Market Overview
The USA Cell Therapy Market is currently valued at approximately USD ~ billion based on a recent historical assessment according to industry reports, reflecting a robust revenue base supported by the expanding adoption of cellular treatments across therapeutic areas such as oncology, rare diseases, and autoimmune disorders. This market scale is largely driven by rapid advancements in cell manipulation techniques, increasing numbers of clinical trials demonstrating therapeutic efficacy, and substantial investment in research and development from both private biotechnology firms and public funding sources.
Dominance in the USA market stems from its strong biotechnology infrastructure, well‑established regulatory frameworks, and concentration of leading cell therapy developers and treatment centers. Major hubs such as Boston, San Francisco, and San Diego host extensive research ecosystems that attract talent, capital, and collaboration between academic institutions and industry. The presence of advanced manufacturing facilities and comprehensive clinical networks further reinforces the country’s leading position, enabling accelerated development and adoption of innovative cell therapy solutions within its healthcare system.
Market Segmentation
By Product Type
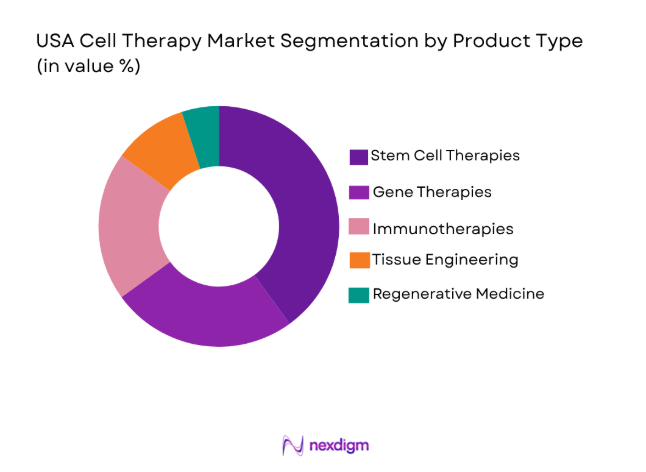
USA Cell Therapy market is segmented by product type into stem cell therapies, gene therapies, immunotherapies, tissue engineering, and regenerative medicine. Recently, stem cell therapies have seen dominant market share due to their wide application in treating a variety of diseases, their ongoing advancements, and their established clinical efficacy. Stem cells are being increasingly used in treating cancer, cardiovascular diseases, and neurological disorders. The demand is also fueled by research funding and the growing number of clinical trials that continue to validate their therapeutic potential.
By Platform Type
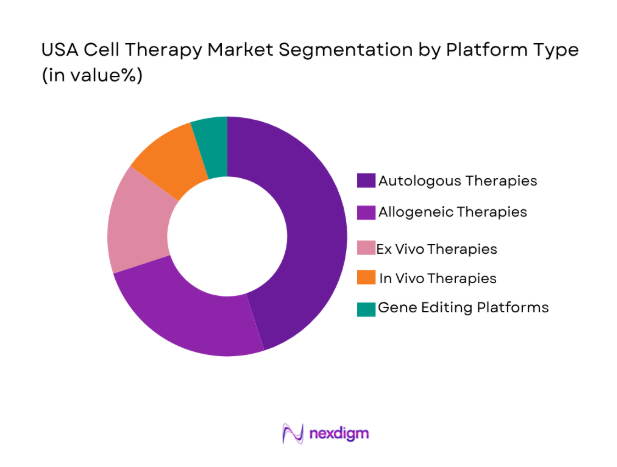
USA Cell Therapy market is segmented by platform type into autologous therapies, allogeneic therapies, ex vivo therapies, in vivo therapies, and gene editing platforms. Recently, autologous therapies have seen a dominant market share due to their personalized approach, wherein the patient’s own cells are used for treatment. This method reduces the risk of immune rejection and has gained traction across various disease treatments, including cancers and autoimmune disorders. The increased adoption of autologous therapies is further fueled by advances in manufacturing processes and successful clinical outcomes, making it the preferred choice for many treatment protocols.
Competitive Landscape
The competitive landscape in the USA Cell Therapy market is characterized by major players heavily investing in research and development, resulting in continuous advancements in therapies and technologies. Market consolidation is evident, with several key players forming strategic partnerships and acquisitions to strengthen their position. The competitive environment is driven by innovation, with companies focusing on enhancing the safety and efficacy of their cell-based therapies.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Key Market-Specific Parameter |
| Gilead Sciences | 1987 | Foster City, USA | ~ | ~ | ~ | ~ | ~ |
| Novartis | 1996 | Basel, Switzerland | ~ | ~ | ~ | ~ | ~ |
| Celgene Corporation | 1986 | Summit, USA | ~ | ~ | ~ | ~ | ~ |
| Kite Pharma | 2009 | Santa Monica, USA | ~ | ~ | ~ | ~ | ~ |
| Bluebird Bio | 1992 | Cambridge, USA | ~ | ~ | ~ | ~ | ~ |
USA Cell Therapy Market Analysis
Growth Drivers
Government Investment in Healthcare
Government investment in healthcare has been a key driver for the USA Cell Therapy market. The federal and state governments have significantly increased funding for cell therapy research and development, providing grants and incentives for clinical trials, product development, and the commercialization of cell-based therapies. This funding has enabled biotechnology companies to advance their research processes and overcome challenges related to high capital expenditure. Programs like the NIH’s Stem Cell Research Initiative and the Cancer Moonshot initiative have been crucial in driving innovation and reducing financial barriers for research entities, thereby ensuring that novel cell therapies progress into clinical applications. The strategic focus on cell and gene therapy by government agencies has fostered an ecosystem conducive to research collaborations, public-private partnerships, and infrastructure development, thus creating a solid foundation for long-term market growth. Additionally, the establishment of clear regulatory pathways for cell therapies has encouraged a consistent flow of public funding into cutting-edge therapies, further accelerating progress. As a result, government investment continues to be a pivotal factor in supporting the commercialization and widespread adoption of cell therapies in the USA.
Technological Advancements in Gene Therapy
Technological advancements in gene therapy have played an instrumental role in driving the USA Cell Therapy market. The advent of CRISPR-Cas9 gene editing, alongside other breakthrough technologies, has revolutionized the potential to treat genetic disorders at the molecular level by directly modifying genes within patients’ cells. This precision-based approach is enabling scientists and clinicians to correct inherited genetic defects, cure certain cancers, and treat viral infections in ways that were previously unimaginable. The increased efficiency and accuracy of these techniques have rapidly accelerated gene therapy adoption across various medical specialties, fueling an expansion in treatment options for rare genetic diseases, complex cancers, and chronic conditions. Additionally, advancements in gene delivery systems and vectors, such as lentiviral and adeno-associated virus vectors, have improved the therapeutic efficacy of gene therapies, making them safer and more reliable for clinical use. As the scientific community continues to refine gene-editing technologies and expand their capabilities, interest in gene therapies continues to grow, leading to increased investments, innovation, and adoption of these treatments. Technological advancements in gene therapy have opened doors to the next generation of medical interventions, solidifying the market’s trajectory toward sustained growth and market penetration.
Market Challenges
High Cost of Cell Therapies
One of the most significant challenges facing the USA Cell Therapy market is the high cost of these therapies. The development, production, and administration of cell therapies require specialized infrastructure, advanced technology, and highly trained professionals, all of which contribute to the overall expense. Cell-based treatments, especially gene therapies and stem cell therapies, involve complex and highly personalized procedures that require tailored manufacturing processes, which makes them considerably more expensive than traditional therapies. These high costs often make it difficult for patients to access cutting-edge treatments, particularly in populations without extensive insurance coverage. Moreover, the reimbursement landscape remains a critical issue, as insurers and government healthcare programs struggle to adopt sustainable coverage models for high-cost cell therapies. Despite the potential long-term benefits and life-saving effects of these treatments, the prohibitive costs create a significant barrier to broader market adoption and patient access. As a result, the USA Cell Therapy market faces an ongoing challenge in balancing the cost of advanced treatments with the need to make them accessible and affordable for a larger patient population. The development of more cost-efficient manufacturing processes and broader insurance coverage will be essential to overcoming this challenge and facilitating widespread use of these innovative therapies.
Regulatory Hurdles and Approval Process
The regulatory hurdles and approval processes for cell therapies represent another key challenge for the USA market. Given the novel and complex nature of cell-based treatments, regulatory agencies like the FDA require extensive clinical data to demonstrate the safety, efficacy, and long-term benefits of these therapies. This lengthy approval process can delay the introduction of new therapies to the market, prolonging patient access to potentially life-saving treatments. Furthermore, the intricacies of manufacturing and distributing cell therapies add another layer of regulatory complexity. As these therapies are often highly personalized, ensuring consistency, quality control, and compliance across production batches becomes a challenging task for manufacturers. Regulatory uncertainties also arise from differing requirements in international markets, which complicates global market expansion for companies. The high cost of clinical trials, coupled with the unpredictable regulatory pathway, can deter smaller biotech firms from pursuing cell therapy development, limiting innovation. Despite these challenges, the FDA has been taking steps to streamline regulatory processes for cell and gene therapies by implementing initiatives like the Regenerative Medicine Advanced Therapy (RMAT) designation. However, regulatory hurdles remain a significant barrier to the widespread commercialization of cell therapies in the USA, and their successful navigation will be crucial for market growth.
Opportunities
Expansion of Cell Therapy Applications
The expansion of cell therapy applications presents one of the most promising opportunities for the USA Cell Therapy market. Initially, cell therapies were focused on treating a narrow set of diseases, but research and clinical trials are increasingly exploring the potential of these therapies across a wide array of therapeutic areas. The growing understanding of the regenerative capabilities of stem cells, alongside advancements in gene therapy, is driving this diversification. For example, cell therapies are being investigated for their potential to treat neurological diseases such as Alzheimer’s, Parkinson’s, and spinal cord injuries, as well as for cardiovascular conditions, autoimmune diseases, and chronic lung diseases. This broadening of applications presents a wealth of opportunities for innovation and market growth. Additionally, the development of combination therapies, which combine cell therapies with other treatment modalities like immunotherapies or targeted drugs, is expected to unlock new avenues for therapeutic advancements. As research continues to uncover the full potential of cell therapies, expanding their use into previously underserved areas will be a key driver for market growth and diversification. Companies that are able to successfully innovate and apply cell therapies to a broader range of diseases will be well-positioned to lead the next phase of growth in the market.
Emerging Markets for Cell Therapies
Emerging markets represent a significant growth opportunity for the USA Cell Therapy market. As healthcare infrastructures improve in developing countries, the demand for advanced and cutting-edge medical treatments is on the rise. Cell therapies, which are often perceived as state-of-the-art treatments, have the potential to address unmet medical needs in regions where traditional therapies may be insufficient. Many emerging markets, particularly in Asia-Pacific, Latin America, and Africa, are experiencing a rise in healthcare spending, driven by an expanding middle class, increased government investments in healthcare, and a growing focus on modernizing healthcare systems. These regions offer an untapped market for cell therapies, where the availability of advanced treatment options is limited, and where governments and private organizations are eager to bring innovative therapies to address the rising burden of chronic diseases. Expanding into these emerging markets presents an opportunity for USA-based biotechnology companies to broaden their market reach and tap into a new and rapidly growing consumer base. By offering affordable, accessible, and effective cell therapies, companies can capture significant market share and contribute to improving global health outcomes in these regions.
Future Outlook
The future of the USA Cell Therapy Market looks promising, with expected growth driven by technological advancements in gene editing and stem cell research. Innovation in gene therapies and their increasing clinical success across various diseases are expected to be key contributors. Alongside technological developments, regulatory support and favorable policies for clinical trials will play a significant role in shaping the market’s expansion. The demand for cell therapies is anticipated to rise, supported by improvements in healthcare infrastructure and access to cutting-edge treatments.
Major Players
- Gilead Sciences
- Novartis
- Celgene Corporation
- Kite Pharma
- Bluebird Bio
- Regeneron Pharmaceuticals
- Biogen
- Merck & Co.
- Amgen
- Abbott Laboratories
- Genentech
- Bristol-Myers Squibb
- Johnson & Johnson
- Vertex Pharmaceuticals
- Eli Lilly & Co.
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Pharmaceutical and biotechnology companies
- Research and academic institutions
- Healthcare providers
- Clinical research organizations
- Hospitals and treatment centers
Research Methodology
Step 1: Identification of Key Variables
We identify key market drivers, trends, and challenges that impact the USA Cell Therapy market by analyzing existing data and expert insights.
Step 2: Market Analysis and Construction
We perform a comprehensive analysis of market conditions, including the study of historical data, competitor strategies, and growth forecasts.
Step 3: Hypothesis Validation and Expert Consultation
We validate market hypotheses and forecasts through consultations with industry experts, including executives, healthcare professionals, and regulatory bodies.
Step 4: Research Synthesis and Final Output
We compile the data, validate it through multiple rounds of expert review, and prepare the final report that includes actionable insights and recommendations.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Advancements in Gene Editing Technology
Rising Prevalence of Chronic Diseases
Increasing Government Investment in Healthcare - Market Challenges
High Cost of Cell Therapy
Complex Regulatory Processes
Limited Infrastructure for Cell Therapy - Market Opportunities
Expansion of Cell Therapy into Emerging Markets
Advancements in Personalized Medicine
Partnerships with Biopharma Companies - Trends
Increased Investment in Stem Cell Research
Adoption of AI and Machine Learning in Cell Therapy - Government Regulations
- SWOT Analysis
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Stem Cell Therapy
Gene Therapy
Immunotherapy
Tissue Engineering
Regenerative Medicine - By Platform Type (In Value%)
Autologous Therapies
Allogeneic Therapies
Ex Vivo Therapies
In Vivo Therapies
Gene Editing Platforms - By Fitment Type (In Value%)
Hospital-based Therapies
Clinic-based Therapies
Research Institutions
Outpatient Care
Long-term Care Facilities - By End User Segment (In Value%)
Hospitals
Research Laboratories
Biopharmaceutical Companies
Clinical Research Organizations
- Market Share Analysis
- Cross Comparison Parameters (Stem Cell Therapy, Gene Therapy, Immunotherapy, Tissue Engineering, Regenerative Medicine, Autologous Therapies, Allogeneic Therapies)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Gilead Sciences
Novartis
Celgene Corporation
Bluebird Bio
Thermo Fisher Scientific
Kite Pharma
Juno Therapeutics
Sangamo Therapeutics
Regeneron Pharmaceuticals
Biogen
Merck & Co.
AbbVie
Amgen
GlaxoSmithKline
Eli Lilly & Co.
- Increasing Demand from Oncology
- Growing Usage in Autoimmune Disorders
- Rising Adoption in Neurological Disorders
- Development of Advanced Cell Therapy Applications
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035


