Market Overview
The USA diagnostic labs market has seen substantial growth, driven by technological advancements and the rising demand for healthcare services. Based on recent historical assessments, the market size is expected to reach USD ~ billion, largely supported by increasing investments in healthcare infrastructure and diagnostics. The growth is also propelled by the rising incidence of chronic diseases, aging populations, and healthcare digitization, with healthcare providers continually upgrading their diagnostic capabilities to meet the demand for accurate and timely results.
The USA diagnostic labs market is dominated by several large urban areas, particularly metropolitan hubs such as New York, Los Angeles, and Chicago. These cities are known for their robust healthcare systems, high patient volumes, and significant investments in diagnostic technologies. The concentration of healthcare facilities and the increasing adoption of digital health solutions in these regions contribute to their dominance. Additionally, government support for healthcare infrastructure and a higher number of diagnostic labs in these regions further strengthens their position in the market.
Market Segmentation
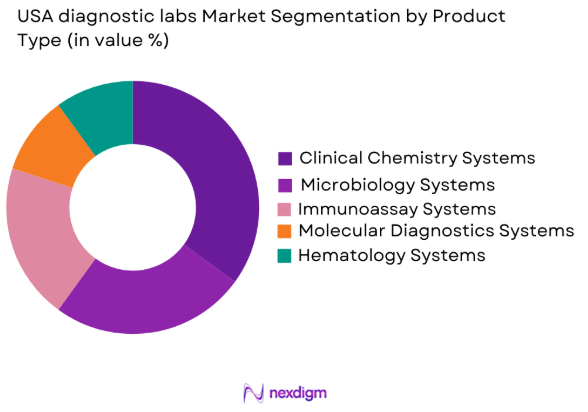
By Product Type
The USA diagnostic labs market is segmented by product type into clinical chemistry systems, microbiology systems, immunoassay systems, molecular diagnostics systems, and hematology systems. Recently, the clinical chemistry systems sub-segment has dominated the market due to the growing need for diagnostic tests related to chronic conditions, such as diabetes and cardiovascular diseases. With the rise in preventive healthcare and disease management, clinical chemistry systems have gained widespread adoption across diagnostic laboratories, driven by their ability to deliver accurate and cost-effective diagnostic results.
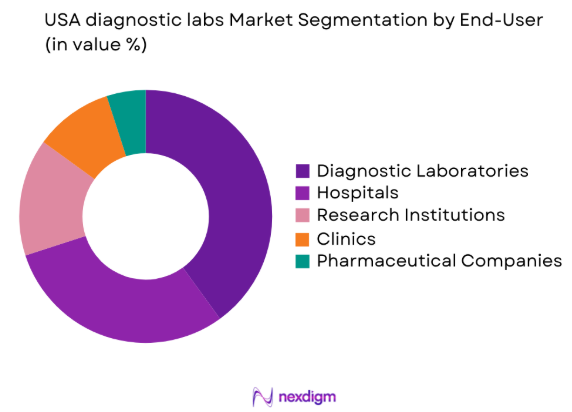
By End-User Segment
The USA diagnostic labs market is segmented by end-user into hospitals, diagnostic laboratories, research institutions, clinics, and pharmaceutical companies. Among these, diagnostic laboratories hold a dominant market share, primarily due to the increasing reliance on specialized diagnostic tests and their high throughput capabilities. As diagnostic labs continue to expand their service offerings, including advanced molecular and genetic testing, they become central players in providing high-quality diagnostic services, thereby increasing their market share.
Competitive Landscape
The USA diagnostic labs market is highly competitive, with numerous global players and regional providers actively involved. The market has witnessed consolidation due to the increasing demand for comprehensive diagnostic services and high-tech diagnostic equipment. Major players dominate the market, with their extensive networks of diagnostic centers and laboratories, and their ability to offer cutting-edge technologies such as molecular diagnostics and automation in testing processes. The market is also influenced by large-scale collaborations and acquisitions aimed at expanding technological capabilities and geographic presence.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD) | Additional Parameter |
| Abbott Laboratories | 1888 | Chicago, Illinois | ~ | ~ | ~ | ~ | ~ |
| Siemens Healthineers | 1847 | Erlangen, Germany | ~ | ~ | ~ | ~ | ~ |
| Roche Diagnostics | 1896 | Basel, Switzerland | ~ | ~ | ~ | ~ | ~ |
| Thermo Fisher Scientific | 1956 | Waltham, Massachusetts | ~ | ~ | ~ | ~ | ~ |
| Danaher Corporation | 1969 | Washington, D.C. | ~ | ~ | ~ | ~ | ~ |
USA diagnostic labs Market Analysis
Growth Drivers
Increased Prevalence of Chronic Diseases
The rise in chronic diseases such as diabetes, cancer, and cardiovascular diseases is a significant growth driver for the diagnostic labs market. With the growing number of patients requiring regular diagnostic tests for disease monitoring and early detection, the demand for diagnostic lab services is expected to rise. Additionally, advancements in diagnostic technology, such as molecular diagnostics and non-invasive testing, enable more accurate and timely detection, making these tests increasingly popular among healthcare providers. As healthcare systems shift toward proactive and preventive care, diagnostic labs play a crucial role in detecting early signs of chronic conditions, which is expected to further boost their demand.
Technological Advancements in Diagnostics
Rapid advancements in diagnostic technologies are reshaping the market, fueling growth in the diagnostic labs sector. Key innovations such as the integration of artificial intelligence (AI) in diagnostic tools, the adoption of molecular diagnostics, and automation of lab processes are improving testing accuracy, speed, and efficiency. These technological advancements enable labs to offer more specialized services, leading to better healthcare outcomes. As patient demand grows for faster, more accurate results, labs investing in cutting-edge technologies like AI-based imaging and genomics are positioned for significant market expansion. By embracing these next-generation solutions, diagnostic labs can stay competitive and meet the increasing expectations for precision and speed in healthcare diagnostics.
Market Challenges
High Operational Costs
A major challenge for diagnostic labs is the high operational costs associated with advanced diagnostic equipment. The purchase, installation, and upkeep of systems, especially those incorporating cutting-edge technologies like molecular diagnostics and AI, can be financially burdensome. Smaller labs, in particular, may find it difficult to afford the latest technologies, which can hinder their ability to compete with larger, well-funded competitors. Additionally, as diagnostic tests become more complex, labor costs for skilled technicians and specialists are increasing, further straining budgets. These rising costs make it challenging for diagnostic labs to maintain profitability while meeting the demand for accurate and timely results, especially for those lacking the financial resources to invest in advanced technologies.
Regulatory and Compliance Issues
Diagnostic labs are subject to strict regulations from authorities like the Food and Drug Administration (FDA) in the USA to ensure the accuracy and reliability of their tests. Compliance with these regulations demands significant investment in meeting certification standards and adhering to evolving regulatory requirements. Failure to comply with these standards can result in costly fines, legal action, or even the suspension of operations. As the healthcare industry evolves, keeping up with regulatory changes and maintaining necessary certifications becomes increasingly challenging for diagnostic labs. The pressure to stay compliant while ensuring operational efficiency adds a layer of complexity, requiring constant vigilance and resources to avoid disruptions and maintain the trust of healthcare providers and patients.
Opportunities
Expansion of Point-of-Care Diagnostics
The growing demand for point-of-care diagnostics offers a significant opportunity for diagnostic labs to expand their services. Point-of-care testing, which delivers rapid results at the patient’s location, is gaining popularity due to its ability to facilitate faster decision-making and improve patient outcomes. Healthcare providers are increasingly adopting this model, and diagnostic labs that offer portable, user-friendly, and precise point-of-care solutions are well-positioned to benefit from the rising demand. This trend is especially notable in home-based diagnostics, where patients can conduct certain tests themselves and send samples to labs for analysis. As patient preferences shift towards convenience and immediacy, diagnostic labs investing in these technologies will be able to capture a larger market share in the evolving healthcare landscape.
Adoption of Artificial Intelligence and Automation
The integration of artificial intelligence (AI) and automation in diagnostic labs presents significant growth potential. AI-powered tools enhance diagnostic accuracy by quickly analyzing complex medical data with greater precision than traditional methods. Automation improves lab efficiency by reducing human error, increasing throughput, and cutting operational costs. As diagnostic labs invest in these technologies, they can provide higher-quality services and streamline operations. This technological advancement not only enhances diagnostic capabilities but also gives labs a competitive edge in a rapidly evolving healthcare market. With the increasing demand for faster and more accurate results, AI and automation are becoming critical for success in the diagnostic industry.
Future Outlook
The future outlook for the USA diagnostic labs market is promising, with expected growth driven by advances in technology and an increasing focus on preventative care. The integration of AI, machine learning, and molecular diagnostics will likely shape the next phase of market evolution, leading to more personalized and accurate diagnostics. Additionally, increased government support for healthcare infrastructure, along with rising demand for home-based diagnostics and point-of-care testing, will drive further market expansion. Over the next few years, the market is expected to experience steady growth, as diagnostic labs adapt to meet the needs of both patients and healthcare providers in a rapidly changing healthcare landscape.
Major Players
- Abbott Laboratories
- Siemens Healthineers
- Roche Diagnostics
- ThermoFisher Scientific
- Danaher Corporation
- Becton,Dickinsonand Company
- Mindray Medical
- Sysmex Corporation
- PerkinElmer
- bioMérieux
- Hitachi Chemical Diagnostics
- Ortho Clinical Diagnostics
- Agilent Technologies
- Hologic, Inc.
- GE Healthcare
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Healthcare providers
- Digital health technology companies
- Pharmaceutical companies
- Medical device manufacturers
- Insurance providers
- Hospitals and clinics
Research Methodology
Step 1: Identification of Key Variables
The first step in our research methodology is to identify the key variables that drive the diagnostic labs market, including technological advancements, market demand, regulatory factors, and competitive dynamics.
Step 2: Market Analysis and Construction
The next step involves constructing the market model by analyzing the historical data, industry trends, and other relevant factors to determine the current market size and growth trajectory.
Step 3: Hypothesis Validation and Expert Consultation
We validate our hypotheses by consulting with industry experts, stakeholders, and market participants to refine our insights and ensure the accuracy of our assumptions.
Step 4: Research Synthesis and Final Output
Finally, the research is synthesized into a comprehensive report, integrating the findings from all previous steps, to deliver a clear and actionable market analysis.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Increase in Prevalence of Chronic Diseases
Technological Advancements in Diagnostic Tools
Rising Healthcare Expenditure
Shift Towards Preventative Healthcare
Growth in Personalized Medicine - Market Challenges
High Initial Setup Costs
Lack of Skilled Labor
Regulatory and Compliance Challenges
Infrastructure Constraints in Rural Areas
Fragmented Market and Lack of Standardization - Market Opportunities
Expansion of Point-of-care Diagnostics
Adoption of Artificial Intelligence in Diagnostics
Growth in Home-based Diagnostic Testing - Trends
Increase in AI and Machine Learning Integration
Shift Towards Molecular Diagnostics
Surge in Telemedicine and Remote Diagnostics - Government Regulations
- SWOT Analysis of Key Competitors
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Clinical Chemistry Systems
Hematology Systems
Microbiology Systems
Immunoassay Systems
Molecular Diagnostics Systems - By Platform Type (In Value%)
Laboratory-based Platforms
Point-of-care Platforms
Home-based Platforms
Mobile Diagnostic Platforms
Integrated Platforms - By Fitment Type (In Value%)
On-premise Solutions
Cloud-based Solutions
Hybrid Solutions
Modular Solutions
Integrated Solutions - By End User Segment (In Value%)
Hospitals
Clinics
Diagnostic Laboratories
Research Institutions
Pharmaceutical Companies - By Procurement Channel (In Value%)
Direct Procurement
Government Tenders
Private Sector Procurement
Online Bidding Platforms
Third-party Distributors
- Market Share Analysis
- Cross Comparison Parameters (System Type, Platform Type, Procurement Channel, EndUser Segment, Fitment Type, Diagnostic Technology, Market Reach, Regulatory Compliance, Adoption Rate, Geographic Coverage)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Abbott Laboratories
Roche Diagnostics
Siemens Healthineers
Thermo Fisher Scientific
Danaher Corporation
Becton, Dickinson and Company
Mindray Medical
Sysmex Corporation
PerkinElmer
bioMérieux
Hitachi Chemical Diagnostics
Ortho Clinical Diagnostics
Agilent Technologies
Hologic, Inc.
GE Healthcare
- Hospitals Increasing Demand for Advanced Diagnostics
- Diagnostic Laboratories’ Expansion of Services
- Research Institutions Investing in Cutting-edge Technology
- Pharmaceutical Companies’ Role in Diagnostics
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035