Market Overview
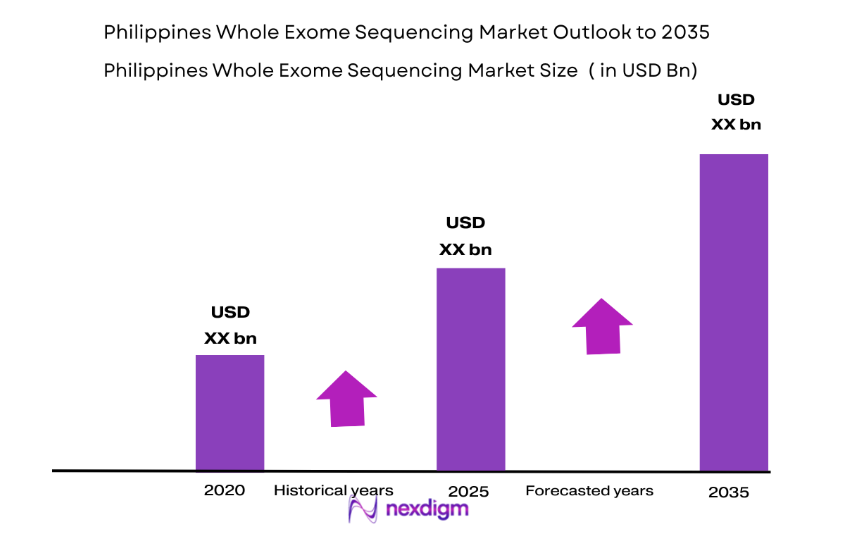
The Philippines Whole Exome Sequencing Market is driven by advancements in genomic research and increased government investment in healthcare infrastructure. Whole exome sequencing has gained significant traction as a tool for early diagnosis and personalized medicine, especially in cancer, genetic disorders, and rare diseases. As the healthcare system modernizes and awareness of genetic testing rises, the demand for whole exome sequencing is expected to grow rapidly. With support from both public and private sectors, the market is set to reach a substantial valuation, estimated at USD ~ million based on recent historical assessments. This growth reflects an expanding commitment to improving healthcare services and diagnostic capabilities across the region.
Dominant regions in the Philippines market include Metro Manila and Cebu, where healthcare infrastructure is most developed. These areas benefit from increased healthcare funding and have access to cutting-edge technology. The growing presence of hospitals, research institutions, and biotech companies in these cities has made them key players in driving the adoption of whole exome sequencing. Localized initiatives by government bodies and the increasing awareness of genetic testing also contribute to the market’s growth in these dominant regions.
Market Segmentation
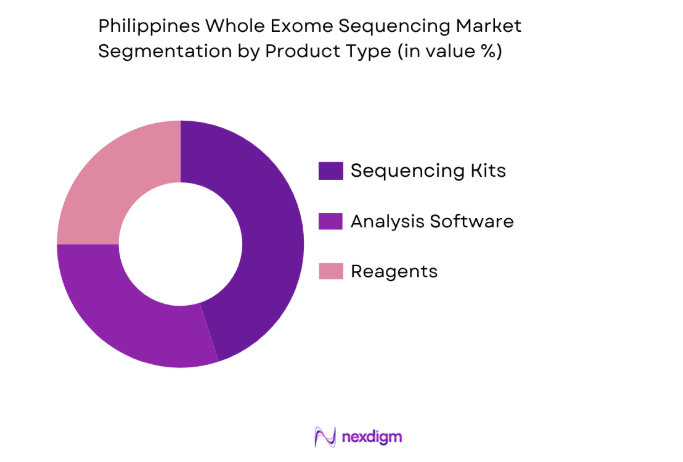
By Product Type:
The Philippines Whole Exome Sequencing market is segmented by product type into sequencing kits, analysis software, and reagents. Recently, sequencing kits have seen a dominant market share due to factors such as ease of use, cost-effectiveness, and the growing number of research institutions adopting them for diagnostic purposes. Furthermore, improvements in sequencing technology have made these kits more accurate and efficient, further driving their adoption across clinical laboratories and healthcare providers. As demand for precision medicine and genetic testing increases, the role of sequencing kits in diagnostics continues to grow, making it the dominant segment in the market.
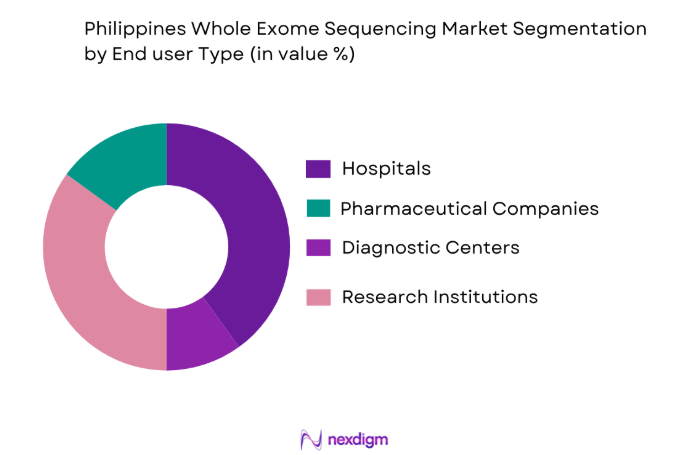
By End-User:
The market is also segmented by end-user into hospitals, diagnostic centers, research institutions, and pharmaceutical companies. Hospitals and diagnostic centers dominate the market share due to the rising demand for genetic testing in clinical settings. These facilities are increasingly adopting whole exome sequencing to offer personalized treatment plans, particularly for oncology and rare genetic disorders. Research institutions also play a crucial role, contributing to the growth by using this technology for advancing genomic research and exploring new disease treatments. As the healthcare sector continues to integrate genomic diagnostics, hospitals and diagnostic centers will remain the leading end-users of this technology.
Competitive Landscape
The Philippines Whole Exome Sequencing Market is characterized by a competitive landscape with both global and regional players. Leading companies are investing heavily in research and development to expand their product offerings and maintain a competitive edge. Strategic partnerships between local healthcare providers and international genomics firms are enhancing market consolidation. The influence of key players such as Illumina, Thermo Fisher Scientific, and Roche has significantly contributed to market growth, with these companies focusing on technology innovation, regulatory compliance, and expanding distribution channels.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Market-Specific Parameter |
| Illumina | 1998 | USA | ~ | ~ | ~ | ~ | ~ |
| Thermo Fisher Scientific | 2006 | USA | ~ | ~ | ~ | ~ | ~ |
| Roche | 1896 | Switzerland | ~ | ~ | ~ | ~ | ~ |
| Agilent Technologies | 1999 | USA | ~ | ~ | ~ | ~ | ~ |
| Bio-Rad Laboratories | 1952 | USA | ~ | ~ | ~ | ~ | ~ |
Philippines Whole Exome Sequencing Market Analysis
Growth Drivers
Technological Advancements in Genomic Sequencing:
The rapid advancements in whole exome sequencing technology are a major growth driver for the market. Continuous improvements in sequencing accuracy, speed, and affordability have significantly increased the adoption of this technology across hospitals and diagnostic centers. With the introduction of next-generation sequencing platforms, the ability to analyze genetic data has become faster and more cost-effective, thus driving demand in clinical diagnostics. This trend is expected to continue as innovations in bioinformatics and sequencing technology enhance diagnostic capabilities, particularly in cancer genomics and rare disease identification. The increasing precision of genomic analysis also facilitates personalized medicine, further boosting the demand for whole exome sequencing.
Government Support and Healthcare Infrastructure Expansion:
Government initiatives and investments in healthcare infrastructure are vital drivers of growth in the Philippines Whole Exome Sequencing Market. With the increasing recognition of the importance of genomics in healthcare, the government has been supporting the integration of genetic testing in public healthcare programs. Investments in healthcare infrastructure, especially in urban areas like Metro Manila and Cebu, enable hospitals and diagnostic centers to adopt advanced technologies, including whole exome sequencing. Government funding aimed at improving healthcare accessibility and precision medicine is expected to fuel further market expansion. The growing focus on disease prevention, early diagnosis, and personalized treatment plans underpins the increased adoption of whole exome sequencing.
Market Challenges
High Initial Cost of Whole Exome Sequencing Systems:
One of the key challenges for the Philippines Whole Exome Sequencing Market is the high initial cost of sequencing systems and associated infrastructure. Despite the declining costs of sequencing over the years, the upfront investment required for establishing and maintaining whole exome sequencing laboratories remains significant. Smaller hospitals and diagnostic centers, particularly in rural areas, face difficulty in affording these technologies. While larger institutions can absorb these costs, the high investment remains a barrier for widespread adoption, limiting the market’s growth potential in less-developed regions. Furthermore, the need for specialized training and expertise to operate these advanced systems adds to the overall expense.
Regulatory Hurdles and Data Privacy Concerns:
The Philippines faces regulatory challenges in the widespread implementation of whole exome sequencing. Stringent government regulations concerning genetic data privacy and protection can slow down the adoption of this technology. The Philippines’ regulatory framework for genetic testing is still evolving, with concerns over data handling and patient consent creating hurdles for market players. The delay in developing clear and standardized regulations for genetic testing and data usage could hinder the expansion of whole exome sequencing technologies in the healthcare sector. Addressing these concerns will require more effective regulatory policies and clearer guidelines for data protection and privacy.
Opportunities
Expansion of Personalized Medicine Initiatives:
The growing demand for personalized medicine presents a significant opportunity for the whole exome sequencing market. Personalized medicine, which tailors medical treatments to individual genetic profiles, relies heavily on genomic testing. As healthcare providers increasingly shift toward individualized treatment plans, the adoption of whole exome sequencing to identify genetic mutations and predict disease susceptibility will continue to rise. This trend is particularly evident in the treatment of cancers, genetic disorders, and rare diseases, where genomic testing plays a crucial role in formulating personalized therapy. The continued expansion of personalized medicine initiatives provides a promising avenue for the market’s growth in the Philippines.
Collaboration Between Healthcare Providers and Biotechnology Firms:
Another significant opportunity lies in the increasing collaboration between healthcare providers and biotechnology firms to integrate whole exome sequencing into clinical practice. Research institutions and biotech companies are actively partnering with hospitals and diagnostic centers to develop targeted therapies and improve patient outcomes through genomic testing. These collaborations help bring cutting-edge sequencing technologies to the market more efficiently, making whole exome sequencing more accessible to a wider population. As these partnerships grow, the market for whole exome sequencing in the Philippines is expected to see rapid expansion.
Future Outlook
Over the next five years, the Philippines Whole Exome Sequencing Market is expected to experience strong growth driven by technological advancements and increasing healthcare infrastructure investments. With ongoing improvements in sequencing accuracy and affordability, whole exome sequencing will become more accessible, especially in urban healthcare settings. Technological developments in AI and bioinformatics will play a significant role in enhancing the capabilities of whole exome sequencing, enabling faster and more precise genetic analysis. Additionally, government initiatives focused on personalized medicine and disease prevention will further support market growth, creating a conducive environment for market players to expand their reach.
Major Players
- Illumina
- ThermoFisher Scientific
- Roche
- Agilent Technologies
- Bio-Rad Laboratories
- Pacific Biosciences
- Qiagen
- PerkinElmer
- Oxford Nanopore Technologies
- CambridgeEpigenetix
- Danaher Corporation
- Illumina Inc.
- Beckman Coulter
- Fluidigm Corporation
- Cepheid
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Large healthcare corporations
- Pharmaceutical companies
- Biotechnology firms
- Diagnostic laboratories
- Research institutions
- Hospitals
Research Methodology
Step 1: Identification of Key Variables
This step involves identifying the key factors that drive the whole exome sequencing market, such as technological advancements, market demand, and regulatory factors. Understanding these variables is essential for building the research framework.
Step 2: Market Analysis and Construction
Market segmentation and construction are based on comprehensive research that includes data from reliable sources, government publications, and industry reports. A detailed understanding of the key market segments is crucial for accurate projections.
Step 3: Hypothesis Validation and Expert Consultation
This step ensures the validity of the market hypotheses by consulting industry experts, researchers, and stakeholders in the healthcare and biotechnology sectors. The feedback gathered helps refine the research model.
Step 4: Research Synthesis and Final Output
In this final step, all the research findings are synthesized into a comprehensive market report, highlighting key trends, growth drivers, challenges, and future outlook, providing actionable insights for stakeholders.
- Executive Summary
- Research Methodology [Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks]
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Growth Drivers
Increasing Adoption of Personalized Medicine
Advancements in Genomic Research
Growing Healthcare Infrastructure in Philippines - Market Challenges
High Initial Cost of Whole Exome Sequencing Systems
Lack of Skilled Personnel in Genomic Research
Regulatory Barriers in Genomic Data Use - Market Opportunities
Government Initiatives in Healthcare Research
Rising Demand for Genetic Testing in Rural Areas
Potential for Collaborative Research with International Institutions - Trends
Integration of AI in Genomic Research
Growth in Non-Invasive Testing Methods
Increase in Health Data Collection via Genomics - Government Regulations
- By Market Value 2020-2025
- By Installed Units 2020-2025
- By Average System Price 2020-2025
- By System Type (In Value%)
Next-Generation Sequencing Systems
Illumina-Based Systems
Ion Torrent Systems
PacBio Systems
Oxford Nanopore Systems - By Platform Type (In Value%)
Clinical Sequencing Platforms
Research Sequencing Platforms
Targeted Sequencing Platforms
Cloud-Based Platforms
Laboratory-Based Platforms - By Fitment Type (In Value%)
Standalone Systems
Integrated Systems
Portable Systems
Cloud-Based Solutions
Modular Systems - By EndUser Segment (In Value%)
Hospitals & Diagnostic Centers
Research Institutions
Biotech & Pharma Companies
Clinical Laboratories
- Market Share Analysis
- CrossComparison Parameters (System Type, Platform Type, Procurement Channel, EndUser Segment, Fitment Type)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Porter’s Five Forces
- Key Players
Illumina
Thermo Fisher Scientific
Pacific Biosciences
Oxford Nanopore Technologies
Qiagen
PerkinElmer
Agilent Technologies
Bio-Rad Laboratories
Promega Corporation
Siemens Healthineers
Danaher Corporation
Becton Dickinson
Roche
Hudson Robotics
Geneious
- Hospitals Increasing Investment in Genomic Diagnostics
- Research Institutions Expanding Genomic Study Programs
- Biotech Companies Developing Genomic-Based Therapies
- Clinical Laboratories Integrating Whole Exome Sequencing Services
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035