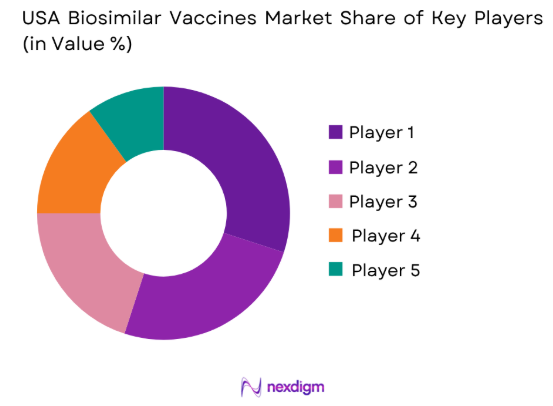
Market Overview
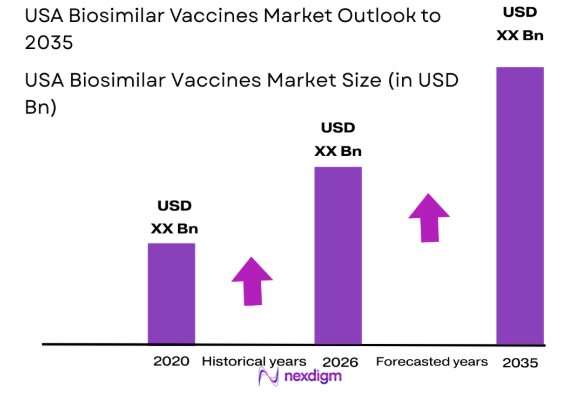
The USA biosimilar vaccines market is experiencing significant growth, driven by increasing healthcare demands and rising vaccine affordability concerns. Based on a recent historical assessment, the market size for biosimilar vaccines is expected to reach USD ~ billion. This growth is fueled by the increasing acceptance of biosimilars in both developed and emerging markets, driven by government initiatives and healthcare systems striving for cost-effective alternatives to originator biologics. Key drivers include the regulatory approval of new biosimilars and the cost-efficiency they bring to healthcare systems.
The dominant regions for biosimilar vaccines are primarily urban areas with robust healthcare infrastructure such as New York, California, and Texas. These cities offer a high demand for vaccines due to large populations and significant investments in healthcare facilities. Additionally, the growing adoption of biosimilar vaccines in the U.S. is further accelerated by regulatory support and initiatives aimed at improving access to affordable healthcare solutions. The U.S. government’s role in implementing cost-containment strategies, along with the increasing prevalence of diseases preventable by vaccines, further solidifies the dominance of these areas.
Market Segmentation
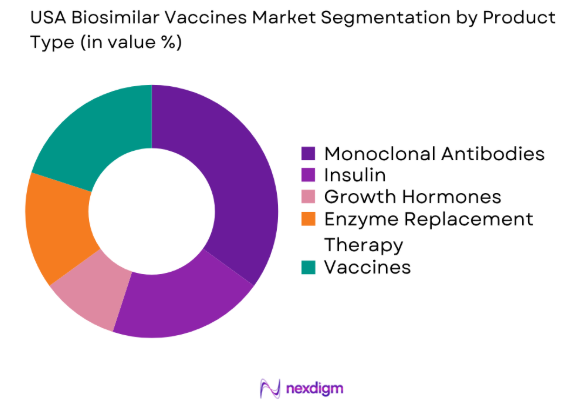
By Product Type
The USA biosimilar vaccines market is segmented by product type into monoclonal antibodies, insulin, growth hormones, enzyme replacement therapy, and vaccines. Recently, biosimilar vaccines have gained a dominant market share due to increased awareness and government policies supporting their use in public health programs. The demand for biosimilar vaccines is particularly high because of their cost-effectiveness compared to original biologics. This has made them an attractive option for healthcare providers and insurers aiming to reduce overall healthcare costs.
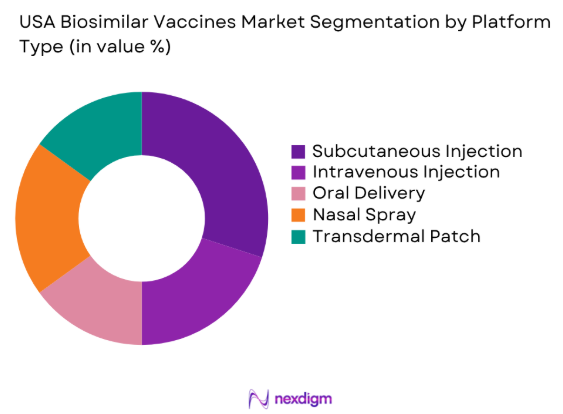
By Platform Type
The USA biosimilar vaccines market is segmented by platform type into subcutaneous injection platforms, intravenous injection platforms, oral delivery platforms, nasal spray platforms, and transdermal patch platforms. The subcutaneous injection platform has a dominant market share because of its ease of administration and increasing demand for injectable treatments, especially in chronic disease management. This platform’s reliability and user-friendliness make it an ideal choice for healthcare providers and patients, driving its market penetration.
Competitive Landscape
The competitive landscape in the USA biosimilar vaccines market is characterized by a few dominant players who influence the market through consolidation and strong partnerships. These companies are focusing on expanding their portfolios by entering into agreements with healthcare providers and governments to promote the uptake of biosimilar vaccines. The increasing competition is expected to drive innovation and lower prices, further fostering market growth.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD Billion) | Additional Market-Specific Parameter |
| Amgen | 1980 | Thousand Oaks, CA | ~ | ~ | ~ | ~ | ~ |
| Sandoz | 2003 | Holzkirchen, Germany | ~ | ~ | ~ | ~ | ~ |
| Biocon | 1978 | Bangalore, India | ~ | ~ | ~ | ~ | ~ |
| Pfizer | 1849 | New York, NY | ~ | ~ | ~ | ~ | ~ |
| Celltrion | 2002 | Incheon, South Korea | ~ | ~ | ~ | ~ | ~ |
USA Biosimilar Vaccines Market Analysis
Growth Drivers
Increased Acceptance of Biosimilars
The rapid adoption of biosimilars in the USA is largely driven by government initiatives to reduce healthcare costs. These efforts have made biosimilars an attractive option for healthcare providers and patients seeking more affordable alternatives to expensive biologics. The FDA’s increasing approval of biosimilars, supported by policies like the Biologics Price Competition and Innovation Act (BPCIA), has facilitated their integration into treatment regimens for chronic conditions such as cancer and autoimmune disorders. With demonstrated efficacy and safety comparable to reference biologics, biosimilars are gaining trust. This trend is expected to continue, as further biosimilars enter the market, driving sustained growth in the sector.
Rising Healthcare Costs
The USA biosimilar vaccines market is benefitting from increasing pressure on healthcare systems to reduce rising costs. As healthcare expenses climb, both public and private sectors are focused on lowering the costs of biologic drugs, which are major contributors to healthcare spending. Biosimilars offer affordable alternatives without sacrificing quality, making them appealing to governments, insurance companies, and healthcare providers. With the USA facing an aging population and growing chronic disease burden, the demand for cost-effective treatment options, like biosimilar vaccines, continues to rise. This is especially true for vaccines that prevent high-cost diseases, such as influenza and pneumococcal infections, boosting biosimilar demand in vaccination programs.
Market Challenges
Regulatory Hurdles
The USA biosimilar vaccines market faces significant challenges due to complex regulatory processes. The FDA’s stringent approval requirements and the extensive clinical trials needed to demonstrate efficacy and safety can delay market entry. While the Biologics Price Competition and Innovation Act (BPCIA) has streamlined approval, navigating the complex regulatory framework remains a challenge for new players. Additionally, post-market surveillance to ensure long-term safety can slow growth. Regulatory hurdles are especially pronounced in the vaccine segment, where high standards for immunogenicity and safety must be met. These barriers can delay the commercialization of promising biosimilars, especially for smaller companies aiming to enter the market.
Market Perception and Acceptance
Despite growing regulatory support, biosimilars often face resistance from physicians and patients who are hesitant to switch from branded biologics to biosimilars. There are concerns about the clinical outcomes and safety profiles of biosimilars, which can slow their adoption. This challenge is particularly prevalent in the vaccine segment, where public trust in vaccines is already sensitive. Furthermore, concerns over the potential for reduced efficacy and adverse effects with biosimilars could limit physician willingness to prescribe them, particularly for high-stakes treatments like vaccines. Overcoming these perceptions will require targeted educational campaigns to inform both healthcare professionals and patients about the benefits and safety of biosimilars.
Opportunities
Expansion of Vaccination Programs
One of the most significant opportunities for the USA biosimilar vaccines market lies in the expansion of vaccination programs in both the public and private sectors. With an increasing focus on preventative healthcare, government initiatives are promoting the use of vaccines to reduce the burden of infectious diseases. Biosimilar vaccines, which provide cost-effective alternatives to traditional vaccines, are well-positioned to benefit from this trend. As the cost-effectiveness of biosimilars becomes more recognized, their inclusion in national vaccination programs, especially in underserved populations, will likely grow. Additionally, the growing trend of travel vaccination, as well as the need for vaccines targeting emerging diseases, provides a lucrative opportunity for biosimilar vaccine development and adoption.
Technological Advancements in Vaccine Development
An opportunity for growth in the USA biosimilar vaccines market lies in technological advancements in vaccine development. New vaccine delivery technologies, such as mRNA-based vaccines and innovative adjuvants, are expected to drive growth in the biosimilar vaccine segment. These advancements enable biosimilar manufacturers to develop vaccines that are more effective, easier to administer, and better accepted by healthcare systems. Additionally, integrating cutting-edge technologies to enhance the production process and scalability of biosimilars can lower production costs, making them more attractive to healthcare providers and patients. As new vaccine technologies continue to emerge, biosimilar manufacturers must leverage these innovations to remain competitive and capture a larger share of the market.
Future Outlook
The future of the USA biosimilar vaccines market looks promising, with continued growth expected over the next five years. Technological innovations, such as mRNA-based vaccines, and increased regulatory support are likely to drive the market’s expansion. As biosimilars become more widely accepted and integrated into healthcare systems, demand for affordable vaccines will increase, particularly in public health programs. Governments and private healthcare systems will continue to push for cost-effective alternatives to high-priced biologics, creating further opportunities for biosimilar manufacturers.
Major Players
- Amgen
- Sandoz
- Biocon
- Pfizer
- Celltrion
- Mylan
- AbbVie
- Merck
- Bristol-Myers Squibb
- AstraZeneca
- Novartis
- GSK
- Roche
- Johnson & Johnson
- Eli Lilly
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Healthcare providers
- Pharmaceutical manufacturers
- Biotechnology firms
- Insurance companies
- Public health organizations
- Private sector technology firms
Research Methodology
Step 1: Identification of Key Variables
Market segmentation, demand drivers, and regulatory landscapes are identified to understand the factors influencing market growth.
Step 2: Market Analysis and Construction
Market trends, opportunities, and challenges are analyzed to construct a detailed picture of the market’s dynamics.
Step 3: Hypothesis Validation and Expert Consultation
Industry experts validate the hypotheses through interviews and consultations to ensure the accuracy of findings.
Step 4: Research Synthesis and Final Output
The research findings are synthesized into actionable insights and the final report is generated to assist stakeholders in decision-making.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Increased Acceptance of Biosimilars
Rising Demand for Cost-Effective Vaccines
Growing Healthcare Accessibility in Emerging Markets - Market Challenges
Regulatory Hurdles and Compliance Issues
Intellectual Property Barriers
Limited Awareness of Biosimilar Benefits - Market Opportunities
Expansion of Vaccination Programs in Developing Regions
Partnerships Between Biosimilar Producers and Governments
Technological Advancements in Vaccine Development - Trends
Rise in Immunization Campaigns
Technological Innovations in Vaccine Formulation
Increased Competition in the Biosimilar Vaccine Market - Government Regulations
- SWOT Analysis of Key Competitors
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Biosimilar Monoclonal Antibodies
Biosimilar Insulin
Biosimilar Growth Hormones
Biosimilar Enzyme Replacement Therapy
Biosimilar Vaccines - By Platform Type (In Value%)
Subcutaneous Injection Platforms
Intravenous Injection Platforms
Oral Delivery Platforms
Nasal Spray Platforms
Transdermal Patch Platforms - By Fitment Type (In Value%)
On-Premise Solutions
Cloud-Based Solutions
Hybrid Solutions
Modular Solutions
Integrated Solutions - By End User Segment (In Value%)
Healthcare Providers
Pharmaceutical Manufacturers
Biotechnology Firms
Government and Non-Governmental Health Agencies
Private Sector / Technology Firms - By Procurement Channel (In Value%)
Direct Procurement
Government Tenders
Private Sector Procurement
Online Bidding Platforms
Third-Party Distributors
- Market Share Analysis
- Cross Comparison Parameters(System Type, Platform Type, Procurement Channel, End User Segment, Fitment Type, Vaccine Type, Regulatory Approval Process, Delivery Method, Pricing Model, Geographic Reach)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Amgen
Sandoz
Biocon
Mylan
Pfizer
Bayer
Boehringer Ingelheim
Teva Pharmaceuticals
Novartis
Celltrion
Samsung Biologics
LG Chem
GSK
Eli Lilly
Roche
- Increase in Healthcare Providers’ Adoption of Biosimilars
- Government Agencies’ Role in Biosimilar Policy Making
- Healthcare Providers’ Focus on Affordable Vaccines
- Private Sector’s Growing Demand for Biosimilars
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035