Market Overview
The UAE Autoimmune Disorder Testing Market is projected to experience significant growth, with a market size based on a recent historical assessment. This expansion is driven by increasing healthcare investments, rising awareness about autoimmune diseases, and technological advancements in diagnostic testing. The growing demand for early diagnosis and the need for precise, reliable test results further fuel this market. The market value for autoimmune disorder testing is estimated to reach USD ~ billion in 2024, as patients and healthcare providers embrace more advanced diagnostic methods.
The UAE remains a dominant player in the Middle East, driven by its advanced healthcare infrastructure, government support, and high demand for cutting-edge diagnostic solutions. Dubai and Abu Dhabi are leading regions, with well-established hospitals and diagnostic centers investing heavily in autoimmune testing. These cities’ prominence is driven by their medical tourism initiatives, strong healthcare policies, and increasing patient awareness about autoimmune diseases, further solidifying their leadership in this growing market.
Market Segmentation
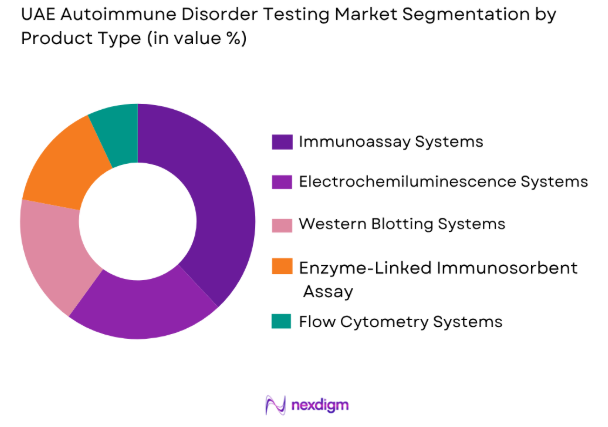
By Product Type
The UAE Autoimmune Disorder Testing Market is segmented by product type into Immunoassay Systems, Electrochemiluminescence Systems, Western Blotting Systems, Enzyme-Linked Immunosorbent Assay (ELISA), and Flow Cytometry Systems. Immunoassay Systems currently dominate the market, driven by their widespread adoption across hospitals and diagnostic centers. These systems offer high sensitivity and accuracy in detecting autoimmune disorders, making them a preferred choice among healthcare providers. Additionally, the continuous advancement of immunoassay technology, coupled with its reliability in diagnosing autoimmune diseases such as lupus, rheumatoid arthritis, and others, positions it as the market leader in the UAE.
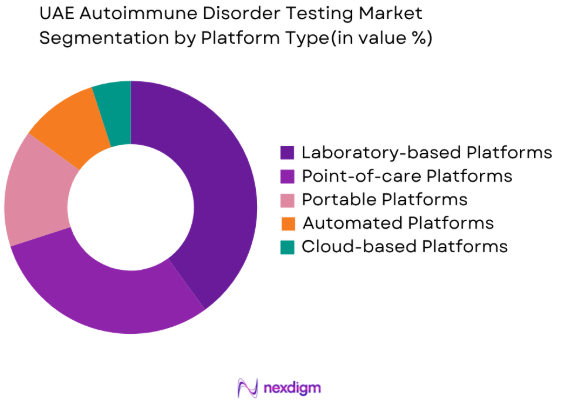
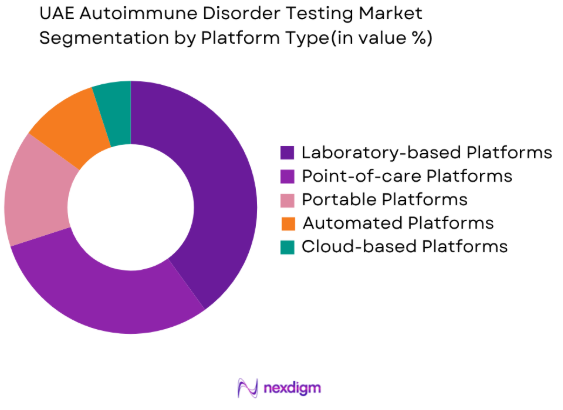
By Platform Type
The market is segmented by platform type into Laboratory-based Platforms, Point-of-care Platforms, Portable Platforms, Automated Platforms, and Cloud-based Platforms. Laboratory-based platforms currently dominate the market, favored for their high throughput and the ability to provide accurate, reproducible results for a wide range of autoimmune disorders. These systems are commonly used in large hospitals and diagnostic labs where a high volume of tests is processed. However, the shift toward point-of-care and portable platforms is increasing, driven by the demand for faster, on-site testing solutions, making them a key emerging trend in the market.
Competitive Landscape
The competitive landscape of the UAE Autoimmune Disorder Testing Market is shaped by both global and regional players, contributing to increased market consolidation. Leading companies are focusing on enhancing their product offerings through research and development, strategic partnerships, and acquisitions to stay competitive. The UAE market is characterized by the presence of major players investing in expanding healthcare infrastructure, leading to innovation in diagnostic solutions. Companies are also leveraging government support and regulations favoring healthcare technology adoption to gain a competitive edge in this growing sector.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Regulatory Compliance |
| Abbott Laboratories | 1888 | USA | ~ | ~ | ~ | ~ | ~ |
| Roche Diagnostics | 1896 | Switzerland | ~ | ~ | ~ | ~ | ~ |
| Thermo Fisher Scientific | 1956 | USA | ~ | ~ | ~ | ~ | ~ |
| Bio-Rad Laboratories | 1952 | USA | ~ | ~ | ~ | ~ | ~ |
| Danaher Corporation | 1969 | USA | ~ | ~ | ~ | ~ | ~ |
UAE Autoimmune Disorder Testing Market Analysis
Growth Drivers
Government Investment in Healthcare Infrastructure
The UAE government has significantly increased its investment in healthcare infrastructure, particularly in diagnostic technologies. This has driven the growth of autoimmune disorder testing by facilitating access to high-quality testing equipment across the country. Government initiatives such as Vision 2021 have also focused on improving healthcare facilities, thereby increasing the demand for advanced diagnostic solutions. Public-private partnerships in healthcare are encouraging more investments, especially in the areas of precision medicine and personalized diagnostics, which include autoimmune disorder testing. The adoption of state-of-the-art technologies such as immunoassays and molecular diagnostics is heavily supported by the government, ensuring broader access to advanced testing methods. This trend is expected to continue, boosting the market for autoimmune disorder testing in the UAE. Furthermore, the push for medical tourism in the UAE has prompted hospitals and diagnostic centers to adopt cutting-edge testing technologies, increasing the demand for autoimmune disorder diagnostics. The availability of funding for healthcare startups and innovations is also playing a pivotal role in accelerating market growth.
Technological Advancements in Diagnostic Systems
Technological advancements are another key growth driver for the UAE Autoimmune Disorder Testing Market. The constant development of more accurate and faster diagnostic tools has made it easier to identify autoimmune disorders early, improving patient outcomes. Innovations in immunoassay technologies, molecular diagnostics, and AI-driven testing platforms have revolutionized the healthcare sector, making it possible to diagnose diseases with higher precision and in a shorter timeframe. These advancements are particularly important for diagnosing chronic autoimmune diseases like rheumatoid arthritis, lupus, and multiple sclerosis. As healthcare providers adopt these advanced testing platforms, patients benefit from improved diagnostic accuracy and more effective treatment options. These innovations also align with the UAE’s focus on becoming a global leader in medical technologies, positioning the market for further growth. With the introduction of automated testing systems and cloud-based platforms, diagnostic procedures have become more efficient, leading to increased adoption in hospitals, research institutions, and diagnostic laboratories. This has driven demand for more advanced testing solutions, making technology a key enabler of market expansion.
Market Challenges
High Cost of Diagnostic Technologies
The high cost of advanced diagnostic technologies remains a significant challenge in the UAE Autoimmune Disorder Testing Market. The purchase, installation, and maintenance of cutting-edge testing equipment such as immunoassay systems, electrochemiluminescence platforms, and flow cytometry systems can be prohibitively expensive for smaller healthcare facilities and diagnostic labs. These costs often deter smaller entities from adopting advanced technologies, limiting access to accurate diagnostic testing for certain patient populations. Although government initiatives aim to subsidize healthcare costs, the high cost of cutting-edge diagnostic technologies continues to be a challenge, especially in less economically developed areas. Healthcare providers in these regions face budget constraints, which restricts their ability to invest in the latest diagnostic systems. The reliance on high-cost technologies also limits market growth in rural areas, where healthcare infrastructure is still developing. The slow pace of technological adoption in smaller healthcare centers has led to disparities in healthcare quality, hindering the widespread use of autoimmune disorder testing. Until affordable alternatives become available, the high costs of diagnostic equipment will continue to limit the market’s overall growth potential.
Regulatory Barriers in Test Approval
Regulatory barriers are another significant challenge in the UAE Autoimmune Disorder Testing Market. The process of obtaining approval for new diagnostic tests and devices is time-consuming, complex, and expensive. Manufacturers must navigate strict regulations to meet safety, efficacy, and quality standards before their products can be marketed and sold in the UAE. This regulatory process delays the availability of new diagnostic technologies, limiting access to the latest innovations. Moreover, compliance with international standards, such as ISO certifications and FDA approvals, is required for market entry, which further complicates the approval process. The regulatory environment is particularly challenging for smaller companies that lack the resources to meet these requirements. While the UAE has made strides in improving its healthcare regulatory environment, the complexity of regulatory approvals remains a barrier to the rapid introduction of new diagnostic technologies. This has a direct impact on the pace of market expansion, limiting the availability of innovative autoimmune disorder tests.
Opportunities
Expansion of Point-of-Care Testing Solutions
The UAE Autoimmune Disorder Testing Market presents significant opportunities for the expansion of point-of-care (POC) testing solutions. POC devices offer the advantage of delivering fast, accurate results in a variety of settings, including hospitals, clinics, and even patients’ homes. These devices have gained popularity due to their ability to provide immediate results, reducing wait times and facilitating timely treatment decisions. As demand for faster and more convenient testing solutions increases, POC devices are becoming a key area of focus for manufacturers in the UAE. Furthermore, government initiatives to promote healthcare accessibility in remote areas are driving the demand for POC solutions. The UAE’s advanced healthcare infrastructure is increasingly adopting POC devices to meet the growing demand for autoimmune disorder testing. This trend is particularly prominent in primary care settings and emergency care units, where rapid diagnostic results are essential for patient management. The continuous advancements in POC technology, including miniaturization and integration with digital platforms, are expected to further drive market growth. As more affordable and portable POC testing devices become available, healthcare providers in the UAE are likely to adopt these solutions more widely, making it a promising area for future market expansion.
Integration of Artificial Intelligence in Diagnostic Systems
Another opportunity in the UAE Autoimmune Disorder Testing Market lies in the integration of artificial intelligence (AI) in diagnostic systems. AI algorithms have the potential to significantly enhance the accuracy and efficiency of autoimmune disorder diagnostics by analyzing complex data from various testing methods, such as immunoassays and molecular diagnostics. AI can assist healthcare professionals in identifying patterns in diagnostic results, reducing human error, and enabling faster decision-making. The increasing adoption of AI-driven diagnostic tools is already transforming the way healthcare providers approach autoimmune disease management, making it an essential component of future diagnostic systems. Additionally, AI integration can help healthcare providers better manage patient data, improving the overall patient experience and treatment outcomes. With AI-powered systems, the time required to diagnose autoimmune diseases can be significantly reduced, making it possible to deliver faster treatments. Furthermore, AI has the potential to reduce costs by automating routine diagnostic processes, freeing up resources for more complex cases. As the UAE continues to invest in digital health solutions, AI-driven diagnostics are expected to play a key role in the country’s healthcare system, creating a significant opportunity for growth in the autoimmune disorder testing market.
Future Outlook
The UAE Autoimmune Disorder Testing Market is expected to witness steady growth in the coming years, driven by technological advancements, government support for healthcare infrastructure, and increasing patient demand for accurate and timely diagnostics. Key developments in diagnostic systems, including the integration of artificial intelligence, are expected to enhance the efficiency and accuracy of autoimmune disorder tests. Furthermore, regulatory support for innovation and the rise of point-of-care testing solutions will contribute to the market’s expansion. The demand for advanced autoimmune testing systems will continue to grow as the UAE strengthens its position as a medical hub in the region, attracting both local and international patients.
Major Players
- Abbott Laboratories
- Roche Diagnostics
- ThermoFisher Scientific
- Bio-Rad Laboratories
- Danaher Corporation
- Siemens Healthineers
- Becton Dickinson
- Medtronic
- BioMerieux
- Ortho Clinical Diagnostics
- Beckman Coulter
- Quidel Corporation
- F. Hoffmann-La Roche
- Hologic
- ELITechGroup
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Diagnostic laboratories
- Hospitals and healthcare providers
- Medical device manufacturers
- Pharmaceuticals and biotech companies
- Research institutions
- Healthcare IT companies
Research Methodology
Step 1: Identification of Key Variables
This step involves identifying the core variables that impact the autoimmune disorder testing market, including technology trends, regulatory factors, and market dynamics.
Step 2: Market Analysis and Construction
Data is collected and analyzed to construct a comprehensive market model that highlights the key trends, growth drivers, and challenges influencing the sector.
Step 3: Hypothesis Validation and Expert Consultation
Industry experts are consulted to validate assumptions and hypotheses, ensuring that the research aligns with real-world market conditions.
Step 4: Research Synthesis and Final Output
The findings are synthesized into a final report that provides actionable insights, market forecasts, and strategic recommendations for stakeholders.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Technological Advancements in Healthcare Infrastructure
Government Investment in Healthcare Infrastructure - Market Challenges
High Cost of Diagnostic Equipment
Regulatory Barriers in Test Approval - Market Opportunities
Expansion of Point-of-care Testing Solutions
Integration of AI in Diagnostic Systems - Trends
Shift toward multiplex and high-throughput testing technologies
Growing preference for point-of-care autoimmune diagnostics - Government Regulations
- SWOT Analysis
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By Product Type (In Value%)
Immunoassay Systems
Electrochemiluminescence Systems
Western Blotting Systems
Enzyme-Linked Immunosorbent Assay (ELISA)
Flow Cytometry Systems - By Platform Type (In Value%)
Laboratory-based Platforms
Point-of-care Platforms
Portable Platforms
Automated Platforms
Cloud-based Platforms - By EndUser Segment (In Value%)
Hospitals
Diagnostic Laboratories
Research Institutions
Outpatient Clinics
Physician Offices - By Procurement Channel (In Value%)
Direct Procurement
Distributor Networks
Online Bidding Platforms
Private Sector Procurement
Government Tenders
- Market Share Analysis
- Cross Comparison Parameters(System Type, Platform Type, Procurement Channel, End User Segment, Fitment Type, Test Accuracy, Turnaround Time, Cost Efficiency, Technology Integration, Product Portfolio, Regulatory Compliance, Market Presence, Distribution Network, Automation Level, After-sales Support)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Abbott Laboratories
Roche Diagnostics
Thermo Fisher Scientific
Siemens Healthineers
Bio-Rad Laboratories
Danaher Corporation
Becton Dickinson
Medtronic
BioMerieux
Ortho Clinical Diagnostics
Beckman Coulter
Quidel Corporation
F. Hoffmann-La Roche
Hologic
ELITech Group
- Hospitals Increasing Adoption of Automation in Testing
- Diagnostic Labs Expanding Testing Capabilities
- Research Institutions Investing in Advanced Testing Solutions
- Outpatient Clinics Offering More Diagnostic Services
- By Market Value,2026-2035
- By Installed Units,2026-2035
- By Average System Price, 2026-2035
- By System Complexity Tier, 2026-2035