Injections
Intravenous Infusions
Topical Formulations - By EndUser Segment (In Value%)
Hospitals
Clinics
Pharmaceutical Companies
Government Healthcare Programs
- Market Share Analysis
- CrossComparison Parameters (Therapeutic Monoclonal Antibodies, Biosimilar Adoption Rate, Healthcare Infrastructure, Government Support, Research & Development Investments)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Porter’s Five Forces
- Key Players
Pfizer
Amgen
Bristol-Myers Squibb
Roche
Sandoz
Celltrion
Mylan
Samsung Bioepis
Eisai
Biocon
Novartis
Biogen
Sun Pharmaceutical
Hikma Pharmaceuticals
Alvogen
- Hospitals Increasing Investment in Biologics
- Rising Number of Biosimilar Clinics and Treatment Centers
- Pharmaceutical Companies’ Shift Towards Biosimilars
- Government Healthcare Programs Promoting Affordable Treatments
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035‘