Market Overview
The Philippines predictive biomarkers market is driven by the increasing adoption of personalized medicine and advances in genomic research. Based on a recent historical assessment, the market size is expected to reach USD ~ billion by the end of the assessment period, supported by technological advancements in diagnostic tools such as next-generation sequencing (NGS) and polymerase chain reaction (PCR). Rising healthcare spending, increased prevalence of chronic diseases, and a shift toward precision medicine are key factors influencing this growth. These technologies are pivotal in improving early disease detection, personalized treatment, and monitoring therapeutic efficacy.
In terms of regional dominance, the Philippines stands out due to its rapidly evolving healthcare infrastructure, especially in urban areas like Metro Manila, Cebu, and Davao. These cities are major hubs for healthcare innovations and offer access to state-of-the-art technologies. A growing number of diagnostic laboratories, healthcare institutions, and biotechnology firms in these regions are investing in predictive biomarkers for early disease detection, driving market growth. The Philippines’ investment in healthcare modernization and government initiatives supporting personalized medicine also contribute to the dominance of these regions.
Market Segmentation
By System Type
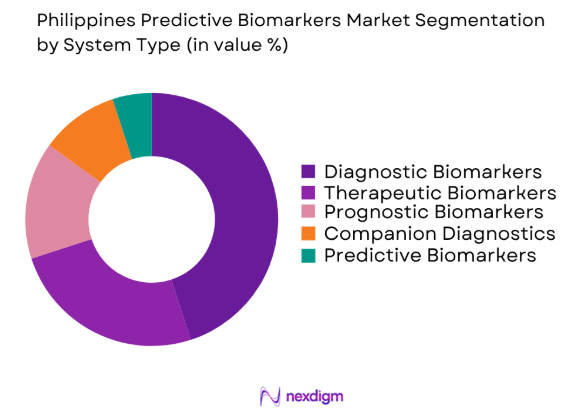
The Philippines predictive biomarkers market is segmented by system type into diagnostic biomarkers, therapeutic biomarkers, prognostic biomarkers, companion diagnostics, and predictive biomarkers. Recently, diagnostic biomarkers have a dominant market share due to factors such as increasing awareness about early disease detection, advancements in diagnostic technologies, and greater healthcare accessibility. These biomarkers are extensively used in identifying the presence of diseases, enabling earlier intervention and more personalized treatment plans.
By Platform Type
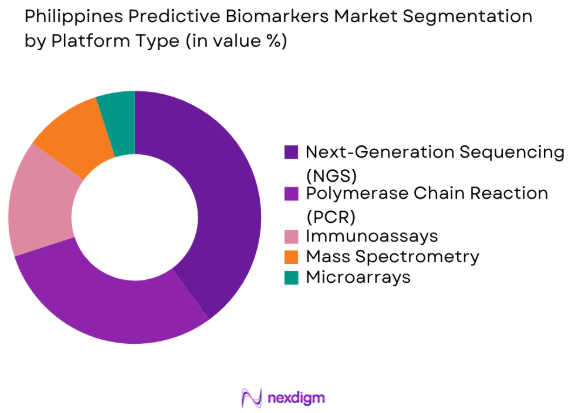
The Philippines predictive biomarkers market is segmented by platform type into polymerase chain reaction (PCR), next-generation sequencing (NGS), microarrays, immunoassays, and mass spectrometry. Recently, next-generation sequencing (NGS) has gained a dominant market share due to its high sensitivity, ability to detect genetic mutations, and efficiency in analyzing large volumes of genetic data. NGS is widely used for cancer diagnostics and personalized treatments, which makes it a preferred technology for clinicians.
Competitive Landscape
The competitive landscape of the Philippines predictive biomarkers market is marked by the presence of several prominent players focused on expanding their market share through technological innovation and strategic collaborations. These players are investing heavily in R&D to enhance their product offerings and drive demand for predictive biomarkers. The market is experiencing consolidation, with larger players acquiring smaller biotech firms to strengthen their portfolios.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD Million) | Additional Market-Specific Parameter |
| Thermo Fisher Scientific | 1956 | Waltham, MA, USA | ~ | ~ | ~ | ~ | ~ |
| Illumina | 1998 | San Diego, CA, USA | ~ | ~ | ~ | ~ | ~ |
| Abbott Laboratories | 1888 | Chicago, IL, USA | ~ | ~ | ~ | ~ | ~ |
| QIAGEN | 1984 | Hilden, Germany | ~ | ~ | ~ | ~ | ~ |
| Merck & Co. | 1891 | Kenilworth, NJ, USA | ~ | ~ | ~ | ~ | ~ |
Philippines Predictive Biomarkers Market Analysis
Growth Drivers
Technological Advancements in Genomic Testing
The continuous development of genomic testing technologies, such as next-generation sequencing (NGS), is one of the primary growth drivers for the Philippines predictive biomarkers market. These advancements allow for more precise detection of genetic mutations and the identification of disease biomarkers at an earlier stage, which enables personalized treatment plans. NGS, in particular, has revolutionized genetic testing by enabling the analysis of large DNA datasets, thus accelerating the discovery of novel biomarkers. This has resulted in a growing demand for predictive biomarkers in areas such as oncology, cardiology, and personalized medicine, fueling market growth. With the government and private sectors focusing on healthcare modernization, the adoption of genomic technologies will continue to expand, propelling the market forward.
Increasing Demand for Personalized Medicine
The growing demand for personalized medicine is a key driver in the expansion of the predictive biomarkers market in the Philippines. Personalized medicine, which tailors treatments based on individual genetic profiles, is rapidly gaining traction as a more effective approach to healthcare. By using biomarkers, healthcare providers can predict disease progression and tailor treatments to suit individual patients, enhancing treatment efficacy and reducing adverse effects. The Philippines’ healthcare system is increasingly focusing on precision medicine to improve treatment outcomes for diseases such as cancer, diabetes, and cardiovascular disorders. This shift towards personalized treatment approaches is driving the demand for predictive biomarkers, further contributing to the market’s growth.
Market Challenges
High Cost of Predictive Biomarker Testing
Despite the promising benefits of predictive biomarkers, the high cost of testing remains a significant challenge for market growth. The advanced technologies used in predictive biomarker testing, such as next-generation sequencing and PCR, require substantial investment in infrastructure and specialized equipment. Additionally, the cost of genetic testing and interpretation of results can be prohibitive, especially in emerging markets like the Philippines. While the market is growing, the accessibility and affordability of these tests remain a major concern. This limits the widespread adoption of predictive biomarkers, especially in rural areas where healthcare infrastructure may not be as developed, hindering overall market penetration.
Regulatory and Compliance Barriers
Regulatory challenges significantly hinder the growth of the predictive biomarkers market in the Philippines. The approval process for new diagnostic tests and biomarkers is lengthy and complex, involving stringent requirements for clinical validation and safety assessments. While these regulations ensure the accuracy and safety of biomarkers, they also delay the market introduction of innovative solutions. Moreover, the absence of harmonized regulations across regions complicates market entry, especially for international companies seeking to expand in the Philippines. These regulatory barriers create additional challenges for both local and global players, affecting the speed and efficiency of market adoption for new biomarker-based diagnostic technologies.
Opportunities
Expansion of Point-of-Care Solutions
Point-of-care (POC) solutions are gaining momentum in the Philippines, offering significant opportunities for the predictive biomarkers market. POC testing provides faster results and is more accessible compared to traditional laboratory-based diagnostic methods. The growing demand for rapid diagnostic results, especially for chronic disease management and emergency care, is driving the adoption of POC solutions in the country. By integrating predictive biomarkers with POC devices, healthcare providers can offer timely and personalized treatment, thus improving patient outcomes. This trend aligns with the government’s push to improve healthcare access, especially in remote areas. As healthcare infrastructure improves and the demand for POC testing grows, the market for predictive biomarkers in POC settings is expected to expand rapidly.
Collaboration with Biotechnology Firms
An emerging opportunity is the growing collaboration between diagnostic companies and biotechnology firms to advance biomarker discovery and validation. As the Philippines shifts toward personalized medicine, there is an increasing demand for accurate and validated biomarkers. Biotechnology firms, with their expertise in molecular biology and genetic research, are teaming up with diagnostic companies to uncover novel biomarkers for disease prediction and prognosis. This collaboration leads to the development of more precise diagnostic tools, crucial for early disease detection and treatment optimization. These partnerships are expected to drive innovation and growth in the predictive biomarkers market, offering new opportunities in the coming years.
Future Outlook
The Philippines predictive biomarkers market is expected to continue its growth trajectory over the next five years, driven by advancements in genomic technologies, increased adoption of personalized medicine, and the expansion of point-of-care solutions. Technological developments, such as the integration of artificial intelligence (AI) and machine learning (ML) in biomarker discovery, will further enhance the precision and efficiency of predictive biomarker tests. Additionally, regulatory support and government initiatives aimed at improving healthcare access and infrastructure will provide a conducive environment for market growth. With the rising demand for early disease detection and personalized treatment, the market is poised for continued expansion.
Major Players
- Thermo Fisher Scientific
- Illumina
- Abbott Laboratories
- QIAGEN
- Merck & Co.
- Roche Diagnostics
- Bio-Rad Laboratories
- PerkinElmer
- Agilent Technologies
- Siemens Healthineers
- GE Healthcare
- Becton Dickinson and Company
- Philips Healthcare
- Medtronic
- Johnson & Johnson
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Healthcare providers
- Pharmaceutical companies
- Biotechnology firms
- Diagnostic laboratories
- Research institutions
- Medical device manufacturers
Research Methodology
Step 1: Identification of Key Variables
Identification of critical variables, including market size, technology trends, and regulatory factors, through secondary research and expert consultations.
Step 2: Market Analysis and Construction
Comprehensive market analysis using data from reputable sources, primary interviews, and market reports to build a foundation for the study.
Step 3: Hypothesis Validation and Expert Consultation
Validating hypotheses with industry experts, key stakeholders, and market leaders to ensure accuracy and relevance.
Step 4: Research Synthesis and Final Output
Synthesizing research findings into a structured report, providing actionable insights, and market recommendations for stakeholders.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Increasing Prevalence of Chronic Diseases
Advances in Genomic Research
Rising Demand for Personalized Medicine - Market Challenges
High Cost of Predictive Biomarker Testing
Regulatory Challenges in Approvals
Technological Barriers and Integration - Market Opportunities
Expansion of Point-of-Care Solutions
Collaboration with Biotechnology Firms
Integration of AI in Predictive Diagnostics - Trends
Advancement of Liquid Biopsy Technology
Personalized Medicine for Cancer Treatment
Integration of Machine Learning in Biomarker Discovery - Government Regulations
- SWOT Analysis of Key Competitors
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Diagnostic Biomarkers
Therapeutic Biomarkers
Prognostic Biomarkers
Companion Diagnostics
Predictive Biomarkers - By Platform Type (In Value%)
Polymerase Chain Reaction (PCR)
Next-Generation Sequencing (NGS)
Microarrays
Immunoassays
Mass Spectrometry - By Fitment Type (In Value%)
Laboratory-based Solutions
Point-of-Care Solutions
Wearable Predictive Systems
Cloud-based Predictive Systems
Portable Predictive Devices - By End User Segment (In Value%)
Hospitals and Clinics
Research Institutions
Pharmaceutical Companies
Biotechnology Firms
Diagnostic Laboratories - By Procurement Channel (In Value%)
Direct Procurement
Online Procurement
Distributor Channels
Government Tenders
Private Sector Procurement
- Market Share Analysis
- Cross Comparison Parameters (System Type, Platform Type, Procurement Channel, End User Segment, Fitment Type, Diagnostic Technology, Market Reach, Regulatory Compliance, Price Sensitivity, Adoption Rate)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Thermo Fisher Scientific
Illumina, Inc.
AbbVie Inc.
Roche Diagnostics
Qiagen
Agilent Technologies
Merck & Co., Inc.
Novartis
Abbott Laboratories
Bio-Rad Laboratories
PerkinElmer
GE Healthcare
Siemens Healthineers
F. Hoffmann-La Roche AG
Becton Dickinson and Company
- Increasing Adoption of Predictive Biomarkers in Healthcare
- Role of Research Institutions in Advancing Biomarker Discovery
- Pharmaceutical Companies Driving Drug Development
- Technological Advancements in Diagnostic Laboratories
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035


