Market Overview
The USA biosimilar recombinant proteins market is valued in the billions ~ USD, with a strong growth trajectory driven by the increasing demand for affordable biologic treatments, especially in oncology and autoimmune disorders. Rising healthcare costs and the pressure to reduce expenses for healthcare providers are fueling the growth of biosimilars, as they offer similar efficacy to their reference biologics at a fraction of the price. Based on a recent historical assessment, the USA biosimilar recombinant proteins market is projected to experience significant growth, driven by the ongoing advancements in recombinant technology and government support for the biosimilars sector.
Dominant cities in the USA, such as New York, San Francisco, and Chicago, have seen rapid adoption of biosimilars, largely due to their established healthcare infrastructure and research institutions. These regions host a critical mass of healthcare facilities and pharmaceutical companies that push forward the adoption of biosimilars. State-level initiatives and healthcare reforms, including the Medicare Part B reimbursement for biosimilars, have contributed to market dominance in these areas. Additionally, factors such as the expansion of clinical trial activities and strategic partnerships with pharmaceutical giants in these cities have boosted market demand.
Market Segmentation
By Product Type
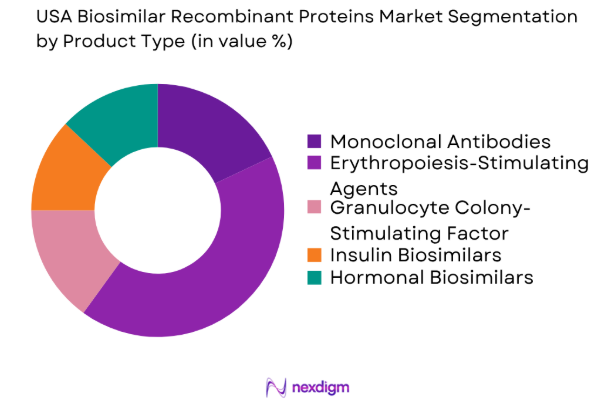
The USA biosimilar recombinant proteins market is segmented by product type into monoclonal antibodies, erythropoiesis-stimulating agents, granulocyte colony-stimulating factors, insulin biosimilars, and hormonal biosimilars. Recently, monoclonal antibodies have dominated the market share due to their broad application in treating cancers and autoimmune diseases. The robust pipeline of monoclonal antibody biosimilars and the high demand for cancer therapies have further cemented their dominant position in the market. Additionally, significant investments from biopharmaceutical companies have contributed to the accelerated development of monoclonal antibody biosimilars.
By Platform Type
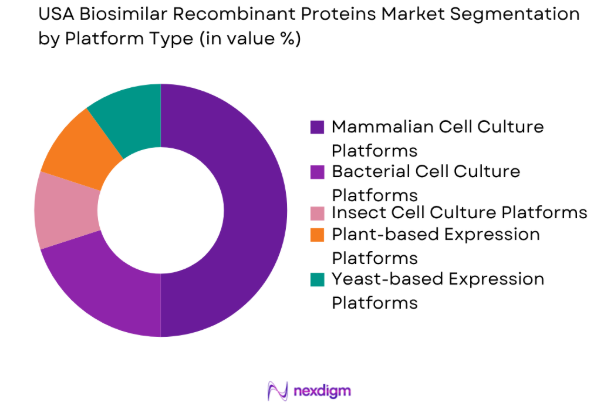
The USA biosimilar recombinant proteins market is segmented by platform type into mammalian cell culture platforms, bacterial cell culture platforms, insect cell culture platforms, plant-based expression platforms, and yeast-based expression platforms. Mammalian cell culture platforms hold the largest market share, driven by their ability to produce high-quality and complex recombinant proteins required for biosimilar production. The increased efficiency and scalability of mammalian cell culture systems, coupled with the growing demand for high-quality biosimilars, make this platform the dominant technology in the market.
Competitive Landscape
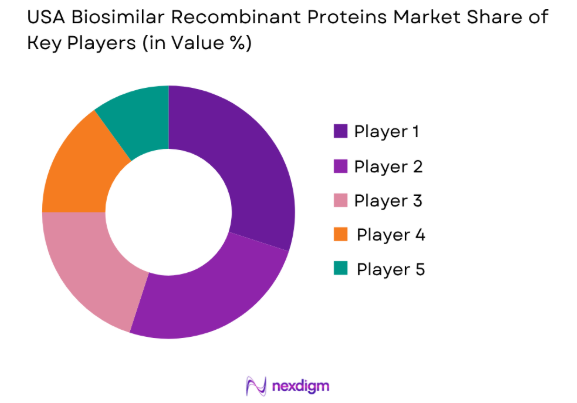
The competitive landscape of the USA biosimilar recombinant proteins market is highly dynamic, with several large pharmaceutical companies leading the market. As competition intensifies, consolidation is occurring through mergers and acquisitions, with the leading companies striving to expand their product portfolios and enhance R&D capabilities. The dominance of companies such as Amgen, Sandoz, and Pfizer shapes the market, ensuring high standards of production and technology. These players continue to leverage their expertise in biologics to develop cost-effective alternatives to reference biologics, further boosting the biosimilar recombinant proteins market.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD) | Additional Market Parameter |
| Amgen Inc. | 1980 | Thousand Oaks, CA | ~ | ~ | ~ | ~ | ~ |
| Sandoz International GmbH | 2003 | Holzkirchen, Germany | ~ | ~ | ~ | ~ | ~ |
| Pfizer Inc. | 1849 | New York, NY | ~ | ~ | ~ | ~ | ~ |
| Biocon Limited | 1978 | Bangalore, India | ~ | ~ | ~ | ~ | ~ |
| Celltrion Healthcare Co. | 2002 | Incheon, South Korea | ~ | ~ | ~ | ~ | ~ |
USA Biosimilar Recombinant Proteins Market Analysis
Growth Drivers
Increase in Chronic Diseases and Aging Population
The rising prevalence of chronic diseases such as diabetes, cancer, and autoimmune disorders, particularly among the aging population, is a significant growth driver for the biosimilar recombinant proteins market. These diseases often require long-term biologic treatments, fueling the shift towards more cost-effective biosimilars. As healthcare systems aim to manage rising costs, biosimilars offer an affordable alternative while maintaining therapeutic efficacy. The increasing adoption of biosimilars for managing chronic conditions is being driven by their ability to lower healthcare expenditures without compromising on quality. With a growing aging population and the rising burden of chronic diseases, the demand for affordable biologic therapies, including biosimilars, is expected to continue increasing, further expanding the market.
Rising Healthcare Cost Pressures
Healthcare systems globally are facing immense pressure to reduce costs, driving the demand for more cost-effective treatment options. Biosimilars address this challenge by providing the same therapeutic efficacy as reference biologics but at a significantly lower price. This cost advantage makes them an attractive alternative, especially as healthcare expenditures continue to rise. Governments and health insurers are increasingly incentivizing the use of biosimilars to ensure that high-quality care remains accessible while reducing overall healthcare spending. The growing focus on cost containment in healthcare is one of the key factors fueling the expansion of the biosimilar recombinant proteins market, benefiting both public and private sectors by offering substantial savings without compromising patient care.
Market Challenges
Regulatory Hurdles for Market Approval
Biosimilars encounter strict regulatory challenges in the USA, with the FDA demanding extensive clinical trial data to verify their safety, efficacy, and quality. These regulatory requirements significantly increase the time and cost involved in obtaining approval, presenting a substantial barrier for new market entrants. The complexity of the approval process can delay the availability of biosimilars, slowing down their market entry. Despite their cost-effectiveness and potential to reduce healthcare expenses, these regulatory hurdles complicate the path to commercialization. As a result, the high cost and lengthy approval process create challenges for expanding the biosimilar recombinant proteins market, limiting the speed at which new products can reach the market and increasing the financial burden on manufacturers.
High R&D Costs for Biosimilar Development
Developing biosimilars requires significant investment in research and development, especially for complex biologics. While biosimilars offer a more affordable alternative to reference biologics, their development costs remain high due to expensive manufacturing processes and the need for extensive clinical trials to ensure safety and efficacy. Regulatory compliance further adds to these costs, creating a considerable financial burden for companies entering the market. The high initial investment required for R&D and manufacturing makes it challenging for many companies to compete, resulting in limited market participation. This financial barrier restricts the growth of the biosimilar recombinant proteins market and slows down the introduction of new, cost-effective alternatives to traditional biologics.
Opportunities
Emerging Markets for Biosimilars
The demand for biosimilars is increasing rapidly in emerging markets, where access to costly biologics is limited. Regions such as Asia-Pacific, Latin America, and Africa offer significant opportunities for the biosimilars market, as these areas face growing healthcare burdens while lacking access to expensive treatments. Governments in these regions are implementing policies to promote biosimilar adoption, creating a supportive environment for market expansion. With favorable regulatory frameworks and increasing healthcare needs, these markets present substantial growth prospects for biosimilar manufacturers. The availability of more affordable biosimilars in these regions is expected to address healthcare access challenges and drive the market forward, making biosimilars an attractive alternative in emerging economies.
Increased Focus on Biosimilar Market Access
As demand for biosimilars grows, there is a heightened focus on improving market access, particularly in the USA. Healthcare systems are increasingly adopting cost-effective treatments, which is expected to drive the expansion of the biosimilars market. This trend is fueled by the need for supportive reimbursement policies, insurance coverage, and government incentives that enable biosimilars to compete with reference biologics. Manufacturers who can secure market access through strategic pricing, partnerships, and regulatory approvals will be well-positioned to capitalize on this opportunity. With favorable policies and increasing healthcare cost pressures, biosimilars are set to play a larger role in the healthcare market, offering affordable alternatives to expensive biologic therapies.
Future Outlook
The future outlook for the USA biosimilar recombinant proteins market is positive, with continued growth expected over the next five years. Technological advancements in recombinant protein production and regulatory support for biosimilars are likely to drive significant growth. Market access improvements, coupled with the increasing demand for affordable biologics, will be key drivers of this expansion. Additionally, the rise of chronic diseases and aging populations will continue to fuel the demand for biosimilars, positioning them as a key component in future healthcare strategies.
Major Players
- Amgen Inc.
- Sandoz International GmbH
- Pfizer Inc.
- Biocon Limited
- Celltrion Healthcare Co.
- SamsungBioepis
- Mylan N.V.
- Teva Pharmaceuticals
- Merck & Co.
- Novartis AG
- Roche Holding AG
- AbbVie Inc.
- Bristol-Myers Squibb
- Eli Lilly and Company
- GSK
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Biopharmaceutical companies
- Healthcare providers
- Pharmaceutical distributors
- Research institutions
- Contract manufacturing organizations
- Healthcare insurance companies
Research Methodology
Step 1: Identification of Key Variables
The initial step involves identifying the key variables affecting the biosimilar recombinant proteins market, including product types, platform technologies, regulatory requirements, and market trends.
Step 2: Market Analysis and Construction
A comprehensive analysis of the current market landscape, including key drivers, challenges, and opportunities, is conducted to construct a detailed market model for biosimilars.
Step 3: Hypothesis Validation and Expert Consultation
Market hypotheses are validated through consultations with industry experts, stakeholders, and key players in the biosimilars market, ensuring the accuracy of the findings.
Step 4: Research Synthesis and Final Output
The final research output synthesizes the data gathered from multiple sources, presenting a comprehensive report on the USA biosimilar recombinant proteins market, including forecasts and strategic recommendations.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Increase in Chronic Diseases and Aging Population
Rising Healthcare Cost Pressures
Government Support for Biosimilar Adoption - Market Challenges
Regulatory Hurdles for Market Approval
High R&D Costs for Biosimilar Development
Price Sensitivity and Market Penetration Barriers - Market Opportunities
Emerging Markets for Biosimilars
Increased Focus on Biosimilar Market Access
Partnerships and Collaborations for Cost Reduction - Trends
Advancement in Biotechnological Innovations
Shift Towards Personalized Medicine
Regulatory Changes Favoring Biosimilars - Government Regulations
- SWOT Analysis of Key Competitors
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Monoclonal Antibodies
Erythropoiesis Stimulating Agents
Granulocyte Colony-Stimulating Factor
Insulin Biosimilars
Hormonal Biosimilars - By Platform Type (In Value%)
Mammalian Cell Culture Platforms
Bacterial Cell Culture Platforms
Insect Cell Culture Platforms
Plant-based Expression Platforms
Yeast-based Expression Platforms - By Fitment Type (In Value%)
On-premise Solutions
Cloud-based Solutions
Hybrid Solutions
Modular Solutions
Integrated Solutions - By End User Segment (In Value%)
Biopharmaceutical Companies
Hospitals and Clinics
Government Health Agencies
Research Institutions
Contract Manufacturing Organizations - By Procurement Channel (In Value%)
Direct Procurement
Government Tenders
Private Sector Procurement
Online Bidding Platforms
Third-party Distributors
- Market Share Analysis
- Cross Comparison Parameters (System Type, Platform Type, Procurement Channel, End User Segment, Fitment Type, Market Share, Regulatory Compliance, R&D Investment, Production Capacity, Geographic Reach)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Amgen Inc.
Sandoz International GmbH
Pfizer Inc.
Bristol-Myers Squibb
Samsung Bioepis
Celltrion Healthcare Co.
Mylan N.V.
Biocon Limited
Eli Lilly and Company
Teva Pharmaceuticals
Merck & Co.
Novartis AG
Roche Holding AG
AbbVie Inc.
Hikma Pharmaceuticals
- Rising Demand for Cost-effective Biologics
- Government Policies Supporting Biosimilar Access
- Increasing Use of Biosimilars in Oncology
- Emerging Focus on Immunology and Autoimmune Disorders
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035