Market Overview
The USA Diagnostic Biomarkers market demonstrates strong maturity with robust integration across clinical diagnostics and pharmaceutical research, supported by technological innovation and high healthcare expenditure. Based on a recent historical assessment, the market size reached approximately USD ~ billion, driven by increasing adoption of precision medicine, rising chronic disease burden, and demand for early detection tools. Significant investments in genomics, proteomics, and digital biomarker technologies further accelerate growth across healthcare ecosystems.
The market is predominantly concentrated in advanced healthcare hubs such as New York, Boston, San Francisco, and San Diego, where biotechnology clusters and research institutions are highly developed. These regions benefit from strong presence of pharmaceutical companies, diagnostic laboratories, and funding ecosystems. High patient awareness, advanced infrastructure, and rapid adoption of innovative diagnostic technologies contribute to their leadership, supported by favorable regulatory frameworks and continuous research collaborations.
Market Segmentation
By Product Type
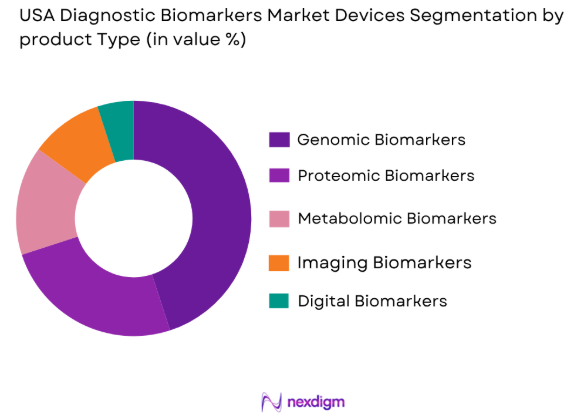
USA Diagnostic Biomarkers market is segmented by product type into genomic biomarkers, proteomic biomarkers, metabolomic biomarkers, imaging biomarkers, and digital biomarkers. Recently, genomic biomarkers has a dominant market share due to factors such as widespread adoption in oncology diagnostics, strong integration with next-generation sequencing technologies, and increasing demand for personalized medicine. Pharmaceutical companies and diagnostic labs heavily rely on genomic data for drug development and disease prediction, supported by extensive research funding and clinical trials, making this segment the most influential in shaping overall market demand.
By End User
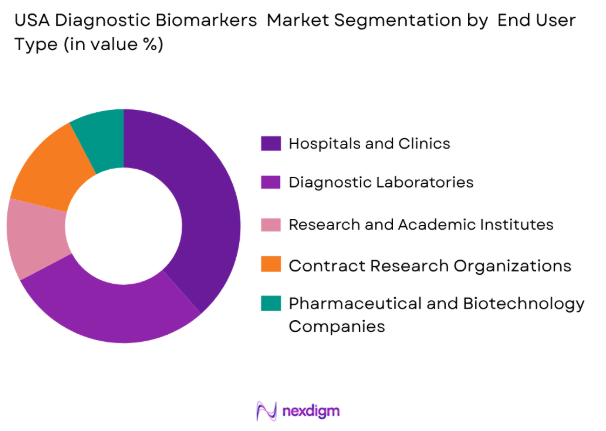
USA Diagnostic Biomarkers market is segmented by end user into hospitals and clinics, diagnostic laboratories, research and academic institutes, pharmaceutical and biotechnology companies, and contract research organizations. Recently, hospitals and clinics has a dominant market share due to factors such as high patient inflow, availability of advanced diagnostic infrastructure, and integration of biomarker-based testing into routine clinical workflows. These facilities are primary points of care where early disease detection and treatment decisions are made, leading to higher utilization of biomarker technologies compared to other end users.
Competitive Landscape
The competitive landscape of the USA Diagnostic Biomarkers Market is characterized by a mix of local and international players, with market consolidation occurring as key players continue to innovate and improve their product offerings. Large multinational companies are establishing a strong presence in the market, aided by their established brand reputations and ability to offer high-quality, technologically advanced systems. Smaller, local players are also contributing to the competition by offering cost-effective and portable solutions for the growing demand in rural and underserved areas. This competitive environment is fostering a dynamic market with continuous improvements in product features and pricing strategies.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Additional Parameter |
| Philips Healthcare | 1891 | Netherlands | ~ | ~ | ~ | ~ | ~ |
| Siemens Healthineers | 1847 | Germany | ~ | ~ | ~ | ~ | ~ |
| GE Healthcare | 1892 | USA | ~ | ~ | ~ | ~ | ~ |
| Canon Medical Systems | 1933 | Japan | ~ | ~ | ~ | ~ | ~ |
| Medtronic | 1949 | Ireland | ~ | ~ | ~ | ~ | ~ |
USA Diagnostic Biomarkers Market Analysis
Growth Drivers
Rising Adoption of Precision Medicine and Personalized Healthcare
The increasing emphasis on precision medicine and personalized healthcare is significantly driving the growth of the USA Diagnostic Biomarkers market as healthcare providers shift toward tailored treatment approaches based on individual patient profiles. This transformation is supported by advancements in genomic sequencing technologies, enabling clinicians to identify disease-specific biomarkers with higher accuracy and efficiency. The growing prevalence of chronic diseases such as cancer, cardiovascular disorders, and neurological conditions has further intensified the need for early and precise diagnostic tools. Biomarkers play a crucial role in predicting disease progression and treatment response, which enhances clinical decision-making and improves patient outcomes. Additionally, pharmaceutical companies are increasingly incorporating biomarkers into drug development processes to enhance clinical trial success rates and reduce development costs. The integration of artificial intelligence and machine learning in biomarker discovery is also accelerating innovation by enabling faster data analysis and pattern recognition. Increased patient awareness regarding early diagnosis and preventive healthcare is further contributing to the demand for biomarker-based diagnostics. Government initiatives supporting precision medicine programs and funding research activities have also created a favorable environment for market expansion. Healthcare systems are continuously investing in advanced diagnostic infrastructure to improve efficiency and reduce long-term treatment costs. As a result, the widespread adoption of precision medicine continues to serve as a primary growth catalyst for the diagnostic biomarkers market across the United States.
Technological Advancements in Multi-Omics and Digital Biomarkers
Continuous advancements in multi-omics technologies, including genomics, proteomics, and metabolomics, are significantly propelling the growth of the USA Diagnostic Biomarkers market by enabling comprehensive disease profiling. These technologies allow researchers and clinicians to analyze complex biological systems and identify multiple biomarkers simultaneously, improving diagnostic accuracy and reliability. The emergence of digital biomarkers derived from wearable devices and mobile health applications is further transforming the diagnostic landscape by enabling real-time health monitoring and remote patient management. This shift toward digital healthcare solutions is particularly beneficial for managing chronic diseases and reducing hospital visits. Advanced data analytics tools are playing a critical role in interpreting large volumes of biological data, leading to more precise and actionable insights. Collaborations between technology companies and healthcare providers are accelerating the development and commercialization of innovative diagnostic solutions. Regulatory bodies are also increasingly recognizing the importance of digital biomarkers, leading to the establishment of guidelines that facilitate their adoption. The integration of cloud computing and big data platforms is enhancing the scalability and accessibility of biomarker-based diagnostics. As a result, technological advancements continue to drive efficiency, innovation, and adoption across various healthcare settings, reinforcing the market’s long-term growth trajectory.
Market Challenges
High Cost of Biomarker Discovery and Clinical Validation Processes
The high cost associated with biomarker discovery and clinical validation represents a significant challenge for the USA Diagnostic Biomarkers market, as the process requires substantial investment in research, technology, and skilled personnel. Developing a clinically validated biomarker involves extensive laboratory testing, data analysis, and regulatory approvals, which can take several years and require significant financial resources. Small and mid-sized biotechnology firms often face difficulties in securing funding for such large-scale projects, limiting innovation and market entry. Additionally, the complexity of biological systems makes it challenging to identify biomarkers that are both highly sensitive and specific, leading to increased research costs and failure rates. Clinical trials for biomarker validation are also expensive and time-consuming, as they require large patient populations and long-term studies to establish reliability. Regulatory requirements further add to the cost burden, as companies must comply with stringent guidelines to ensure safety and efficacy. The lack of standardized protocols across different diagnostic platforms can also increase costs by requiring additional validation efforts. Furthermore, reimbursement challenges can hinder the adoption of expensive biomarker tests, particularly in cost-sensitive healthcare settings. As a result, the high cost of development and validation remains a critical barrier to market growth and innovation.
Regulatory Complexities and Data Privacy Concerns
Regulatory complexities and data privacy concerns pose significant challenges to the growth of the USA Diagnostic Biomarkers market, as companies must navigate a highly regulated environment to bring new diagnostic products to market. Regulatory bodies such as the FDA impose stringent requirements for the approval of biomarker-based tests, including extensive clinical validation and documentation. These processes can be time-consuming and may delay product launches, affecting overall market growth. Additionally, the evolving nature of biomarker technologies often outpaces regulatory frameworks, creating uncertainty for companies regarding compliance requirements. Data privacy is another critical concern, particularly with the increasing use of digital biomarkers and health data collected from wearable devices. Ensuring the security and confidentiality of patient data is essential to maintain trust and comply with regulations such as HIPAA. The integration of advanced technologies such as AI and cloud computing further complicates data management and security. Companies must invest heavily in cybersecurity measures and compliance systems to mitigate risks, increasing operational costs. Cross-border data sharing for research and clinical trials also presents regulatory challenges due to varying international standards. As a result, regulatory and privacy-related issues continue to act as significant obstacles for market expansion and innovation.
Opportunities
Expansion of Liquid Biopsy Technologies in Oncology Diagnostics
The expansion of liquid biopsy technologies presents a significant opportunity for the USA Diagnostic Biomarkers market, particularly in the field of oncology diagnostics where non-invasive testing methods are gaining widespread acceptance. Liquid biopsies enable the detection of cancer-related biomarkers from blood samples, eliminating the need for invasive tissue biopsies and improving patient comfort. This technology allows for early cancer detection, monitoring of disease progression, and assessment of treatment response, making it a valuable tool in precision oncology. Increasing research investments and clinical trials focused on liquid biopsy applications are accelerating technological advancements and commercialization. Pharmaceutical companies are also leveraging liquid biopsy biomarkers to enhance drug development and clinical trial efficiency. The growing demand for personalized cancer treatment is further driving the adoption of these technologies across healthcare settings. Regulatory agencies are gradually establishing frameworks to support the approval and adoption of liquid biopsy tests, which is expected to boost market growth. Collaborations between biotech firms and diagnostic companies are fostering innovation and expanding product pipelines. Additionally, the integration of AI and advanced analytics is improving the accuracy and reliability of liquid biopsy results. As awareness and accessibility increase, liquid biopsy technologies are expected to create substantial growth opportunities in the diagnostic biomarkers market.
Integration of Artificial Intelligence in Biomarker Discovery and Diagnostics
The integration of artificial intelligence in biomarker discovery and diagnostics offers a transformative opportunity for the USA Diagnostic Biomarkers market by enhancing efficiency, accuracy, and scalability. AI algorithms can analyze vast datasets from genomic, proteomic, and clinical sources to identify patterns and correlations that may not be detectable through traditional methods. This capability significantly accelerates the biomarker discovery process, reducing time and cost associated with research and development. In clinical settings, AI-powered diagnostic tools can assist healthcare professionals in making more accurate and timely decisions, improving patient outcomes. The adoption of AI is also enabling the development of predictive models for disease risk assessment and treatment planning. Technology companies are increasingly collaborating with healthcare providers to integrate AI solutions into diagnostic workflows. Regulatory bodies are beginning to recognize the potential of AI in healthcare, leading to the development of guidelines that support its adoption. Cloud-based AI platforms are further enhancing accessibility and scalability, allowing smaller organizations to leverage advanced analytics. As AI technology continues to evolve, it is expected to play a crucial role in shaping the future of biomarker-based diagnostics and driving market growth.
Future Outlook
The USA Diagnostic Biomarkers market is expected to witness sustained growth over the next five years, driven by continuous advancements in precision medicine and multi-omics technologies. Increasing adoption of digital biomarkers and AI-based diagnostics will enhance clinical decision-making and efficiency. Regulatory support for innovative diagnostic solutions and growing investments in healthcare infrastructure will further accelerate market expansion. Additionally, rising demand for early disease detection and personalized treatment will continue to shape the market landscape.
Major Players
- Roche Diagnostics
- Abbott Laboratories
- Thermo Fisher Scientific
- Danaher Corporation
- Bio-Rad Laboratories
- Siemens Healthineers
- Agilent Technologies
- Illumina Inc.
- QIAGEN N.V.
- PerkinElmer Inc.
- Hologic Inc.
- Myriad Genetics
- Exact Sciences Corporation
- Guardant Health
- BioMérieux SA
Key Target Audience
- Pharmaceutical and biotechnology companies
- Diagnostic laboratories
- Hospitals and healthcare providers
- Contract research organizations
- Medical device manufacturers
- Investments and venture capitalist firm
- Government and regulatory bodies
- Healthcare insurance companies
Research Methodology
Step 1: Identification of Key Variables
Key variables influencing the market such as technology adoption, disease prevalence, regulatory environment, and investment trends are identified. Both dependent and independent variables are mapped to understand their impact on market growth and structure. This step ensures a strong analytical foundation for further research.
Step 2: Market Analysis and Construction
Comprehensive data is collected from primary and secondary sources, including company reports, healthcare databases, and industry publications. The market is constructed using validated data points, ensuring accurate representation of size, segmentation, and trends. Analytical models are applied to derive meaningful insights.
Step 3: Hypothesis Validation and Expert Consultation
Initial findings and assumptions are validated through consultations with industry experts, healthcare professionals, and key stakeholders. Feedback is incorporated to refine the analysis and ensure reliability. This step helps in eliminating biases and strengthening the accuracy of conclusions.
Step 4: Research Synthesis and Final Output
All validated data and insights are compiled into a structured format to create the final report. The research is synthesized to provide actionable intelligence, clear trends, and strategic recommendations. Quality checks are conducted to ensure consistency, accuracy, and completeness of the report.
- Executive Summary
- Research Methodology (DefinitionXs, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Driver
Rising prevalence of chronic and infectious diseases
Advancements in precision medicine and personalized diagnostics
Increasing adoption of early disease detection technologies - Market Challenges
High cost of biomarker discovery and validation
Regulatory complexities and approval delays
Limited standardization across diagnostic platforms - Market Opportunities
Expansion of liquid biopsy applications
Growing investments in biomarker research and development
Integration of AI and big data analytics in diagnostics - Trends
Shift towards non-invasive diagnostic techniques
Increasing use of multi-omics approaches
Adoption of digital and wearable-based biomarkers - Government Regulations
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Porter’s Five Forces
- By Market Value 2020-2025
- By Installed Units 2020-2025
- By Average System Price 2020-2025
- By System Complexity Tier 2020-2025
- By Product Type (In Value%)
Genomic Biomarkers
Proteomic Biomarkers
Metabolomic Biomarkers
Imaging Biomarkers
Digital Biomarkers - By Platform Type (In Value%)
PCR-Based Platforms
Next-Generation Sequencing Platforms
Immunoassay Platforms
Microarray Platforms
Mass Spectrometry Platforms - By Fitment Type (In Value%)
Standalone Diagnostic Systems
Integrated Laboratory Systems
Point-of-Care Devices
Portable Diagnostic Devices
Cloud-Connected Diagnostic Platforms - By EndUser Segment (In Value%
Hospitals and Clinics
Diagnostic Laboratories
Research and Academic Institutes
Pharmaceutical and Biotechnology Companies
Contract Research Organizations - By Procurement Channel (In Value%)
Direct Procurement from Manufacturers
Distributor-Based Procurement
Online Procurement Platforms
Government Tenders and Contracts
Group Purchasing Organizations
- Market Share Analysis
- Cross Comparison Parameters (Product Portfolio Breadth, Technology Innovation, Regulatory Approvals, Pricing Strategy, Geographic Presence)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Roche Diagnostics
Abbott Laboratories
Thermo Fisher Scientific
Danaher Corporation
Bio-Rad Laboratories
Siemens Healthineers
Agilent Technologies
Illumina Inc.
QIAGEN N.V.
PerkinElmer Inc.
Hologic Inc.
Myriad Genetics
Exact Sciences Corporation
Guardant Health
BioMérieux SA
- Hospitals leading adoption due to advanced diagnostic infrastructure
- Diagnostic labs driving volume through routine testing services
- Pharma companies leveraging biomarkers for drug development
- Research institutes focusing on innovation and biomarker discovery
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035