Market Overview
The Philippines cell therapy market is projected to experience significant growth due to advancements in biotechnology and an increasing demand for personalized medicine. With ongoing investments in research and development, the market is expected to expand as new therapies are introduced for chronic diseases and genetic disorders. Additionally, the country’s growing healthcare infrastructure and government initiatives supporting biotech innovations contribute to the market’s upward trajectory. The market size for 2024 is expected to reach USD ~ billion, driven by both domestic and international demand for advanced medical treatments.
The Philippines is rapidly emerging as a key player in the Southeast Asian healthcare sector, with major cities like Manila and Cebu leading the growth in cell therapy adoption. Factors such as a strong healthcare system, supportive regulatory framework, and increasing healthcare expenditure have created a conducive environment for cell therapy market expansion. Furthermore, the demand for cell-based therapies is expected to rise as the government emphasizes improving healthcare access and as multinational pharmaceutical companies establish a stronger presence in the region. This growth is also supported by partnerships between local biotech firms and international research institutions.
Market Segmentation
By Product Type:
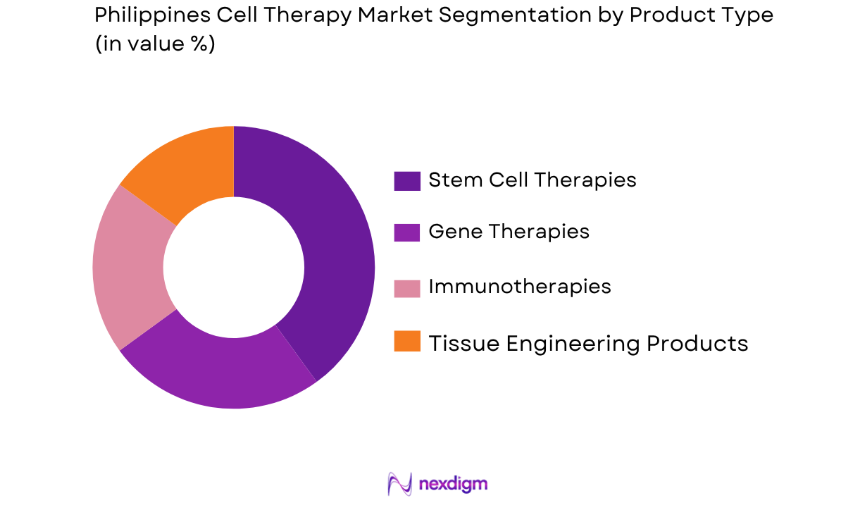
The Philippines cell therapy market is segmented by product type into stem cell therapies, gene therapies, immunotherapies, and tissue engineering products. Recently, stem cell therapies have dominated the market due to their application in a variety of conditions, from degenerative diseases to injuries requiring regenerative treatments. The increasing prevalence of conditions such as arthritis, heart disease, and neurological disorders has propelled the demand for stem cell therapies. Furthermore, advancements in stem cell research and growing consumer awareness are expected to keep this segment at the forefront in the coming years.
By Platform Type:
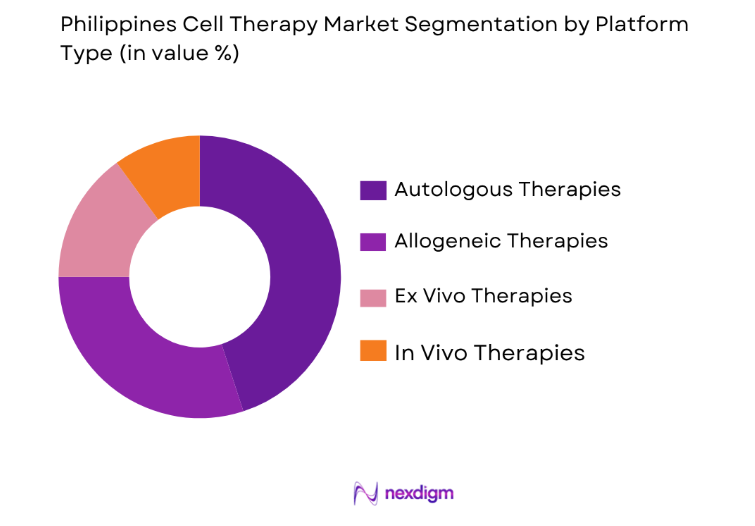
The Philippines cell therapy market is segmented by platform type into autologous therapies, allogeneic therapies, ex vivo therapies, and in vivo therapies. Among these, autologous therapies have the highest market share due to their ability to reduce the risk of immune rejection by utilizing a patient’s own cells for treatment. This platform’s dominance is further supported by advancements in cell isolation and expansion technologies, making autologous therapies increasingly efficient and accessible. Moreover, the preference for personalized medicine is expected to keep autologous therapies as the leading platform type in the market.
Competitive Landscape
The Philippines cell therapy market is highly competitive, with key players focusing on strategic collaborations and acquisitions to strengthen their market position. Major pharmaceutical and biotechnology companies are increasingly investing in local partnerships and establishing manufacturing facilities to support the growing demand for advanced therapies. Consolidation in the market is expected to intensify, as companies look to leverage their technological capabilities and enhance their portfolio of cell therapy treatments.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Additional Market-Specific Parameter |
| Novartis | 1996 | Switzerland | ~ | ~ | ~ | ~ | ~ |
| Gilead Sciences | 1987 | USA | ~ | ~ | ~ | ~ | ~ |
| Kite Pharma | 2009 | USA | ~ | ~ | ~ | ~ | ~ |
| Regeneron Pharmaceuticals | 1988 | USA | ~ | ~ | ~ | ~ | ~ |
| Mesoblast Limited | 2004 | Australia | ~ | ~ | ~ | ~ | ~ |
Philippines Cell Therapy Market Analysis
Growth Drivers
Government Support for Biotechnology Initiatives:
The Philippine government has been actively investing in biotechnology through programs aimed at strengthening the country’s healthcare infrastructure. This includes increased funding for biotech research, regulatory support for new treatments, and partnerships with global pharmaceutical companies. This government support is a critical driver for the adoption and expansion of cell therapy technologies, enabling more clinical trials and the introduction of new therapies in the market. With increasing government incentives, the market for cell therapy is expected to grow significantly, particularly in the areas of stem cell research and regenerative medicine. The Philippine Department of Health has also emphasized developing regulatory frameworks that allow for faster approval of new therapies, which will further boost the adoption of cell therapies.
Technological Advancements in Cell Therapy Platforms:
As cell therapy technologies continue to evolve, the market is seeing significant improvements in the efficiency and effectiveness of various therapies. New breakthroughs in gene editing, stem cell differentiation, and the use of immunotherapy have led to the development of more effective and targeted treatments. Technological advancements in cell processing and genetic engineering, such as CRISPR, are expected to make cell therapies more accessible and cost-effective. These innovations are creating new avenues for treating chronic and genetic diseases, further driving the market’s growth.
Market Challenges
Regulatory Challenges in Cell Therapy Approvals:
One of the significant challenges faced by the cell therapy market in the Philippines is the complex regulatory environment. The approval process for new therapies can be lengthy and challenging, with stringent requirements for clinical trials and safety data. This slow pace of approval can delay the introduction of promising cell therapies to the market and hinder the ability of companies to meet the growing demand for innovative treatments. The Philippines’ regulatory framework, although improving, still presents hurdles for rapid market access, particularly for new and experimental therapies. This challenge may impede the market’s potential growth in the short term.
High Cost of Cell Therapy Treatments:
Despite technological advancements, the high cost of cell therapy treatments remains a significant barrier to widespread adoption in the Philippines. The production and administration of cell-based therapies are expensive, often limiting access to wealthier segments of the population. The lack of sufficient insurance coverage for these therapies adds to the financial burden, making them less accessible for the general public. The high treatment costs also prevent healthcare systems from integrating these therapies on a large scale, slowing down the overall market expansion.
Opportunities
Expanding Personalized Medicine:
The increasing trend toward personalized medicine presents a significant opportunity for the Philippines cell therapy market. As patients increasingly demand treatments tailored to their individual genetic makeup, the demand for autologous therapies and gene therapies is expected to rise. Personalized medicine offers the potential to improve patient outcomes by using cell therapies that are specifically designed to meet the unique needs of each individual. The Philippine healthcare system is beginning to prioritize personalized care, which will further support the market for cell therapies and drive future demand for personalized treatments.
Emerging Biotechnology Startups and Partnerships:
Another promising opportunity for the Philippines cell therapy market is the emergence of local biotechnology startups focused on developing innovative cell-based treatments. These startups are driving innovation and offering new solutions that address unmet medical needs. Additionally, the rise of strategic partnerships between local and international biotech firms is creating opportunities for knowledge transfer, resource sharing, and the acceleration of clinical trials. These collaborations are likely to enhance the availability of cutting-edge cell therapies in the market and offer substantial growth prospects for the sector in the coming years.
Future Outlook
The future of the Philippines cell therapy market looks promising, with significant growth expected in the coming years. As technological advancements in cell-based treatments continue, new therapies are expected to emerge that can address a wider range of diseases, including cancer, cardiovascular diseases, and genetic disorders. Government support for biotechnology, coupled with the rise of personalized medicine, will further drive market expansion. Regulatory improvements and greater adoption of these therapies by healthcare providers will also contribute to a positive market outlook, making it a dynamic and rapidly evolving sector in the Philippines.
Major Players
- Novartis
- Gilead Sciences
- Kite Pharma
- Regeneron Pharmaceuticals
- Mesoblast Limited
- Celgene Corporation
- Takeda Pharmaceutical Company
- Amgen
- Bristol-Myers Squibb
- BioMarin Pharmaceutical Inc.
- Gene Therapy, Inc.
- ImmunoCellularTherapeutics
- Humacyte
- Sangamo Therapeutics
- Bluebird Bio
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Pharmaceutical companies
- Biotechnology firms
- Healthcare providers
- Research institutions
- Contract manufacturing organizations (CMOs)
- End-users (hospitals, clinics, etc.)
Research Methodology
Step 1: Identification of Key Variables
This step involves identifying the key market variables such as product types, platform types, and end-user segments that impact market dynamics. It includes the analysis of current trends and regulatory influences.
Step 2: Market Analysis and Construction
Data is collected and analyzed from both primary and secondary sources to construct an accurate market model. This includes evaluating market size, competitive landscape, and growth potential.
Step 3: Hypothesis Validation and Expert Consultation
Expert consultations with industry leaders, healthcare professionals, and regulatory bodies help validate market hypotheses. This step ensures the accuracy and relevance of the research findings.
Step 4: Research Synthesis and Final Output
The research findings are synthesized into a comprehensive report, providing insights into the market’s current state, future projections, and actionable recommendations for stakeholders.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Growth Drivers
Rising Prevalence of Chronic Diseases
Technological Advancements in Gene Editing
Increasing Government Support for Biotech Initiatives - Market Challenges
High Cost of Treatment
Regulatory Hurdles in Cell Therapy Approval
Limited Healthcare Infrastructure - Market Opportunities
Expansion of Personalized Medicine
Rising Investment in Biotechnology Startups
Emerging Demand for Cell-based Regenerative Therapies - Trends
Growth in Regenerative Medicine Applications
Increased Focus on Immunotherapy Approaches
Adoption of AI and Machine Learning in Cell Therapy Research - Government Regulations
- SWOT Analysis of Key Competitors
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Stem Cell Therapy
Gene Therapy
Tissue Engineering
Immunotherapy
Cellular Immunotherapy - By Platform Type (In Value%)
Autologous Therapy
Allogeneic Therapy
Ex Vivo Therapy
In Vivo Therapy
Gene Editing Platforms - By Fitment Type (In Value%)
Hospital-based Solutions
Outpatient Clinic Solutions
Home-based Solutions
Laboratory-based Solutions
Contract Manufacturing Solutions - By EndUser Segment (In Value%)
Hospitals
Research Institutes
Biotechnology Companies
Contract Research Organizations (CROs)
- Market Share Analysis
- CrossComparison Parameters (System Type, Platform Type, Procurement Channel, EndUser Segment, Fitment Type)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Porter’s Five Forces
- Key Players
Novartis
Kite Pharma
Gilead Sciences
Celgene
Mesoblast Limited
Biologics, Inc.
AlloVir
Regeneron Pharmaceuticals
ImmunoCellular Therapeutics
Thermo Fisher Scientific
Bristol-Myers Squibb
Sanofi
Takeda Pharmaceuticals
Amgen
Bluebird Bio
- Growing Adoption of Stem Cell Therapies in Hospitals
- Increasing Research Collaboration in Biotech Companies
- Private Sector’s Role in Funding Regenerative Medicine
- Outpatient Clinics Leading the Adoption of Personalized Therapies
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035