Market Overview
The USA biosimilar monoclonal antibodies market is projected to reach USD ~ billion based on a recent historical assessment, driven by increasing demand for affordable therapeutic alternatives, particularly in oncology and immunology. The expansion of the market is further fueled by the ongoing development of new biosimilar therapies, advancements in biologics manufacturing, and favorable regulatory frameworks. This surge is accompanied by rising healthcare expenditures, pushing the need for cost-effective treatment options. Market dynamics are also shaped by the growing preference for biologic treatments and improved patient access to biosimilars.
The USA remains a dominant player in the biosimilar monoclonal antibodies market, with significant contributions from regions like California, New Jersey, and Massachusetts. These areas are home to leading biopharma companies that focus on research and development, regulatory compliance, and distribution. Additionally, the robust healthcare infrastructure and growing adoption of biosimilars across hospitals, clinics, and specialty pharmacies have driven the USA’s market dominance. A favorable regulatory environment and government policies, including the Biologics Price Competition and Innovation Act, continue to propel market expansion.
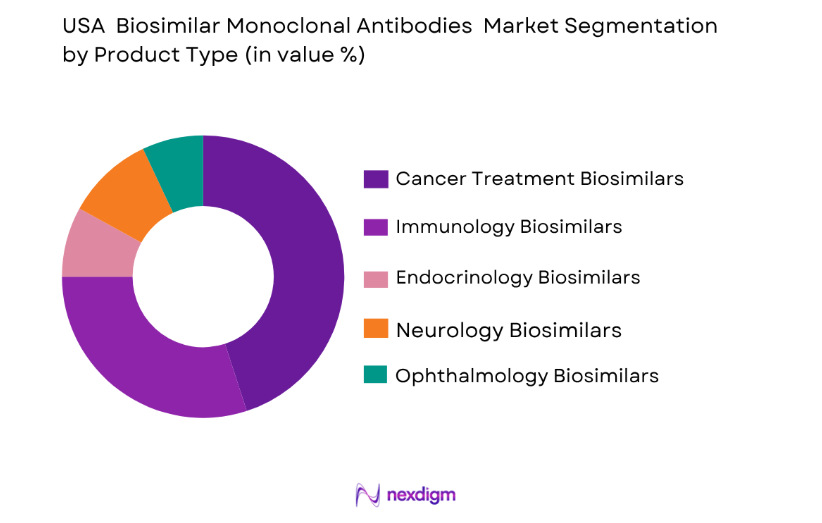
Market Segmentation
By Product Type:
USA biosimilar monoclonal antibodies market is segmented by product type into various sub-segments. Recently, cancer treatment biosimilars have captured the largest market share, driven by the high demand for oncology therapies and the success of biosimilars in treating various cancers. Factors such as the growing prevalence of cancer, increased patient access to affordable alternatives, and supportive regulatory approvals contribute to the dominance of cancer treatment biosimilars in the market. With the patent expiration of key monoclonal antibodies, biosimilars in oncology are expected to lead the market for the foreseeable future.
By Distribution Channel:
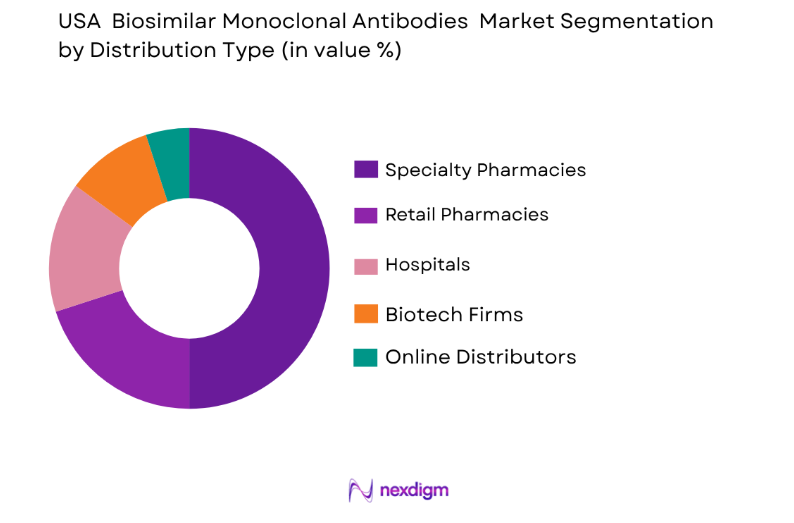
USA biosimilar monoclonal antibodies market is segmented by distribution channel into several sub-segments. Recently, specialty pharmacies have seen the largest market share due to their critical role in delivering complex biosimilar treatments to patients, especially for conditions like cancer and rheumatoid arthritis. Specialty pharmacies are strategically located and equipped to manage the distribution of biosimilars with the necessary support services for both patients and healthcare providers, making them a key driver of market growth. Additionally, the convenience of home delivery and personalized care in these pharmacies is accelerating the preference for this distribution channel.
Competitive Landscape
The USA biosimilar monoclonal antibodies market is highly competitive, with major biopharma companies driving innovation and market penetration. The consolidation of the industry has been evident, with a few large players dominating the market, and smaller firms focusing on niche segments or specific therapeutic areas. The dominance of established companies like Amgen, Sandoz, and Pfizer is reinforced by their extensive experience in monoclonal antibody development, regulatory approvals, and manufacturing capabilities. New entrants are leveraging advancements in biologics manufacturing to capture market share, resulting in increased competition. Ongoing mergers and acquisitions among key players further strengthen market consolidation and expand their portfolio of biosimilar products.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Regulatory Approvals |
| Amgen | 1980 | Thousand Oaks, CA | ~ | ~ | ~ | ~ | ~ |
| Sandoz | 2006 | Holzkirchen, Germany | ~ | ~ | ~ | ~ | ~ |
| Pfizer | 1849 | New York, NY | ~ | ~ | ~ | ~ | ~ |
| Celltrion | 2002 | Incheon, South Korea | ~ | ~ | ~ | ~ | ~ |
| Mylan | 1961 | Canonsburg, PA | ~ | ~ | ~ | ~ | ~ |
USA Biosimilar Monoclonal Market Analysis
Growth Drivers
Rising Prevalence of Chronic Diseases:
The growing prevalence of chronic diseases, especially cancer, autoimmune disorders, and other long-term conditions, is a significant growth driver for the USA biosimilar monoclonal antibodies market. With the increasing burden of diseases such as cancer and rheumatoid arthritis, the demand for affordable and effective treatment options continues to surge. Biosimilars offer a cost-effective alternative to reference biologics, making them an attractive option for patients and healthcare providers alike. As healthcare costs rise, insurance companies and payers are increasingly encouraging the use of biosimilars to reduce the financial strain of biologic treatments. The approval of new biosimilars and their integration into clinical practice further fuels this demand. The shift towards biosimilars helps address the financial challenges in healthcare systems and ensures better access to life-saving treatments for patients, contributing to market growth. Additionally, patients benefit from more treatment options, leading to higher adoption rates. Furthermore, governments’ support for biosimilars in healthcare policy is accelerating the market’s expansion. Increased competition within the biosimilars space is anticipated to drive innovation and foster further growth in the coming years, as companies seek to capture market share in this rapidly evolving sector.
Regulatory Advancements:
Another critical growth driver for the USA biosimilar monoclonal antibodies market is the favorable regulatory environment that supports the development and approval of biosimilars. In the United States, the Biologics Price Competition and Innovation Act (BPCIA) has provided a structured pathway for biosimilar approvals, which has fostered greater confidence among biopharma companies to invest in the development of biosimilars. The FDA’s increasing acceptance of biosimilars and its active role in facilitating their introduction into the market have provided a much-needed boost to the industry. Moreover, the growing recognition of biosimilars’ therapeutic equivalence to their reference biologics by the FDA, coupled with streamlined approval processes, has enhanced market access. This regulatory framework allows for faster market entry, reducing the time required to bring biosimilars to market while maintaining high standards of safety and efficacy. Regulatory incentives, such as exclusivity periods and fast-track approval processes, further attract investment in biosimilar research and development. The evolving regulatory landscape is expected to continue supporting market expansion and innovation, ensuring that patients have access to affordable, high-quality biosimilar treatments.
Market Challenges
High Production Costs:
One of the significant challenges in the USA biosimilar monoclonal antibodies market is the high production costs associated with developing and manufacturing biosimilars. The production process for monoclonal antibodies is complex and requires advanced biotechnological infrastructure, skilled labor, and high-quality raw materials, all of which contribute to the overall cost of biosimilars. These high production costs can limit the profitability of biosimilar manufacturers and slow down the pace at which new products are introduced into the market. While biosimilars offer cost savings compared to reference biologics, their production still involves substantial investment in research and development, regulatory compliance, and manufacturing processes. Additionally, the need to maintain stringent quality control standards and ensure the biosimilarity of the product to the reference biologic increases the cost burden on manufacturers. As a result, biosimilar manufacturers face pressure to balance production costs with market pricing, which can impact their competitive position, particularly against well-established biologic brands. The challenge of managing production costs while ensuring product quality and affordability remains a critical barrier to market growth.
Intellectual Property and Patent Issues:
Intellectual property (IP) and patent disputes represent another significant challenge to the USA biosimilar monoclonal antibodies market. Many reference biologics are protected by patents, which can make it difficult for biosimilar manufacturers to enter the market without facing costly litigation or delays. Patent exclusivity periods for biologics are often lengthy, and even after patents expire, additional patents related to specific formulations, delivery systems, or manufacturing processes may remain in effect. This creates barriers for biosimilar companies looking to develop and commercialize products in the same therapeutic areas. Patent litigation is a common occurrence in the biosimilar industry, with manufacturers of reference biologics often challenging biosimilar companies on the grounds of patent infringement. These legal challenges can delay the market entry of biosimilars, increasing the time it takes for patients to benefit from more affordable treatment options. The evolving landscape of intellectual property laws, including the protection of trade secrets and biosimilar manufacturing processes, further complicates the market. Manufacturers must navigate these complex patent issues to gain regulatory approval and commercialize their products, which can significantly increase costs and slow down market growth.
Opportunities
Increasing Adoption of Personalized Medicine:
The increasing adoption of personalized medicine presents a significant opportunity for the USA biosimilar monoclonal antibodies market. As the healthcare industry moves towards more individualized treatment plans, there is a growing need for biologic therapies that can be tailored to specific patient populations. Biosimilars, being highly similar to their reference biologics, can be incorporated into personalized treatment regimens, offering cost-effective alternatives for patients with specific medical conditions, including cancer, autoimmune diseases, and neurological disorders. The use of biosimilars in personalized medicine is particularly appealing as it allows for the customization of therapy based on genetic, environmental, and lifestyle factors, ensuring more effective treatment outcomes. Personalized medicine is gaining traction across both clinical and research settings, and biosimilars are poised to play a crucial role in this transformation by providing affordable and accessible treatment options. With the growing emphasis on precision healthcare, the demand for biosimilars as part of personalized treatment plans is expected to rise, offering substantial growth opportunities in the coming years.
Expansion into Emerging Markets:
Another promising opportunity for the USA biosimilar monoclonal antibodies market is the expansion into emerging markets. As healthcare systems in developing countries evolve, there is increasing demand for cost-effective alternatives to expensive reference biologics. Biosimilars are seen as a viable solution to address the high treatment costs associated with biologics, especially in low- and middle-income countries. These markets are characterized by a large patient population suffering from chronic diseases, such as cancer, diabetes, and rheumatoid arthritis, where biosimilars can provide affordable treatment options. Additionally, the regulatory environment in these countries is becoming more conducive to the approval of biosimilars, with many governments introducing policies to support the adoption of biosimilars as a way to control healthcare expenditures. As healthcare infrastructure improves and access to biosimilars increases, there is significant potential for biosimilar manufacturers to capture market share in these regions. The expansion of biosimilars into emerging markets offers a tremendous opportunity for growth, both for companies looking to diversify their product offerings and for patients seeking more affordable treatment options.
Future Outlook
The future outlook for the USA biosimilar monoclonal antibodies market is promising, with sustained growth expected over the next five years. This growth will be driven by continued advancements in biologic therapies, an expanding pipeline of biosimilars, and increasing adoption across both developed and emerging markets. Technological innovations in biologics manufacturing and enhanced regulatory support are also anticipated to fuel market expansion. As the market matures, we expect further competition, which will lead to price reductions and improved patient access to these life-saving therapies. The regulatory landscape is expected to evolve, with further improvements in the approval process for biosimilars, creating a favorable environment for market growth. Additionally, the increasing prevalence of chronic diseases, including cancer, is expected to sustain the demand for monoclonal antibodies, with biosimilars playing an essential role in improving healthcare affordability.
Major Players
- Amgen
- Sandoz
- Pfizer
- Celltrion
- Mylan
- Biocon
- Novartis
- Merck
- AbbVie
- Teva Pharmaceuticals
- Roche
- GSK
- Eli Lilly
- Boehringer Ingelheim
- Regeneron Pharmaceuticals
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Biotech companies
- Pharmaceutical companies
- Healthcare providers
- Hospitals
- Pharmaceutical distributors
- Private equity firms
Research Methodology
Step 1: Identification of Key Variables
Identifying the key variables involves understanding market trends, growth drivers, challenges, and opportunities within the biosimilar monoclonal antibodies sector. These variables form the foundation for further analysis.
Step 2: Market Analysis and Construction
Market analysis involves evaluating historical data, reviewing regulatory guidelines, and analyzing competitor performance to build an accurate market model for biosimilars.
Step 3: Hypothesis Validation and Expert Consultation
This step involves validating market hypotheses through expert consultation with industry professionals, academics, and analysts to ensure the accuracy of the data.
Step 4: Research Synthesis and Final Output
Synthesis of research findings leads to the creation of the final report, including detailed market analysis, competitor insights, and future outlook, ensuring comprehensive coverage of the biosimilars market.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Growth Drivers
Rising demand for cost-effective therapies
Advancements in monoclonal antibody production technologies
Growing prevalence of chronic diseases and cancer - Market Challenges
High manufacturing costs
Regulatory hurdles and approval processes
Intellectual property challenges - Market Opportunities
Expansion of biosimilars into emerging markets
Strategic collaborations between biopharma companies
Technological advancements in antibody engineering - Trends
Increasing adoption of biosimilars in oncology
Rising healthcare awareness and affordability
Emerging focus on personalized medicine - Government Regulations
- SWOT Analysis of Key Competitors
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Cancer Treatment Biosimilars
Immunology Biosimilars
Neurology Biosimilars
Endocrinology Biosimilars
Ophthalmology Biosimilars - By Platform Type (In Value%)
Biologic Drug Development Platforms
Cell Culture Platforms
Expression System Platforms
Antibody Engineering Platforms
Biosimilar Manufacturing Platforms - By Fitment Type (In Value%)
Intravenous Injection
Subcutaneous Injection
Intramuscular Injection
Oral Administration
Topical Application - By EndUser Segment (In Value%)
Hospitals
Clinics
Specialty Pharmacies
Retail Pharmacies
Biotech Firms
- Market Share Analysis
- CrossComparison Parameters (System Type, Platform Type, Procurement Channel, EndUser Segment, Fitment Type)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Porter’s Five Forces
- Key Players
Amgen
Sandoz
Celltrion Healthcare
Mylan
Pfizer
Samsung Bioepis
Eli Lilly
Biocon
Novartis
Boehringer Ingelheim
Merck
Roche
GSK
AbbVie
Teva Pharmaceuticals
- Hospitals shifting towards cost-effective biosimilars
- Biotech firms investing in biosimilar development
- Specialty pharmacies driving distribution
- Retail pharmacies expanding biosimilar portfolios
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035