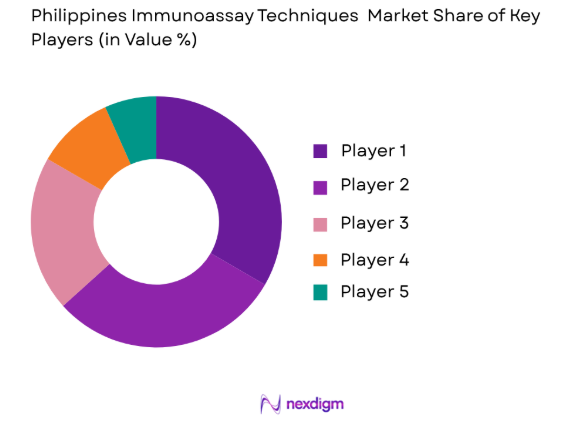
Market Overview
The Philippines Immunoassay Techniques Market is projected to reach a size of USD ~ billion based on a recent historical assessment. The market growth is driven by the increasing demand for diagnostic solutions, particularly for chronic diseases such as diabetes and cardiovascular conditions. Technological advancements in immunoassay systems, including the integration of artificial intelligence and automated platforms, have further accelerated market adoption. The rising healthcare expenditure in the Philippines, along with government initiatives to improve medical infrastructure, has contributed to this market expansion.
The dominant regions in the Philippines for immunoassay techniques are Metro Manila and Cebu. These areas are home to the most advanced healthcare facilities and the highest concentration of diagnostic laboratories. Metro Manila, in particular, leads due to its status as the country’s capital, offering a developed healthcare ecosystem supported by both public and private investments. Cebu follows as a growing hub for healthcare services, driven by both local and international demand for advanced diagnostic solutions.
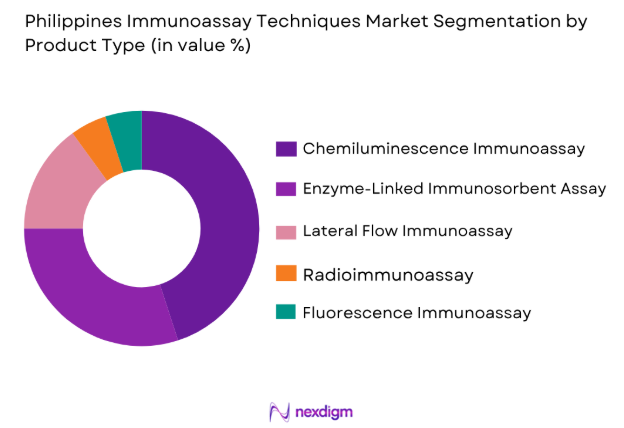
Market Segmentation
By Product Type
The Philippines Immunoassay Techniques Market is segmented by product type into various categories, including enzyme-linked immunosorbent assay (ELISA), chemiluminescence immunoassay (CLIA), and lateral flow immunoassay (LFIA). Currently, CLIA dominates the market share due to its high sensitivity and ability to detect lower levels of biomarkers, which is essential for diagnostic precision in clinical applications. CLIA’s widespread use in hospitals and diagnostic centers, especially for the detection of infectious diseases and biomarkers for cancer, contributes to its market leadership. The growing preference for automated platforms that offer higher throughput and enhanced accuracy further strengthens CLIA’s market position.
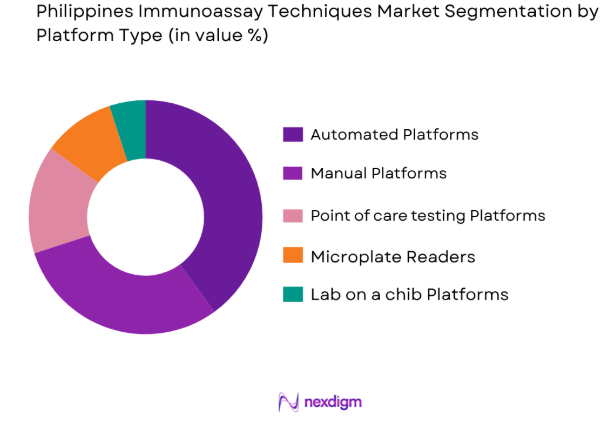
By Platform Type
The market is segmented by platform type into automated platforms, manual platforms, and point-of-care testing platforms. Automated platforms are dominating the market share due to their ability to handle high throughput, which meets the increasing demand for efficient and rapid diagnostic testing. The convenience, reduced human error, and faster results provided by automated systems are key factors in their dominance. Hospitals and diagnostic laboratories in the Philippines are increasingly adopting these platforms to improve operational efficiency and patient outcomes.
Competitive Landscape
The competitive landscape of the Philippines Immunoassay Techniques Market is marked by significant consolidation among key players. Major players dominate the market, often engaging in strategic collaborations, mergers, and acquisitions to expand their presence and enhance technological capabilities. The influence of these major players is further amplified through their strong distribution networks, partnerships with healthcare institutions, and continuous advancements in immunoassay technology.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD) | Regulatory Certifications |
| Roche Diagnostics | 1896 | Basel, Switzerland | ~ | ~ | ~ | ~ | ~ |
| Abbott Laboratories | 1888 | Chicago, USA | ~ | ~ | ~ | ~ | ~ |
| Siemens Healthineers | 1847 | Erlangen, Germany | ~ | ~ | ~ | ~ | ~ |
| Bio-Rad Laboratories | 1952 | Hercules, USA | ~ | ~ | ~ | ~ | ~ |
| PerkinElmer | 1937 | Waltham, USA | ~ | ~ | ~ | ~ | ~ |
Philippines Immunoassay Techniques Market Analysis
Growth Drivers
Technological Advancements in Immunoassay Platforms
The ongoing innovation in immunoassay technologies is a major growth driver in the Philippines market. The introduction of automated systems that integrate artificial intelligence and machine learning is transforming the landscape of diagnostics. These advancements enhance the accuracy, speed, and efficiency of testing, making them invaluable in detecting diseases at an earlier stage. The growing adoption of such systems in hospitals and diagnostic centers is a clear indication of their effectiveness in improving patient outcomes. Additionally, the development of multiplex assays, which allow for the detection of multiple biomarkers simultaneously, is expanding the scope of immunoassay applications in various medical fields, including oncology, cardiology, and infectious diseases. These innovations are fostering market growth by enabling faster diagnosis, better treatment plans, and more effective disease management strategies.
Government Healthcare Initiatives
Another key growth driver in the Philippines Immunoassay Techniques Market is the strong support from government healthcare initiatives aimed at improving the accessibility and quality of healthcare services. The Philippine government has been actively investing in healthcare infrastructure, focusing on expanding medical coverage and improving diagnostic capabilities, particularly in rural areas. Such initiatives include the National Health Insurance Program and the establishment of more diagnostic centers across the country, ensuring that the population has better access to advanced immunoassay testing. Furthermore, the government’s efforts to integrate digital health technologies into public healthcare systems further facilitate the growth of the immunoassay market. With an increasing emphasis on early detection and prevention of diseases, these government-driven efforts are expected to support the continued expansion of immunoassay solutions.
Market Challenges
High Cost of Advanced Immunoassay Systems
One of the primary challenges facing the Philippines Immunoassay Techniques Market is the high cost of advanced immunoassay systems. While newer systems offer superior performance in terms of accuracy, speed, and automation, they often come with a hefty price tag, making them less accessible to smaller healthcare facilities and diagnostic centers. The initial investment required for purchasing and setting up these advanced systems, along with the ongoing maintenance costs, can be a significant financial burden. This challenge is particularly evident in rural areas where healthcare budgets are limited. As a result, healthcare providers may be forced to rely on older, less efficient diagnostic systems, which could delay the adoption of newer technologies and hinder overall market growth.
Regulatory Hurdles in Device Approvals
Another challenge in the Philippines Immunoassay Techniques Market is the complex and often lengthy process of regulatory approvals for medical devices and diagnostic systems. The Philippine Food and Drug Administration (FDA) has strict regulations for medical devices, and navigating through these regulatory requirements can be a time-consuming and costly process. Delays in the approval process can slow down the introduction of new technologies into the market, hindering the growth of the immunoassay sector. Additionally, the need for continuous compliance with local regulations, including certifications and quality control standards, adds to the operational challenges faced by manufacturers and suppliers of immunoassay systems.
Opportunities
Growing Demand for Point-of-Care Diagnostics
The increasing demand for point-of-care (POC) diagnostics presents a significant opportunity for the Philippines Immunoassay Techniques Market. POC testing allows for rapid diagnostics at the patient’s location, providing immediate results without the need for central laboratory services. This is particularly valuable in rural areas where access to healthcare facilities may be limited. The Philippine government’s focus on improving healthcare accessibility in remote regions, combined with the growing demand for convenient and affordable testing solutions, is creating a favorable environment for the expansion of POC immunoassay systems. This trend is also being supported by the increasing prevalence of chronic diseases, which require regular monitoring, further driving the demand for POC diagnostic solutions.
Expansion of Immunoassay Solutions in Infectious Disease Testing
Another promising opportunity in the Philippines Immunoassay Techniques Market lies in the growing focus on infectious disease testing. With the rise of global health concerns such as the COVID-19 pandemic, there has been an increasing demand for fast and reliable diagnostic tools for detecting infectious diseases. Immunoassay techniques, particularly those based on lateral flow assays and ELISA, are well-suited for the detection of various pathogens, including viruses, bacteria, and parasites. As the Philippines continues to strengthen its public health infrastructure and improve disease surveillance, the demand for immunoassay-based testing solutions for infectious diseases is expected to rise. This presents an opportunity for market players to develop specialized diagnostic systems tailored to the needs of public health organizations and healthcare providers.
Future Outlook
The Philippines Immunoassay Techniques Market is expected to grow significantly over the next five years, driven by continued technological advancements, increased healthcare spending, and government initiatives to improve public health. Technological innovations, particularly in automation and AI integration, are set to enhance diagnostic accuracy and speed. Regulatory support and rising demand for point-of-care solutions will further propel the market forward, making immunoassay techniques increasingly accessible to a wider population. The increasing focus on infectious disease detection and early diagnosis will also contribute to market growth, with the Philippines emerging as a key player in Southeast Asia’s healthcare landscape.
Major Players
- Roche Diagnostics
- Abbott Laboratories
- Siemens Healthineers
- Bio-Rad Laboratories
- PerkinElmer
- ThermoFisher Scientific
- Becton Dickinson and Company
- Ortho Clinical Diagnostics
- Danaher Corporation
- Sysmex Corporation
- Fujifilm Holdings Corporation
- Mitsubishi ChemicalMedienceCorporation
- Medtronic
- Beckman Coulter
- ChembioDiagnostics
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Healthcare providers and hospitals
- Diagnosticcenters
- Pharmaceutical companies
- Research institutions
- Medical device distributors
- Point-of-care testing solution providers
Research Methodology
Step 1: Identification of Key Variables
The research process begins by identifying key market variables that affect the Philippines Immunoassay Techniques Market. These include technology trends, healthcare policies, and disease prevalence data.
Step 2: Market Analysis and Construction
Market data is collected through both primary and secondary research, including interviews with healthcare professionals, industry reports, and statistical data from reliable sources.
Step 3: Hypothesis Validation and Expert Consultation
A hypothesis is formulated based on the collected data, which is then validated through consultation with industry experts, medical professionals, and regulatory bodies.
Step 4: Research Synthesis and Final Output
The final research output is synthesized, integrating market insights, forecasts, and expert opinions, ensuring the report provides a comprehensive view of the market dynamics.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Increasing prevalence of chronic diseases
Advancements in diagnostic technology
Growing healthcare expenditure - Market Challenges
High cost of advanced immunoassay systems
Regulatory complexities in diagnostic approvals
Lack of skilled professionals in immunoassay techniques - Market Opportunities
Rising demand for point-of-care testing
Government initiatives for healthcare improvement
Expansion of diagnostic centers in rural areas - Trends
Integration of AI in diagnostic platforms
Increase in use of multiplex assays
Shift toward home-based diagnostic testing - Government Regulations
- SWOT Analysis of Key Competitors
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Enzyme-Linked Immunosorbent Assay (ELISA)
Radioimmunoassay (RIA)
Chemiluminescence Immunoassay (CLIA)
Fluorescence Immunoassay (FIA)
Lateral Flow Immunoassay (LFIA) - By Platform Type (In Value%)
Automated Platforms
Manual Platforms
Microplate Readers
Portable Devices
Lab-on-a-Chip Platforms - By Fitment Type (In Value%)
Benchtop Systems
Handheld Devices
Portable Kits
Fully Integrated Systems
Point-of-Care Systems - By End User Segment (In Value%)
Clinical Laboratories
Hospitals
Research Institutions
Diagnostic Centers
Pharmaceutical Companies - By Procurement Channel (In Value%)
Direct Procurement
Government Tenders
Online Procurement
Private Sector Procurement
Distributors & Resellers
- Market Share Analysis
- Cross Comparison Parameters (System Type, Platform Type, Procurement Channel, End User Segment, Fitment Type)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Porter’s Five Forces
Key Players
Roche Diagnostics
Abbott Laboratories
Siemens Healthineers
Bio-Rad Laboratories
Thermo Fisher Scientific
Danaher Corporation
PerkinElmer
Quidel Corporation
Ortho Clinical Diagnostics
Horiba Ltd
Becton Dickinson and Company
Sysmex Corporation
Fujifilm Holdings Corporation
Mitsubishi Chemical Medience Corporation
Medtronic
- Adoption of immunoassay techniques in hospitals
- Research institutions investing in immunoassay technology
- Pharmaceutical companies using immunoassays for R&D
- Growth of diagnostic centers in urban and rural regions
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035