Market Overview
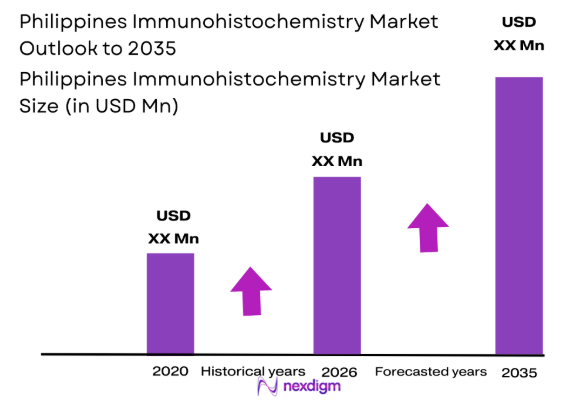
The Philippines Immunohistochemistry market is growing steadily, with increasing demand for advanced diagnostic tools in cancer and other diseases. Based on a recent historical assessment, the market size is valued at USD ~ million, driven by technological advancements, rising healthcare investments, and the increasing focus on personalized medicine. This growth is further supported by enhanced healthcare infrastructure, improved awareness of early disease detection, and the continuous innovation in immunohistochemistry technologies.
The Philippines market is dominated by key urban centers such as Metro Manila, Cebu, and Davao. These regions benefit from advanced medical infrastructure, significant investments in healthcare, and the presence of leading diagnostic and pharmaceutical companies. The accessibility of modern healthcare services in these cities, combined with a growing preference for early-stage diagnostics, further supports their dominance. Government initiatives promoting healthcare improvements also play a vital role in the market’s development.
Market Segmentation
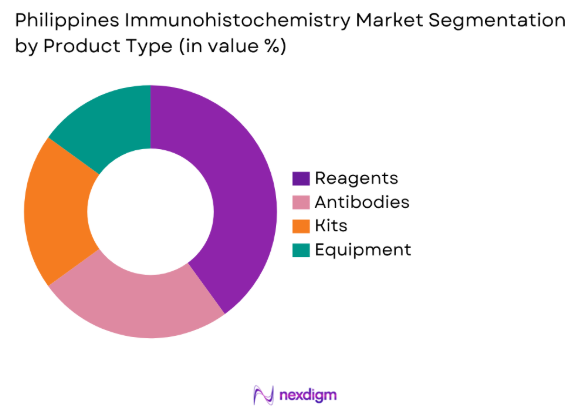
By Product Type
The Philippines Immunohistochemistry market is segmented by product type into reagents, antibodies, kits, and equipment. Reagents have a dominant market share due to their wide applicability and frequent usage in various diagnostic applications. The increasing adoption of IHC reagents in clinical diagnostics, particularly in oncology, has contributed significantly to their prevalence. The growing focus on early-stage disease detection and the preference for targeted treatments further boosts the demand for high-quality reagents in the healthcare system.
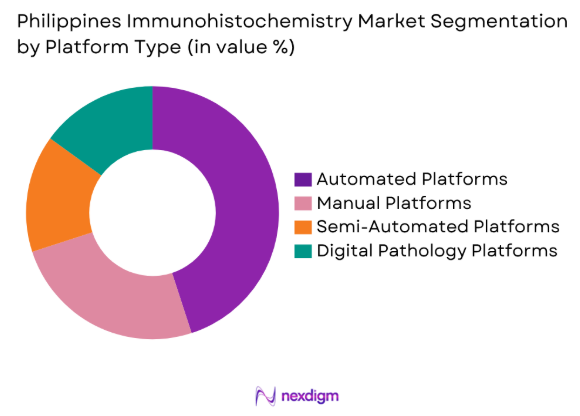
By Platform Type
The Philippines Immunohistochemistry market is segmented by platform type into manual, automated, semi-automated, and digital pathology platforms. Automated platforms have a significant share due to their efficiency, high throughput, and reduced human error. As the healthcare sector in the Philippines shifts towards digitalization and more streamlined processes, automated platforms are increasingly becoming the preferred choice in diagnostic laboratories. These platforms offer faster results and higher precision, contributing to their growing adoption.
Competitive Landscape
The competitive landscape in the Philippines immunohistochemistry market is characterized by the presence of both international and local players. The market is seeing consolidation as companies strive to expand their portfolios and technological capabilities through mergers, partnerships, and acquisitions. Major players are focusing on enhancing their product offerings through innovation and forming strategic alliances to increase market penetration.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Additional Parameter |
| Roche Diagnostics | 1896 | Switzerland | ~ | ~ | ~ | ~ | ~ |
| Abbott Laboratories | 1888 | USA | ~ | ~ | ~ | ~ | ~ |
| Thermo Fisher Scientific | 1956 | USA | ~ | ~ | ~ | ~ | ~ |
| Agilent Technologies | 1999 | USA | ~ | ~ | ~ | ~ | ~ |
| Bio-Rad Laboratories | 1952 | USA | ~ | ~ | ~ | ~ | ~ |
Philippines Immunohistochemistry Market Analysis
Growth Drivers
Technological Advancements in IHC
Technological advancements in immunohistochemistry are significantly driving market growth. Innovations like digital pathology platforms and automated IHC systems are transforming diagnostics by providing faster and more accurate results. The integration of artificial intelligence (AI) enhances IHC systems’ capabilities, enabling precision medicine and personalized treatments. Additionally, advances in biomarker discovery are fueling IHC demand, especially in oncology, where accurate disease staging and diagnosis are essential for tailored treatment plans. These technological improvements contribute to the increasing adoption of immunohistochemistry in routine diagnostics across hospitals, diagnostic centers, and research institutions, further supporting the growth of the market. As technology continues to evolve, the role of immunohistochemistry in disease detection and personalized treatment will expand, driving further market expansion.
Rising Demand for Personalized Medicine
The growing emphasis on personalized medicine is a key driver for the immunohistochemistry market. Personalized medicine relies on molecular diagnostics, with IHC playing a vital role in identifying molecular markers for diseases like cancer. By aligning treatments with an individual’s genetic profile, IHC enables more precise therapies, resulting in better patient outcomes. As chronic diseases and cancer continue to rise in the Philippines, the demand for personalized diagnostics is expected to increase significantly. This growing need for accurate, targeted treatments will drive further market expansion, positioning immunohistochemistry as an essential tool for enhancing personalized medicine and improving healthcare outcomes in the region.
Market Challenges
High Cost of IHC Products
A key challenge in the immunohistochemistry market is the high cost of IHC reagents, antibodies, and equipment. These expenses pose a significant barrier to widespread adoption, particularly in developing regions like the Philippines, where budget limitations impact healthcare infrastructure. Additionally, the costs of regular updates, training for healthcare professionals, and maintaining advanced IHC equipment further increase the overall financial burden. Despite the clear value of IHC in providing accurate diagnoses, the substantial upfront investment required by hospitals and laboratories can deter smaller institutions from adopting these technologies. As a result, cost-related concerns remain a critical challenge to the broader integration of immunohistochemistry in healthcare systems.
Regulatory Constraints and Quality Control
The regulatory environment for immunohistochemistry products is stringent, especially in the Philippines, where both local and international standards must be met to ensure patient safety and diagnostic accuracy. Obtaining regulatory approval for new IHC products, reagents, and equipment can be time-consuming and expensive. Additionally, strict quality control measures are required to maintain compliance with industry standards, further increasing costs. These regulatory constraints pose significant challenges for new market entrants and slow the pace of innovation, which limits the overall growth potential of immunohistochemistry applications. The regulatory complexities, while necessary for safety, can delay product introductions and hinder the expansion of the market.
Opportunities
Expansion of Diagnostic Capabilities
As hospitals and diagnostic centers in the Philippines upgrade their facilities, there is a significant opportunity for the expansion of immunohistochemistry products and technologies. The country’s healthcare system is growing rapidly, especially in urban areas, where advanced diagnostic methods like IHC are becoming essential. Public and private investments in healthcare infrastructure, along with government initiatives to improve accessibility, are creating opportunities for IHC companies to broaden their market reach. Hospitals, diagnostic centers, and academic institutions are increasingly adopting IHC technologies to improve disease diagnosis and management. This trend is expected to drive the continued growth of the immunohistochemistry market as demand for accurate diagnostics rises.
Growing Focus on Cancer Diagnostics
Cancer is a leading cause of death in the Philippines, making early detection essential for improving patient outcomes. Immunohistochemistry plays a vital role in diagnosing various cancers, particularly in tumor staging and determining appropriate treatment options. As awareness of early cancer detection grows and healthcare services continue to improve, the demand for immunohistochemistry in oncology diagnostics is expected to rise. This increasing demand presents a valuable opportunity for market players to innovate and develop specialized IHC solutions tailored to cancer diagnostics. By focusing on precision and personalized treatments, companies can position themselves for long-term success in this expanding market. The focus on early-stage cancer diagnosis will drive further adoption of IHC technologies in the Philippines healthcare system.
Future Outlook
The future of the Philippines immunohistochemistry market looks promising with continued advancements in diagnostic technologies, especially in digital pathology and AI integration. The next five years are expected to witness growth driven by rising healthcare investments, a growing focus on personalized medicine, and improved infrastructure in urban areas. Additionally, regulatory support for advanced diagnostic techniques will further accelerate market development. Increasing healthcare awareness and the demand for early-stage diagnostics, especially in cancer, will continue to drive market expansion.
Major Players
- Roche Diagnostics
- Abbott Laboratories
- Thermo Fisher Scientific
- Agilent Technologies
- Bio-Rad Laboratories
- PerkinElmer
- Danaher Corporation
- Leica Biosystems
- Fujifilm Holdings
- Merck KGaA
- Johnson & Johnson
- Bio-Techne Corporation
- Hologic
- Siemens Healthineers
- Beckman Coulter
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Hospitals and diagnostic centers
- Research institutes and academic institutions
- Pharmaceutical and biotechnology companies
- Healthcare technology developers
- Diagnostics equipment manufacturers
- System integrators
Research Methodology
Step 1: Identification of Key Variables
This step involves identifying the key factors influencing the immunohistochemistry market, including technological developments, regulatory trends, and market demand.
Step 2: Market Analysis and Construction
An in-depth market analysis is conducted to evaluate the market size, growth rate, and key factors contributing to the market’s evolution. This is followed by constructing a detailed market model.
Step 3: Hypothesis Validation and Expert Consultation
The developed hypotheses and models are validated through consultations with market experts and industry stakeholders to ensure their accuracy and relevance.
Step 4: Research Synthesis and Final Output
The final output includes synthesizing research findings into a comprehensive report that outlines market trends, growth drivers, challenges, and opportunities.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Increased Cancer Diagnoses
Technological Advancements in IHC
Rising Demand for Personalized Medicine - Market Challenges
High Cost of IHC Products
Regulatory Constraints on New Reagents
Limited Skilled Professionals - Market Opportunities
Expansion of Diagnostic Capabilities
Growing Prevalence of Chronic Diseases
Increased Investment in Healthcare Infrastructure - Trends
Shift Towards Digital Pathology
Advances in Companion Diagnostics
Increased Use of Monoclonal Antibodies - Government Regulations
- SWOT Analysis of Key Competitors
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Immunohistochemistry Stains
IHC Reagents
IHC Antibodies
IHC Kits
IHC Equipment - By Platform Type (In Value%)
Manual Platforms
Automated Platforms
Semi-Automated Platforms
Integrated Platforms
Digital Pathology Platforms - By Fitment Type (In Value%)
On-Premise Solutions
Cloud-Based Solutions
Hybrid Solutions
Integrated Solutions
Modular Solutions - By End User Segment (In Value%)
Hospitals
Diagnostic Laboratories
Research Institutes
Pharmaceutical Companies
CROs - By Procurement Channel (In Value%)
Direct Procurement
Distributor Networks
Online Procurement
Tendering and Bidding Platforms
Third-Party Vendors
- Market Share Analysis
- Cross Comparison Parameters (System Type, Platform Type, Procurement Channel, End User Segment, Fitment Type, Region, Price Range, Market Reach, Technological Integration, Service Support)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Roche Diagnostics
Abbott Laboratories
Thermo Fisher Scientific
Agilent Technologies
Bio-Rad Laboratories
Cell Signaling Technology
Merck KGaA
Johnson & Johnson
PerkinElmer
Leica Biosystems
Danaher Corporation
GE Healthcare
Fujifilm Holdings Corporation
Becton Dickinson
Sakura Finetek
- Increasing Demand from Hospitals
- Growth of Diagnostic Laboratories
- Emerging Role of CROs
- Research Institutes’ Need for High-Quality Diagnostics
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035