Market Overview
The Turkey medical devices market has witnessed substantial growth, driven by a combination of factors such as advancements in healthcare infrastructure, the rise in chronic diseases, and technological innovations. The market size has been estimated to reach billions ~ USD, reflecting a steady demand for medical technologies, particularly in the areas of diagnostics, imaging, and monitoring systems. Government initiatives and an increasing number of healthcare investments are expected to drive the market’s continued expansion, with key players capitalizing on the growing healthcare needs.
Turkey remains a key player in the medical devices market in the Middle East and North Africa region due to its strategic geographic location, modern healthcare infrastructure, and increasing patient population. Major cities like Istanbul, Ankara, and Izmir have seen a rapid growth in healthcare facilities, attracting investments in medical technologies. This dominance is further supported by the country’s healthcare reforms, the expansion of hospitals, and medical tourism, which has bolstered the demand for high-quality medical devices across both public and private sectors.
Market Segmentation
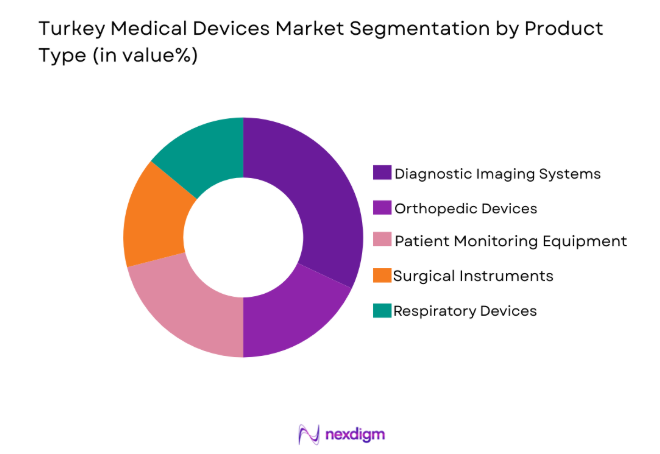
By Product Type
The Turkey medical devices market is segmented by product type into diagnostic imaging systems, orthopedic devices, patient monitoring equipment, surgical instruments, and respiratory devices. Recently, diagnostic imaging systems have seen a dominant market share due to high demand for accurate and early diagnosis, driven by increased awareness of healthcare, aging populations, and the prevalence of chronic diseases. The robust infrastructure for medical imaging, along with advances in technology such as MRI and CT scanning, has fueled growth in this segment.
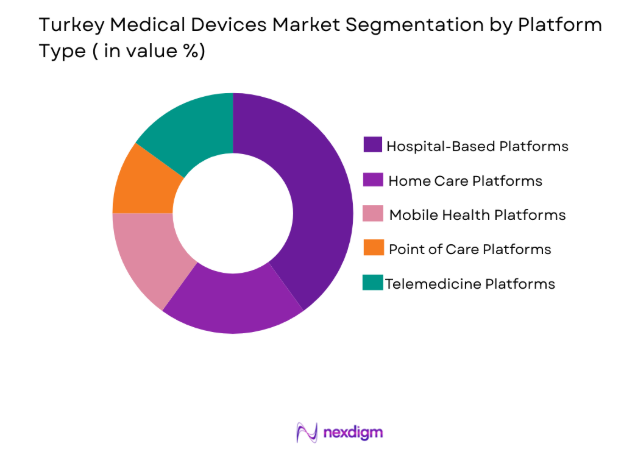
By Platform Type
The market is segmented into hospital-based platforms, home care platforms, mobile health platforms, point of care platforms, and telemedicine platforms. Hospital-based platforms have a dominant market share as healthcare facilities in Turkey increasingly adopt advanced medical devices for comprehensive patient care. The growth in public and private hospital sectors and government investments into healthcare infrastructure have fueled the adoption of integrated, hospital-based platforms that enhance the effectiveness of diagnosis and treatment.
Competitive Landscape
The competitive landscape of the Turkey medical devices market is diverse, with global and local companies striving to meet the growing demand for advanced healthcare solutions. The market has witnessed some consolidation, with larger multinational corporations strengthening their presence, while smaller companies focus on niche segments or technology innovations. Major players in the market influence product development, pricing strategies, and market expansion through their extensive research capabilities and distribution networks.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Additional Parameter |
| Siemens Healthineers | 1847 | Germany | ~ | ~ | ~ | ~ | ~ |
| Philips Healthcare | 1891 | Netherlands | ~ | ~ | ~ | ~ | ~ |
| Medtronic | 1949 | Ireland | ~ | ~ | ~ | ~ | ~ |
| GE Healthcare | 1892 | USA | ~ | ~ | ~ | ~ | ~ |
| Stryker | 1941 | USA | ~ | ~ | ~ | ~ | ~ |
Turkey Medical Devices Market Analysis
Growth Drivers
Technological Advancements
Technological advancements in medical devices have become a major growth driver in Turkey’s medical market. As healthcare providers increasingly invest in cutting-edge technologies like AI-driven diagnostic tools, robotic surgery systems, and connected healthcare devices, there is a significant improvement in patient outcomes, surgical precision, and operational efficiencies. These technologies contribute to shorter recovery times, reduced complications, and enhanced diagnostic capabilities, all of which drive market growth. The growing preference for minimally invasive procedures further supports this trend, as hospitals adopt technologies that offer enhanced accuracy and faster patient recovery. Additionally, medical devices such as wearable sensors, telemedicine platforms, and mobile health solutions are gaining traction, offering patients more convenient and accessible healthcare options. Government incentives, grants, and subsidies have further stimulated investment in these technological advancements, further propelling the market’s growth. With healthcare innovation continually progressing, the demand for these advanced technologies is expected to remain high.
Rising Demand for Healthcare Services
The rising prevalence of chronic diseases and an aging population in Turkey has significantly increased the demand for improved healthcare services and medical devices. As healthcare infrastructure is under growing pressure to meet these needs, there is a surge in investments aimed at enhancing diagnostic capabilities, improving patient care, and reducing operational inefficiencies. Chronic conditions such as diabetes, hypertension, and cardiovascular diseases require continuous monitoring, and devices like blood glucose monitors, ECG machines, and diagnostic imaging systems are in high demand. Healthcare reforms, along with increased public health awareness, are driving the adoption of medical devices across both public and private healthcare sectors. As the demand for reliable and efficient healthcare systems rises, medical technologies are becoming a vital component in managing these conditions. This increase in healthcare needs is fueling the market for medical devices, creating both opportunities and challenges for the industry as a whole.
Market Challenges
Regulatory Challenges
Despite rapid growth in Turkey’s medical devices market, regulatory challenges remain a significant barrier. The stringent regulatory framework for medical device approval can slow market entry for new products, as companies must comply with strict guidelines set by the Turkish Medicines and Medical Devices Agency. Regulatory complexity, particularly in areas such as safety, performance, and manufacturing standards, can add considerable time and cost to the approval process. Furthermore, ensuring compliance with international certifications such as CE or FDA adds another layer of complexity for foreign companies seeking market access. These regulatory challenges can significantly delay the introduction of innovative products, reduce market access, and impact overall growth potential for many companies in the Turkish medical devices sector.
High Capital Investment
A major barrier to growth in Turkey’s medical devices market is the high capital investment required to acquire advanced medical equipment. While large hospitals and healthcare centers have the financial capacity to adopt cutting-edge medical technologies, smaller healthcare facilities and startups face challenges in procuring and maintaining these devices. The substantial initial cost of medical equipment, as well as ongoing maintenance and operational costs, can deter investment, particularly in rural or less-developed regions. Smaller healthcare organizations often struggle with financial constraints, limiting their ability to offer the latest diagnostic, surgical, and patient monitoring systems. Additionally, economic instability and fluctuating currency exchange rates may impact the purchasing power of healthcare providers, further restricting growth in this market segment.
Opportunities
Growth in Medical Tourism
Turkey’s rising prominence as a medical tourism hub presents significant growth opportunities for the medical devices market. With an increasing number of international patients seeking affordable yet high-quality medical care, especially for procedures like cosmetic surgery, orthopedics, and fertility treatments, the demand for advanced medical equipment is growing. As Turkey attracts more patients from neighboring countries and Europe, healthcare facilities are increasing their investments in state-of-the-art technologies, particularly in diagnostic imaging, surgery, and patient monitoring devices. This growth in medical tourism enhances the need for the latest medical devices and technologies to meet the demand for specialized treatments. With continuous investments in healthcare infrastructure, Turkey is well-positioned to become a key player in the global medical tourism market, driving demand for high-quality medical devices.
Government Support for Healthcare Innovation
The Turkish government’s commitment to modernizing healthcare infrastructure presents a significant opportunity for the medical devices market. With an increasing focus on improving care quality and expanding access to healthcare technologies, the government is fostering innovation through investments in public healthcare initiatives and public-private partnerships. Additionally, government support for research and development in the medical devices sector, including grants, subsidies, and regulatory support, provides both domestic and international players with the opportunity to expand their product offerings. As Turkey continues to prioritize the modernization of its healthcare system, demand for advanced medical technologies, especially in diagnostics, patient monitoring, and treatment, will continue to grow, further propelling market expansion.
Future Outlook
The future outlook for Turkey’s medical devices market is promising, with continued growth expected due to technological advancements, government investments, and rising demand for healthcare services. The increasing focus on early diagnosis, personalized medicine, and telehealth will shape the future landscape, with innovations in AI, robotics, and connected health platforms. Regulatory support, along with rising healthcare spending, will create favorable conditions for market expansion. Additionally, the country’s strategic location as a medical tourism hub will continue to drive demand for high-quality medical devices.
Major Players
- Siemens Healthineers
- Philips Healthcare
- Medtronic
- GE Healthcare
- Stryker
- Zimmer Biomet
- Johnson & Johnson Medical Devices
- Abbott Laboratories
- Becton Dickinson
- Fresenius Medical Care
- Cardinal Health
- Smith & Nephew
- Boston Scientific
- Cook Medical
- Terumo Corporation
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Healthcare providers and hospitals
- Medical device distributors
- Medical equipment procurement agencies
- Private healthcare organizations
- Pharmaceutical companies
Research Methodology
Step 1: Identification of Key Variables
Identifying the primary factors influencing the medical devices market in Turkey, including technology adoption, regulatory frameworks, and healthcare demand trends.
Step 2: Market Analysis and Construction
Analyzing historical data, current market conditions, and future trends to construct a comprehensive market model for forecasting.
Step 3: Hypothesis Validation and Expert Consultation
Validating hypotheses through consultations with industry experts, medical professionals, and key stakeholders in the healthcare sector.
Step 4: Research Synthesis and Final Output
Synthesizing data from primary and secondary research to provide a final output in the form of actionable insights, market trends, and projections.
- Executive Summary
- Research Methodology
(Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Growing Elderly Population
Increase in Chronic Diseases
Technological Advancements in Medical Devices - Market Challenges
High Initial Investment Costs
Regulatory Hurdles
Market Fragmentation - Market Opportunities
Expansion in Home Healthcare Devices
Partnerships with Tech Firms for Telemedicine
Government Investments in Healthcare Infrastructure - Trends
Integration of AI in Diagnostic Tools
Rise of Wearable Health Monitoring Devices - Government Regulations
Data Protection and Privacy Regulations
Export Control and Compliance Policies
Government Funding for Medical Innovation - SWOT Analysis
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Diagnostic Imaging Systems
Orthopedic Devices
Patient Monitoring Equipment
Surgical Instruments
Respiratory Devices - By Platform Type (In Value%)
Hospital-based Platforms
Home Care Platforms
Mobile Health Platforms
Point of Care Platforms
Telemedicine Platforms - By Fitment Type (In Value%)
Standalone Systems
Integrated Systems
Modular Systems
Wearable Devices - By End User Segment (In Value%)
Hospitals
Clinics
Home Healthcare Providers
- Market Share Analysis
- Cross Comparison Parameters
(System Type, Platform Type, Procurement Channel, End User Segment, Fitment Type, Technology Adoption, Regulatory Compliance) - SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Siemens Healthineers
Philips Healthcare
Medtronic
GE Healthcare
Stryker
Zimmer Biomet
Johnson & Johnson Medical Devices
Abbott Laboratories
Becton Dickinson
Fresenius Medical Care
Cardinal Health
Smith & Nephew
Boston Scientific
Cook Medical
Terumo Corporation
- Increasing Demand from Rural Areas
- Rising Patient Preferences for Home-based Healthcare
- Surge in Telemedicine Adoption
- Growing Healthcare Accessibility through Technology
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035


