Market Overview
The Philippines Diagnostic Biomarkers Market is currently experiencing significant growth, driven by advancements in medical technology and the increasing need for early disease detection. Based on a recent historical assessment, the market size in 2024 is valued at USD ~ billion. This growth is primarily fueled by an expanding healthcare infrastructure, rising awareness about personalized medicine, and the growing adoption of diagnostic biomarkers in cancer detection, genetic testing, and other chronic diseases. The demand for non-invasive diagnostic tests further accelerates the market’s expansion, supported by government initiatives aimed at improving healthcare access.
The Philippines is one of the dominant players in the Southeast Asian region for diagnostic biomarkers due to its improving healthcare infrastructure and increasing investment in advanced diagnostic technologies. Cities like Manila and Davao are at the forefront of this market, owing to their higher concentration of healthcare facilities, diagnostic centers, and research institutions. The government’s focus on enhancing healthcare services and investing in research and development initiatives has created a favorable environment for the market to thrive. Additionally, rising healthcare awareness and the shift towards personalized medicine are contributing to the growing adoption of diagnostic biomarkers across the country.
Market Segmentation
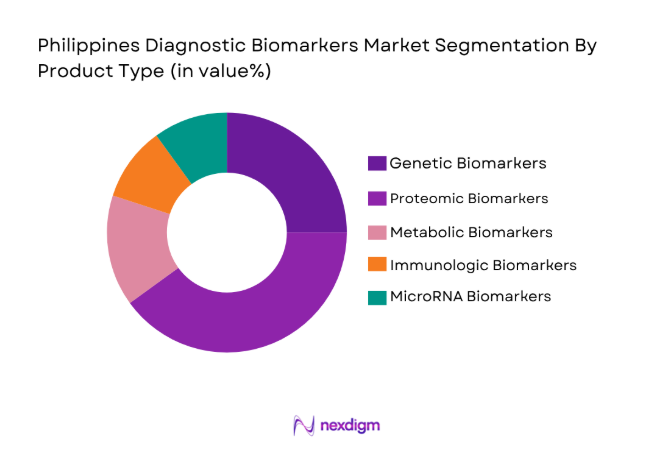
By Product Type
The Philippines Diagnostic Biomarkers market is segmented by product type into various sub-segments. Recently, the proteomic biomarkers sub-segment has gained a dominant market share due to the increasing use of proteomics in cancer diagnosis and other chronic disease monitoring. With the expanding scope of proteomics in early-stage disease detection, hospitals and diagnostic centers are increasingly adopting proteomic tests for their high precision and non-invasive nature. Furthermore, the increased demand for personalized medicine is driving the shift towards proteomic biomarkers, as they offer more targeted and efficient treatment approaches.
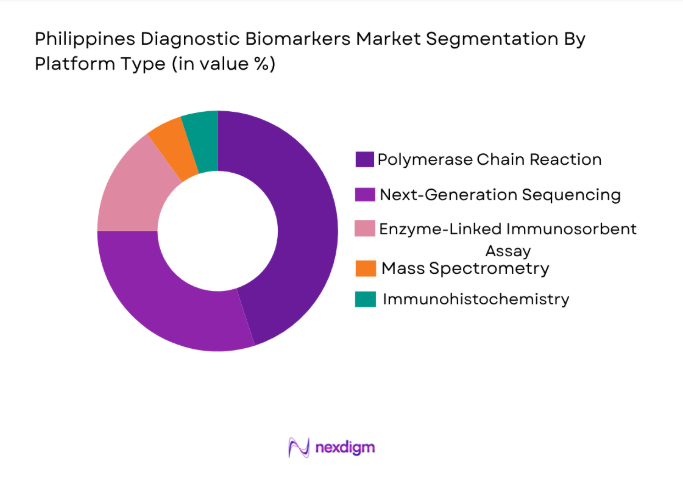
By Platform Type
The market is segmented into various platform types, with the polymerase chain reaction (PCR) platform leading the charge in the Philippines Diagnostic Biomarkers market. PCR is widely used due to its high sensitivity and ability to detect genetic markers associated with various diseases. The PCR platform is also versatile, allowing for the detection of a wide range of diseases, including cancer and genetic disorders, which further bolsters its adoption. Hospitals and diagnostic centers prefer PCR for its quick results, accuracy, and cost-effectiveness in diagnosing chronic diseases and infections.
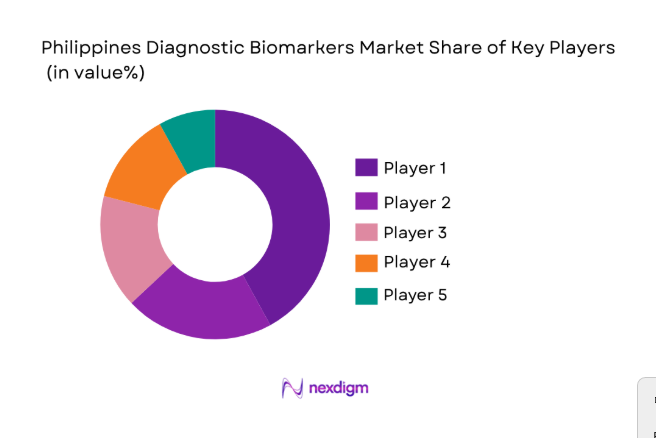
Competitive Landscape
The Philippines Diagnostic Biomarkers market is highly competitive, with both local and international players striving to capitalize on the increasing demand for advanced diagnostic solutions. Major players are focusing on strategic partnerships, technological innovations, and acquisitions to enhance their market position. This has led to a gradual consolidation of the market as companies work towards integrating advanced diagnostic technologies like artificial intelligence and machine learning in their platforms to improve accuracy and efficiency.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Market-Specific Parameter |
| Thermo Fisher Scientific | 1956 | USA | ~ | ~ | ~ | ~ | ~ |
| Abbott Laboratories | 1888 | USA | ~ | ~ | ~ | ~ | ~ |
| Roche | 1896 | Switzerland | ~ | ~ | ~ | ~ | ~ |
| Siemens Healthineers | 1847 | Germany | ~ | ~ | ~ | ~ | ~ |
| Bio-Rad Laboratories | 1952 | USA | ~ | ~ | ~ | ~ | ~ |
Philippines Diagnostic Biomarkers Market Analysis
Growth Drivers
Increasing Healthcare Awareness
As healthcare awareness continues to rise across the Philippines, patients are increasingly seeking early diagnostic solutions for chronic diseases. This growing awareness is driving the demand for diagnostic biomarkers, particularly in the fields of cancer, diabetes, and cardiovascular diseases, which have seen an increase in prevalence. Hospitals and diagnostic laboratories are responding by investing in state-of-the-art diagnostic equipment and systems to cater to the rising demand for accurate and reliable diagnostic tests. The awareness among patients about the benefits of early disease detection is leading to higher adoption rates of biomarker testing, as it helps in the timely identification and management of various diseases. This increased focus on early detection is contributing significantly to the growth of the diagnostic biomarkers market. Additionally, government initiatives focused on improving healthcare services across the country, particularly in underserved regions, are also playing a crucial role in further supporting the widespread use of diagnostic biomarkers, which in turn is driving market growth. The national healthcare policy’s emphasis on preventive care and the integration of diagnostic biomarkers into routine healthcare practices is poised to accelerate the market even further in the coming years.
Advancements in Personalized Medicine
Personalized medicine, which tailors treatments to individual patients based on genetic, proteomic, and other molecular factors, has become a key driver of growth in the Philippines Diagnostic Biomarkers market. The rising capabilities of genomic and proteomic research have paved the way for targeted therapies based on a patient’s unique genetic profile, leading to more effective treatments with fewer side effects. As healthcare providers increasingly adopt personalized medicine approaches, diagnostic biomarkers have become essential tools for identifying the right treatment for each patient. This shift away from traditional one-size-fits-all treatment models towards a more patient-centered approach has led to a significant increase in the demand for diagnostic biomarkers. The availability of biomarkers that can indicate disease susceptibility, guide treatment decisions, and monitor therapeutic effectiveness is driving the widespread adoption of personalized medicine across the country. Furthermore, the growing focus on precision healthcare, supported by both public and private sectors, is expected to continue to propel the demand for diagnostic biomarkers. This transformation in healthcare practices is not only changing treatment paradigms but also creating new opportunities for diagnostic biomarker providers in the Philippines market.
Market Challenges
High Cost of Diagnostic Tests
A major challenge facing the Philippines Diagnostic Biomarkers market is the high cost of advanced diagnostic tests. While the demand for diagnostic biomarkers is growing rapidly, the associated costs, especially for tests such as PCR, NGS, and proteomic analysis, remain a significant barrier. This challenge is particularly acute in rural areas where healthcare budgets are limited, and the affordability of these tests is a major concern. Many advanced diagnostic tests require sophisticated equipment and highly trained professionals, both of which contribute to elevated costs. These expenses often restrict access to cutting-edge diagnostic solutions, making it difficult for large segments of the population to benefit from early detection and personalized medicine. Despite efforts by the government to subsidize healthcare and make it more accessible, the high costs continue to be a critical challenge, limiting the adoption of diagnostic biomarkers among a significant portion of the population. This affordability issue not only affects individuals but also limits the expansion of diagnostic facilities, especially in underserved regions.
Regulatory and Approval Hurdles
Another key challenge in the Philippines Diagnostic Biomarkers market is the complex and lengthy regulatory process for the approval of diagnostic tests and biomarkers. Regulatory bodies, such as the Food and Drug Administration (FDA) Philippines, require comprehensive clinical trial data, rigorous documentation, and detailed testing before granting approval for new diagnostic products. This often results in delayed market entry for innovative biomarker technologies, which can slow down the overall growth of the market. The approval process is not only time-consuming but also resource-intensive, requiring significant investments in research and compliance. The lengthy timelines for regulatory approval and the difficulty of navigating regulatory requirements often discourage companies from introducing new and potentially game-changing diagnostic products. This regulatory bottleneck impacts the speed at which advancements in diagnostic biomarkers reach the healthcare sector, slowing down the potential benefits these technologies can offer in improving patient care and outcomes.
Opportunities
Expansion of Point-of-Care Diagnostics
The expansion of point-of-care (POC) diagnostics presents a significant opportunity in the Philippines Diagnostic Biomarkers market. POC diagnostic systems allow for faster, more accurate testing outside of traditional laboratory settings, making them particularly valuable in rural areas and remote locations with limited access to healthcare facilities. These systems enable immediate diagnosis, which is crucial in managing infectious diseases and chronic conditions in a timely manner. With the growing demand for rapid diagnostic solutions, especially in the management of infectious diseases like tuberculosis, dengue, and COVID-19, POC diagnostic biomarkers are becoming increasingly popular. The Philippine government’s focus on enhancing healthcare infrastructure, especially in underserved areas, creates an ideal environment for the adoption of POC diagnostic systems. The deployment of these systems in community health centers, rural clinics, and mobile units could significantly boost access to essential diagnostic services, improving healthcare outcomes for a larger portion of the population. The demand for these quick, affordable, and convenient diagnostic solutions presents a compelling growth opportunity for the market, driving the adoption of diagnostic biomarkers and expanding market penetration.
Increasing Adoption of Liquid Biopsy
The increasing adoption of liquid biopsy presents another promising opportunity for growth in the Philippines Diagnostic Biomarkers market. Liquid biopsy is a non-invasive testing method that uses blood or other bodily fluids to detect biomarkers associated with cancer and other diseases, offering a more convenient and less invasive alternative to traditional tissue biopsies. As liquid biopsy technology continues to improve, its ability to accurately detect and monitor diseases such as cancer has become more widely recognized. This method not only allows for earlier detection of cancers, but it also enables the monitoring of disease progression and therapeutic responses without the need for invasive procedures. The growing adoption of liquid biopsy in cancer diagnostics, along with its expanding applications in other medical fields, presents a significant opportunity for the market. With increasing technological advancements, higher accuracy, and reduced costs, liquid biopsy is expected to revolutionize the way diagnostic biomarkers are used in the Philippines, driving market growth and further integrating these solutions into routine clinical practice.
Future Outlook
The Philippines Diagnostic Biomarkers market is expected to witness robust growth over the next five years, driven by advancements in medical technology, an increasing shift towards personalized medicine, and expanding healthcare access in the region. Regulatory support and technological innovations are likely to drive demand for non-invasive diagnostic solutions, such as liquid biopsy and point-of-care diagnostics, which are gaining traction due to their cost-effectiveness and patient-friendly nature. Moreover, the government’s continued focus on healthcare infrastructure improvements and disease prevention will contribute to a favorable environment for market growth.
Major Players
- Thermo Fisher Scientific
- Abbott Laboratories
- Roche
- Siemens Healthineers
- Bio-Rad Laboratories
- Illumina
- Agilent Technologies
- PerkinElmer
- QIAGEN
- Luminex Corporation
- Beckman Coulter
- Danaher Corporation
- Abcam
- VWR International
- Labcorp
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Hospitals and diagnostic centers
- Research organizations and institutes
- Biotechnology companies
- Healthcare service providers
- Pharmaceutical companies
Research Methodology
Step 1: Identification of Key Variables
Identify critical variables that impact the diagnostic biomarkers market, including disease prevalence, healthcare infrastructure, and government policies.
Step 2: Market Analysis and Construction
Conduct comprehensive market analysis using both primary and secondary research methodologies to estimate market size and dynamics.
Step 3: Hypothesis Validation and Expert Consultation
Validate hypotheses through expert interviews and consultations with industry leaders, clinicians, and regulatory bodies.
Step 4: Research Synthesis and Final Output
Synthesize research findings into a comprehensive report, focusing on actionable insights, growth drivers, challenges, and opportunities.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Increasing Prevalence of Chronic Diseases
Technological Advancements in Biomarker Discovery
Government Initiatives for Healthcare Improvement - Market Challenges
High Costs of Diagnostic Tests
Regulatory Hurdles and Approval Delays
Limited Availability of Skilled Professionals - Market Opportunities
Expansion of Personalized Medicine
Growing Demand for Non-Invasive Diagnostic Solutions
Advancements in AI for Diagnostics - Trends
Integration of Artificial Intelligence in Diagnostics
Rising Demand for Liquid Biopsy in Cancer Diagnosis - Government Regulations
Data Protection and Privacy Regulations
Regulatory Framework for Biomarker Approvals
Government Funding for Healthcare Innovations - SWOT Analysis
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Genetic Biomarkers
Proteomic Biomarkers
Metabolic Biomarkers
Immunologic Biomarkers
MicroRNA Biomarkers - By Platform Type (In Value%)
Polymerase Chain Reaction (PCR)
Next-Generation Sequencing (NGS)
Enzyme-Linked Immunosorbent Assay (ELISA)
Mass Spectrometry
Immunohistochemistry - By Fitment Type (In Value%)
Laboratory-Based Diagnostics
Point-of-Care Diagnostics
Home-Based Diagnostics
Mobile Diagnostics - By End User Segment (In Value%)
Hospitals
Diagnostic Laboratories
Research Institutes
- Market Share Analysis
- Cross Comparison Parameters (System Type, Platform Type, End User Segment, Procurement Channel, Fitment Type, Technology Integration, Pricing Model)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Philippine Diagnostic Laboratories
Philippine Biotech
Sanofi
Pfizer
AbbVie
Roche
Thermo Fisher Scientific
Bio-Rad Laboratories
Abbott Laboratories
Siemens Healthineers
Illumina
Agilent Technologies
GE Healthcare
Becton Dickinson
Merck Group
- Increasing Adoption of Diagnostic Biomarkers in Clinical Settings
- Growth of Diagnostic Laboratories in Urban Areas
- Shift Toward Home-Based Diagnostic Solutions
- Rising Healthcare Awareness Among Consumers
- Increasing Adoption of Diagnostic Biomarkers in Clinical Settings
- Growth of Diagnostic Laboratories in Urban Areas
- Shift Toward Home-Based Diagnostic Solutions
- Rising Healthcare Awareness Among Consumers


