Market Overview
The USA biosimilar insulin market is experiencing significant growth, driven by factors such as rising diabetic prevalence and the increasing adoption of cost-effective biosimilars. As of the most recent historical assessment, the market is valued at USD ~ billion, with key drivers including regulatory support for biosimilar approval and the push for affordable diabetes care solutions. These factors are expected to drive market expansion as healthcare providers increasingly shift toward biosimilar options to lower costs while maintaining treatment effectiveness for patients with diabetes.
The USA dominates the biosimilar insulin market, supported by a robust healthcare infrastructure and government initiatives focused on reducing healthcare costs. Major cities like New York, Los Angeles, and Chicago contribute significantly to the market’s growth, driven by high demand for affordable diabetes treatments and increasing healthcare access. The country’s commitment to expanding biosimilar adoption, along with widespread access to advanced healthcare facilities, positions it as a leader in the global biosimilar insulin market.
Market Segmentation
By Product Type
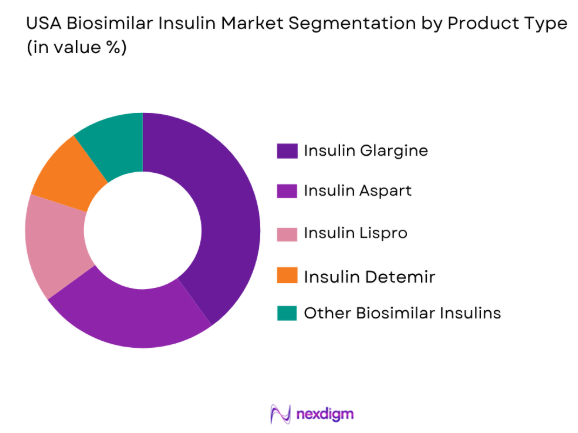
The USA biosimilar insulin market is segmented by product type into insulin glargine, insulin aspart, insulin lispro, insulin detemir, and other biosimilar insulin products. Currently, insulin glargine holds a dominant market share due to its extensive use in diabetes management, driven by its high efficacy and cost-effectiveness compared to branded insulins. The widespread acceptance of insulin glargine among healthcare providers, its long market presence, and its stable demand from both public and private healthcare sectors have contributed to its leadership in this market segment.
By Delivery Device
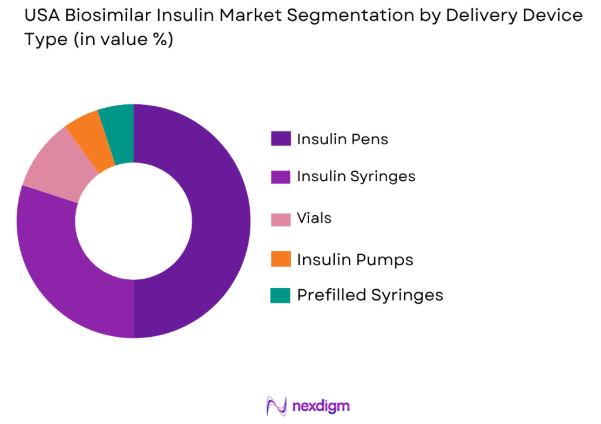
The biosimilar insulin market in the USA is also segmented by delivery device, including insulin pens, insulin syringes, vials, insulin pumps, and prefilled syringes. Insulin pens dominate this segment, driven by their convenience, ease of use, and precision in dosage delivery. Their adoption has been increasing among both healthcare providers and patients due to their user-friendly design, compact size, and reduced risk of dosage errors. These advantages make insulin pens the preferred delivery device in the USA market.
Competitive Landscape
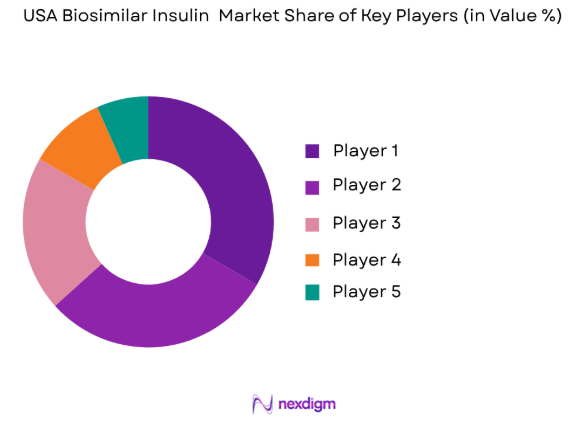
The USA biosimilar insulin market is highly competitive, characterized by a mix of large multinational pharmaceutical companies and emerging biosimilar producers. Market consolidation is evident as major players acquire smaller biosimilar firms to expand their portfolios and strengthen their market presence. Key players focus on advancing their biosimilar offerings while competing on price, product efficacy, and market penetration. The competitive landscape also highlights the regulatory challenges in the approval process, which affect market entry and innovation.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD Billion) | Additional Parameter |
| Sanofi | 2004 | Paris, France | ~ | ~ | ~ | ~ | ~ |
| Novo Nordisk | 1923 | Bagsværd, Denmark | ~ | ~ | ~ | ~ | ~ |
| Boehringer Ingelheim | 1885 | Ingelheim, Germany | ~ | ~ | ~ | ~ | ~ |
| Mylan | 1961 | Canonsburg, USA | ~ | ~ | ~ | ~ | ~ |
| Sandoz | 2003 | Holzkirchen, Germany | ~ | ~ | ~ | ~ | ~ |
USA Biosimilar Insulin Market Analysis
Growth Drivers
Increased Diabetic Prevalence
The increasing prevalence of diabetes in the USA is a primary driver of the biosimilar insulin market. With over 30 million people affected by diabetes, the demand for effective and affordable insulin treatments has surged. Biosimilars, being more cost-effective compared to branded insulins, are increasingly viewed as a viable option by both healthcare providers and patients. Government efforts to curb healthcare spending have fueled the adoption of biosimilars, making them an attractive choice for healthcare systems across the country. As the diabetic population continues to grow, biosimilars are expected to play an increasingly important role in managing the disease and improving patient access to care. With new innovations in insulin delivery systems and regulatory support, the market for biosimilar insulin will continue to expand rapidly. This trend will further accelerate as more patients shift to affordable insulin alternatives.
Government Support for Biosimilars
Another key growth driver for the USA biosimilar insulin market is the increasing government support for biosimilar adoption. Government initiatives like the Biologics Price Competition and Innovation Act (BPCIA) have streamlined the approval process for biosimilars, making them more accessible to patients. These policies are intended to foster competition and reduce the overall cost of biologic drugs, including insulin. The USA government has also made substantial investments in healthcare programs aimed at improving access to insulin, particularly in underserved areas. The combination of favorable regulatory frameworks and government-backed initiatives will continue to boost the adoption of biosimilar insulin, ensuring a steady growth trajectory for the market.
Market Challenges
Regulatory Barriers
Despite the favorable regulatory changes, significant barriers remain in the approval and market entry of biosimilar insulin products. The FDA approval process for biosimilars, though streamlined, is still time-consuming and complex. The need for extensive clinical trials and data to prove the biosimilarity of products to reference insulins can delay market introduction and increase costs. Furthermore, the regulatory landscape for biosimilars is continually evolving, which creates uncertainties for manufacturers looking to enter the market. As a result, new entrants face considerable challenges in navigating the regulatory environment, which impacts the speed of innovation and market competition in the biosimilar insulin space.
Price Competition from Branded Insulin
Another challenge faced by the USA biosimilar insulin market is the ongoing price competition from established branded insulin products. While biosimilars offer a more affordable alternative, the market dominance of well-established brands like Novo Nordisk and Sanofi, which have strong brand loyalty and extensive distribution networks, makes it difficult for biosimilars to gain significant market share. These brands continue to offer competitive pricing and have established relationships with healthcare providers, making it challenging for newer players to displace them in the marketplace. The persistence of high-priced branded insulins in the market limits the overall adoption of biosimilars, preventing the full potential of cost savings from being realized.
Opportunities
Expansion in Rural and Underserved Areas
A significant opportunity for the biosimilar insulin market lies in the expansion of availability in rural and underserved areas. Many patients in these regions lack access to affordable insulin treatments, leading to suboptimal diabetes management. Biosimilars present a cost-effective solution to this problem, and expanding distribution channels to these areas could significantly increase market penetration. This expansion can be supported by government programs and partnerships with healthcare providers, ensuring that more patients gain access to affordable insulin. Moreover, the growing number of healthcare initiatives focused on expanding coverage in rural America presents a valuable growth avenue for biosimilar insulin producers.
Technological Advancements in Delivery Devices
Another opportunity for growth in the USA biosimilar insulin market is the integration of technological advancements in insulin delivery devices. The rise of smart insulin pens, which track usage and provide reminders for patients, has the potential to increase the efficiency and appeal of biosimilar insulin. These devices can improve patient compliance and provide valuable data for healthcare providers to better manage diabetes care. As the market for diabetes management tools continues to evolve, the development of more advanced insulin delivery technologies presents a substantial opportunity for biosimilar insulin manufacturers to differentiate their products and enhance patient outcomes.
Future Outlook
The USA biosimilar insulin market is expected to experience steady growth over the next five years, driven by increasing diabetic prevalence and government initiatives to support biosimilar adoption. Technological advancements in insulin delivery systems, coupled with stronger regulatory frameworks, will likely drive further market expansion. As the demand for affordable and effective diabetes treatments rises, biosimilar insulin will continue to play a key role in reshaping the landscape of diabetes care in the USA.
Major Players
- Sanofi
- Novo Nordisk
- Boehringer Ingelheim
- Mylan
- Sandoz
- Eli Lilly
- Teva Pharmaceuticals
- Merck & Co.
- AbbVie
- Amgen
- Pfizer
- Regeneron Pharmaceuticals
- Ferring Pharmaceuticals
- GSK
- Celltrion
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Healthcare providers
- Pharmaceutical manufacturers
- Insurance companies
- Diabetes care organizations
- Retail pharmacies
- Biotech firms
Research Methodology
Step 1: Identification of Key Variables
This step involves identifying the critical variables that drive the market, including patient demographics, insulin adoption rates, and regulatory frameworks.
Step 2: Market Analysis and Construction
In this phase, we conduct a comprehensive market analysis using primary and secondary research to assess market size, growth drivers, challenges, and opportunities.
Step 3: Hypothesis Validation and Expert Consultation
We validate our findings by consulting with industry experts and key stakeholders in the biosimilar insulin and diabetes care sectors to refine assumptions and ensure accuracy.
Step 4: Research Synthesis and Final Output
Finally, we synthesize the data collected and present the findings in a structured report format that highlights key insights, market trends, and strategic recommendations.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Increase in Diabetes Prevalence
Government support of Biosimilars - Market Challenges
Regulatory Barriers
Price Competition from Branded Insulin - Market Opportunities
Expansion in Rural and Underserved Areas
Price Competition from Branded Insulin - Trends
Increased Adoption of Insulin Pens
Integration of Digital Health Features in Insulin Devices
Focus on Sustainability and Eco-friendly Insulin Devices - Government Regulations
- SWOT Analysis of Key Competitors
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By Product Type (In Value%)
Insulin Glargine
Insulin Lispro
Insulin Aspart
Insulin Detemir
Other Biosimilar Insulin - By Device Type (In Value%)
Injection Pens
Syringes
Vials
Prefilled Syringes
Insulin Pumps - By End User Segment (In Value%)
Hospitals
Clinics
Home Care
Retail Pharmacies
Diabetes Care Centers
- Market Share Analysis
- Cross Comparison Parameters (Product Type, Insulin Analog Type, Delivery Device Type, Distribution Channel, End User Segment, Dosage Strength, Pricing Competitiveness, Regulatory Approvals, Manufacturing Capacity, Product Availability, Brand Positioning, Market Presence, Distribution Network, Patient Support Programs, Partnerships & Collaborations)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Sanofi
Novo Nordisk
Boehringer Ingelheim
Mylan
Sandoz
Baxter International
Lilly
Bristol-Myers Squibb
Fresenius Kabi
Teva Pharmaceuticals
Celltrion
Korean Biologics
Hikma Pharmaceuticals
Samsung Biologics
Amgen
- Hospitals Increasing Biosimilar Insulin Usage
- Pharmacy Demand for Biosimilar Insulin
- Shift Towards Home Care and Self-Monitoring
- Regional Disparities in Access to Biosimilar Insulin
- By Market Value,2026-2035
- By Installed Units,2026-2035
- By Average System Price, 2026-2035
- By System Complexity Tier, 2026-203