Market Overview
The South Korea diagnostic labs market is experiencing significant growth driven by the increasing demand for diagnostic services and technological advancements in medical devices. Based on a recent historical assessment, the market size in South Korea is expected to reach USD ~ billion. This growth is largely propelled by factors such as the aging population, rising healthcare needs, and government initiatives aimed at improving healthcare infrastructure. Innovations in diagnostic technologies such as AI and automation have also bolstered the market’s expansion.
Dominant cities in South Korea, such as Seoul and Busan, continue to lead the diagnostic labs market due to their robust healthcare infrastructure and higher concentration of hospitals and healthcare facilities. The capital city, Seoul, is at the forefront of adopting advanced diagnostic technologies, which is further supported by government policies and investments in health technology. Additionally, the presence of major healthcare companies and research institutions in these regions contributes to their dominance, making them key players in the national healthcare landscape.
Market Segmentation
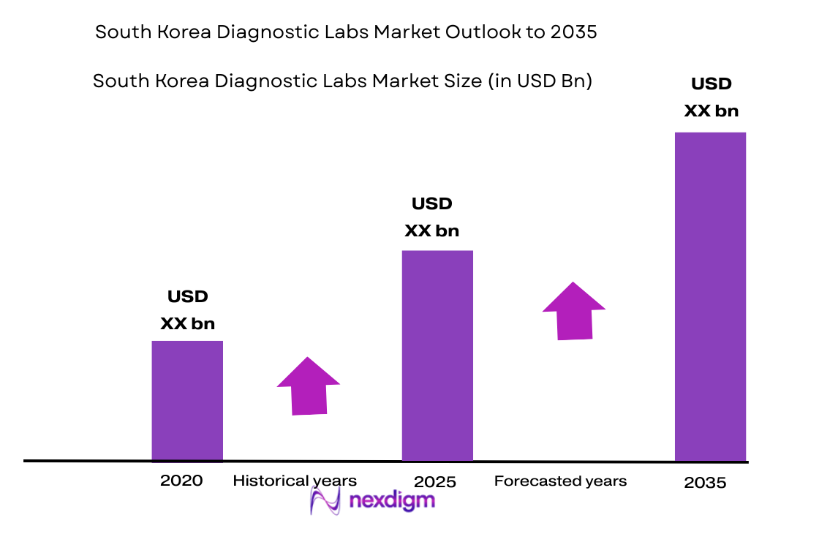
By Product Type
The South Korea diagnostic labs market is segmented by product type into clinical chemistry systems, hematology systems, immunoassay systems, microbiology systems, and molecular diagnostics systems. Recently, clinical chemistry systems have captured the largest share of the market due to the rising demand for laboratory-based tests and diagnostics that are essential for monitoring chronic conditions. These systems are highly valued for their accuracy, reliability, and broad application across various medical conditions, making them a top choice among healthcare providers.
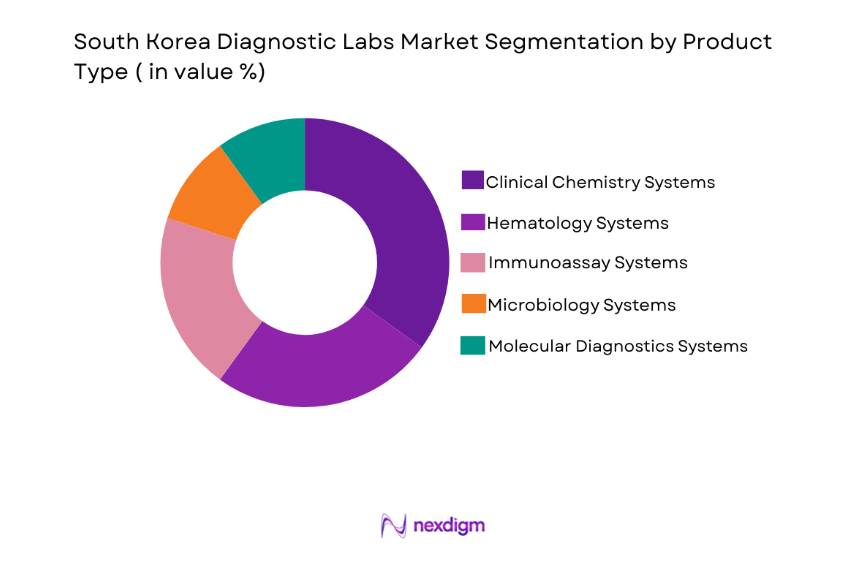
By Platform Type
The South Korea diagnostic labs market is also segmented by platform type into automated diagnostic platforms, point-of-care diagnostic platforms, laboratory diagnostic platforms, telemedicine diagnostic platforms, and wearable diagnostic platforms. Recently, automated diagnostic platforms have gained significant traction due to their efficiency, accuracy, and the increasing adoption of automation in medical diagnostics. These platforms enhance laboratory workflows and are widely used for high-throughput testing, making them dominant in both hospital settings and diagnostic labs.
Competitive Landscape
The South Korea diagnostic labs market is highly competitive, with consolidation occurring due to the influence of major healthcare players and global diagnostic companies. The presence of leading companies with strong research and development capabilities has created a competitive environment where innovation plays a key role in market positioning. The market dynamics are shaped by technological advancements, partnerships between local and international players, and the increasing focus on patient-centered care.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD) | Additional Parameter |
| Samsung Medison | 1985 | Seoul, South Korea | ~ | ~ | ~ | ~ | ~ |
| LG Electronics | 1958 | Seoul, South Korea | ~ | ~ | ~ | ~ | ~ |
| Siemens Healthineers | 1847 | Erlangen, Germany | ~ | ~ | ~ | ~ | ~ |
| Roche Diagnostics | 1896 | Basel, Switzerland | ~ | ~ | ~ | ~ | ~ |
| Thermo Fisher | 1956 | Waltham, USA | ~ | ~ | ~ | ~ | ~ |
South Korea Diagnostic Labs Market Analysis
Growth Drivers
Increasing Healthcare Investments
The South Korean government has made substantial investments in healthcare infrastructure, diagnostic technologies, and medical research, significantly driving the growth of the diagnostic labs market. These investments are part of broader national initiatives aimed at improving healthcare access, reducing health disparities, and ensuring that high-quality healthcare is available to all citizens, particularly the aging population. The government has also prioritized the adoption of state-of-the-art technologies, including AI-powered diagnostics, which have been instrumental in enhancing the accuracy, efficiency, and accessibility of diagnostic services. These investments are directed toward the establishment of advanced diagnostic labs equipped with the latest technologies, allowing healthcare providers to offer better and more accurate diagnostic services. Furthermore, government initiatives are fostering collaboration between private and public entities, enabling the development and integration of innovative solutions into diagnostic processes. As these healthcare investments continue to grow, the demand for sophisticated diagnostic services has also risen, further fueling the market’s expansion. With better infrastructure and modern diagnostic tools, diagnostic labs have become an integral part of South Korea’s evolving healthcare ecosystem, making them central to the country’s efforts to provide top-tier healthcare services.
Rising Chronic Diseases and Aging Population
South Korea’s rapidly aging population, coupled with the rising prevalence of chronic diseases such as diabetes, hypertension, and heart disease, has significantly contributed to the increasing demand for diagnostic services. The country’s elderly population, which is one of the fastest-growing demographic groups, requires constant monitoring for chronic conditions and early detection of diseases, thereby creating a robust need for routine diagnostic tests. Furthermore, chronic diseases demand long-term care and regular screenings, which has heightened the demand for reliable and timely diagnostic results. As a result, diagnostic labs are seeing a surge in patient visits and the need for more specialized tests, including molecular diagnostics, genetic testing, and other advanced technologies. This demographic shift has put increased pressure on healthcare systems to provide high-quality and efficient diagnostic services. Consequently, diagnostic labs are being pushed to not only expand their service offerings but also enhance their technological capabilities to keep up with the growing demand. As the healthcare system adapts to these changes, diagnostic labs will continue to play a crucial role in managing the health of the aging population, further driving market growth.
Market Challenges
Regulatory Challenges
The South Korean diagnostic labs market faces significant regulatory challenges, which create barriers to market growth. Diagnostic equipment and testing technologies are subject to stringent regulatory standards and certification processes, which can delay product approval and market entry. The regulatory framework in South Korea is complex, with rules and guidelines that are constantly evolving to keep up with advancements in healthcare and technology. Companies seeking to enter or expand in the market must navigate these regulatory hurdles, which can be resource-intensive and time-consuming. These compliance requirements can also be particularly challenging for smaller companies, which may lack the resources to manage complex regulatory demands. Additionally, the need to comply with international standards for data security and privacy, especially in relation to patient data, adds another layer of complexity to the regulatory environment. As regulations continue to tighten globally, diagnostic labs in South Korea will need to invest in staying ahead of compliance requirements, further increasing operational costs. These regulatory challenges could slow the growth of new diagnostic technologies and equipment, limiting the speed at which the market can evolve and meet growing demand.
Skilled Workforce Shortage
A significant challenge facing the South Korean diagnostic labs market is the shortage of skilled technicians and healthcare professionals who are qualified to operate advanced diagnostic equipment and interpret complex test results. The rapid pace of technological advancements in the diagnostic field requires a workforce that is highly trained and capable of handling sophisticated tools, from automated testing platforms to AI-based diagnostic systems. However, the demand for such skilled professionals far exceeds the available supply. As new technologies become more widespread, the need for workers who are adept at utilizing and maintaining these systems has intensified. Without a sufficient number of trained professionals, diagnostic labs face difficulties in providing fast, accurate, and high-quality services, which impacts both operational efficiency and patient outcomes. Additionally, the shortage of skilled workers hampers the ability of diagnostic labs to expand their operations or scale up to meet increasing demand. To address this, the industry will need to focus on educational initiatives and workforce development, including specialized training programs to equip professionals with the skills needed for modern diagnostic environments. Until these challenges are overcome, the growth potential of diagnostic labs will remain constrained.
Opportunities
Expansion of At-Home Diagnostic Services
As consumer demand for convenience and accessibility increases, at-home diagnostic services are emerging as a significant growth opportunity in South Korea’s diagnostic labs market. These services allow individuals to perform diagnostic tests and monitor their health from the comfort of their homes, offering a more personalized and timely approach to healthcare. The ability to conduct tests at home can save time, reduce travel costs, and provide a more convenient solution for patients who require frequent monitoring, such as those with chronic conditions. Furthermore, with the rapid advancement of telemedicine, at-home diagnostic services can integrate seamlessly with virtual consultations, allowing for a more holistic healthcare experience. South Korean consumers are becoming increasingly comfortable with home-based solutions, especially as digital health tools and devices become more widespread. For diagnostic labs, this trend presents an opportunity to diversify their service offerings by providing kits for home use, offering guidance through telemedicine consultations, and enabling remote testing. As telemedicine and home testing technologies continue to advance, diagnostic labs can tap into this growing segment, broadening their market reach while enhancing service delivery. The expansion of at-home diagnostic services is not only an attractive growth opportunity but also aligns with the broader trend of personalized healthcare, positioning diagnostic labs to meet evolving consumer preferences.
Integration of Artificial Intelligence and Automation
The integration of artificial intelligence (AI) and automation into diagnostic platforms presents significant opportunities for growth in the South Korean diagnostic labs market. As the healthcare industry increasingly adopts AI-powered technologies, diagnostic labs can leverage AI to improve the accuracy of diagnoses, reduce human error, and accelerate the processing of diagnostic tests. AI algorithms can analyze large volumes of data from diagnostic tests, such as imaging, pathology, and genomics, to provide quicker, more accurate results, while also identifying patterns that may not be immediately apparent to human clinicians. Automation, on the other hand, enhances operational efficiency by streamlining lab workflows, minimizing manual intervention, and allowing labs to handle larger volumes of tests. Automated systems can be particularly valuable in high-demand diagnostic environments, enabling labs to process tests more rapidly and cost-effectively. With the continued advancements in AI and machine learning, diagnostic labs have the potential to not only improve patient outcomes but also optimize resource allocation, making operations more sustainable in the long term. As AI technology becomes more advanced and accessible, diagnostic labs can integrate these innovations into their systems, enhancing their competitive edge and positioning themselves for future success.
Future Outlook
The South Korea diagnostic labs market is poised for steady growth over the next few years, with technological advancements and increased demand for personalized healthcare driving the market. Continued investments in healthcare infrastructure and diagnostic technology will play a crucial role in shaping the market’s future. The adoption of AI, automation, and telemedicine will further enhance diagnostic capabilities, creating a more efficient and patient-centered healthcare system. Strong regulatory support will ensure the sector’s expansion while maintaining high standards for quality and accuracy in diagnostics.
Major Players
- Samsung Medison
- LG Electronics
- Siemens Healthineers
- Roche Diagnostics
- Thermo Fisher
- GE Healthcare
- Philips Healthcare
- Mindray
- Sysmex Corporation
- Beckman Coulter
- Hitachi High-Tech
- Agilent Technologies
- Bio-Rad Laboratories
- Abbott Laboratories
- Panasonic Healthcare
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Healthcare providers and hospitals
- Diagnostic lab operators
- Healthcare technology developers
- Medical equipment distributors
- Pharmaceutical companies
Research Methodology
Step 1: Identification of Key Variables
The identification of key variables involved in market dynamics, including market drivers, challenges, and trends, was performed through primary and secondary research.
Step 2: Market Analysis and Construction
The market was analyzed based on historical data and current trends. Market segmentation was also constructed using input from industry experts and reliable sources.
Step 3: Hypothesis Validation and Expert Consultation
Hypotheses about market trends and growth were validated through expert consultations, including discussions with industry professionals, analysts, and company representatives.
Step 4: Research Synthesis and Final Output
The final research synthesis was prepared by combining all data and insights, ensuring accuracy and consistency, and presenting it in a comprehensive report.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Growth Drivers
Increased Government Spending on Healthcare Infrastructure
Rising Prevalence of Chronic Diseases
Technological Advancements in Diagnostic Equipment - Market Challenges
High Operational Costs
Regulatory Compliance Challenges
Shortage of Skilled Technicians - Market Opportunities
Expansion of Telemedicine Services
Growth of At-Home Diagnostic Services
Increased Adoption of AI and Automation in Diagnostics - Trends
Integration of AI in Diagnostic Platforms
Shift Toward Personalized Healthcare - Government Regulations
- SWOT Analysis
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Clinical Chemistry Systems
Hematology Systems
Immunoassay Systems
Microbiology Systems
Molecular Diagnostics Systems - By Platform Type (In Value%)
Automated Diagnostic Platforms
Point-of-Care Diagnostic Platforms
Laboratory Diagnostic Platforms
Telemedicine Diagnostic Platforms
Wearable Diagnostic Platforms - By Fitment Type (In Value%)
In-House Diagnostics
Outsourced Diagnostics
Home-Based Diagnostics
Mobile Diagnostics - By End User Segment (In Value%)
Hospitals
Diagnostic Centers
Research Laboratories
- Market Share Analysis
- Cross Comparison Parameters (System Type, Platform Type, Fitment Type, End User Segment, Technology Adoption, Regulatory Compliance, Pricing Strategy)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Samsung Medison
LG Electronics
Siemens Healthineers
Abbott Laboratories
Roche Diagnostics
Thermo Fisher Scientific
Danaher Corporation
Philips Healthcare
GE Healthcare
Mindray
Sysmex Corporation
Beckman Coulter
Hitachi High-Tech
Agilent Technologies
Bio-Rad Laboratories
- Hospitals’ Growing Investment in Diagnostic Equipment
- Shift Toward Specialization in Diagnostic Centers
- Adoption of Advanced Diagnostics in Research Laboratories
- Increased Integration of Diagnostics in Primary Care Settings
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035