Market Overview
The USA EEG Machines Equipment Market is valued at USD ~ billion, driven by growing demand for advanced neurological diagnostic tools, increasing prevalence of neurological disorders like epilepsy, and rising awareness regarding brain health. The market is propelled by continuous advancements in EEG technology, including wireless and portable devices, which offer better flexibility and ease of use. The rising number of hospitals, diagnostic centers, and neurological clinics is also fueling the demand for EEG machines across the USA, ensuring long-term market growth. These factors, combined with ongoing research and innovation, contribute significantly to the market’s expansion.
The USA remains the dominant market for EEG machines, particularly in major urban centers such as New York, Los Angeles, and Chicago. These cities are home to a large number of medical facilities, including hospitals, neurological research centers, and diagnostic clinics, which are the key consumers of EEG equipment. The strong healthcare infrastructure, coupled with a high rate of neurological disorders, drives the demand for EEG devices. Additionally, these cities benefit from being innovation hubs where advanced medical technologies are rapidly adopted.
Market Segmentation
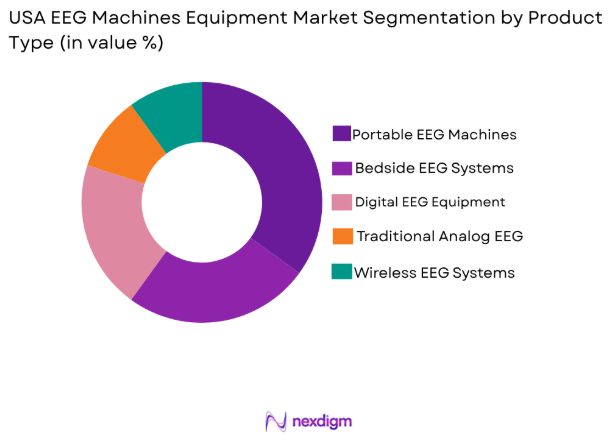
By Product Type
The USA EEG Machines Equipment market is segmented by product type into portable EEG machines, bedside EEG systems, digital EEG equipment, traditional analog EEG equipment, and wireless EEG systems. Portable EEG machines have the largest market share in the USA EEG market due to the growing preference for mobility and flexibility in medical diagnostics. Portable systems enable healthcare providers to conduct EEG tests at patients’ homes, during transport, or in ambulatory settings. This convenience, along with the decreasing costs of portable devices, makes them increasingly popular, especially among healthcare facilities offering at-home care and outpatient services.
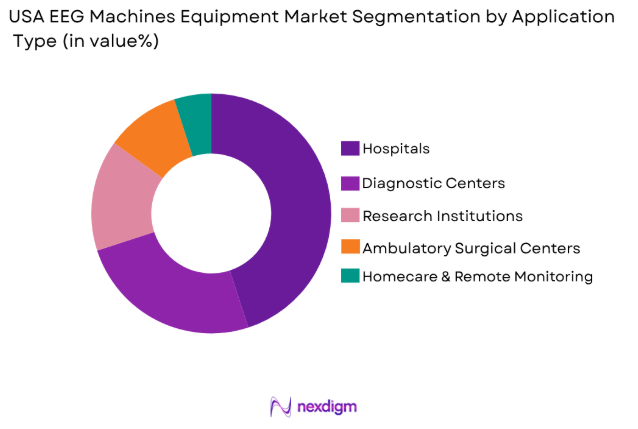
By Application Type
The USA EEG Machines Equipment market is also segmented by application into hospitals, diagnostic centers, research institutions, ambulatory surgical centers, and homecare and remote monitoring. Hospitals hold the dominant share of the market, accounting for a significant percentage of EEG machine purchases. The high volume of patients requiring EEG tests for neurological conditions such as epilepsy, sleep disorders, and brain injuries drives the demand within these facilities. Hospitals are equipped with advanced EEG systems to offer comprehensive patient care and accurate diagnosis, making them a primary segment in the EEG market.
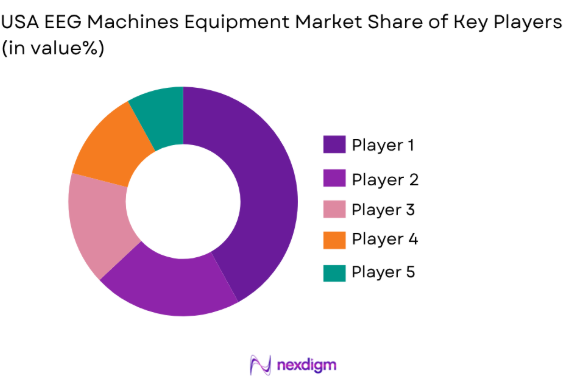
Competitive Landscape
The USA EEG Machines Equipment market is competitive, with key players dominating the landscape due to their advanced technological offerings, extensive distribution networks, and strong market presence. Leading companies such as Philips Healthcare, Medtronic, and Nihon Kohden Corporation hold significant market shares. These companies are at the forefront of EEG innovation, introducing wireless and portable systems to cater to the increasing demand for patient-centric care. The market sees a mix of global players and specialized firms focusing on high-end neurological diagnostic equipment.
| Company | Establishment Year | Headquarters | Product Offerings | Market Share | Technology | Distribution Network | Annual Revenue | Customer Base | Regional Reach |
| Philips Healthcare | 1891 | Amsterdam, Netherlands | EEG Machines, Diagnostic Equipment | 25% | ~ | ~ | ~ | ~ | ~ |
| Medtronic | 1949 | Dublin, Ireland | EEG Machines, Neurosurgery Equipment | ~ | ~ | ~ | ~ | ~ | ~ |
| Nihon Kohden Corporation | 1951 | Tokyo, Japan | EEG Systems, Electrocardiograph | ~ | ~ | ~ | ~ | ~ | ~ |
| Cadwell Laboratories | 1979 | Kennewick, USA | EEG Machines, Polysomnography Devices | ~ | ~ | ~ | ~ | ~ | ~ |
| BioSemi | 1996 | Amsterdam, Netherlands | EEG Amplifiers, Wireless EEG | ~ | ~ | ~ | ~ | ~ | ~ |
USA EEG Machines Equipment Market Analysis
Growth Drivers
Increasing Prevalence of Neurological Disorders
The U.S. EEG machines equipment market is supported first by the sheer clinical volume of neurological conditions that require brain-function monitoring, seizure confirmation, differential diagnosis, ICU follow-up, or longitudinal review. CDC states that about 2.9 million U.S. adults had active epilepsy in 2021, and 456,000 U.S. children had active epilepsy in 2022. CDC also reports that the United States records more than 795,000 strokes each year, with 610,000 counted as first or new strokes. On the dementia side, the National Institute on Aging reported 6.9 million Americans living with Alzheimer’s in 2024. These are not fringe-use cases for EEG; they are core demand pools for routine EEG, ambulatory EEG, continuous EEG, seizure detection workflows, and neurocritical care monitoring. The underlying macro base is also large enough to sustain equipment deployment: the World Bank lists the U.S. population at 340,110,988 in 2024, with GDP at $28.75 trillion and GDP per capita at $84,534. In parallel, CMS reports national health expenditure of $5.3 trillion in 2024, including $1,118.0 billion in Medicare spending and $931.7 billion in Medicaid spending. For the EEG equipment market, this matters because neurological disease burden is rising inside a healthcare system that still has scale, reimbursement depth, and institutional capacity to keep investing in diagnostic neurotechnology for emergency, inpatient, outpatient, and aging-care settings.
Advancements in EEG Technology
Technology improvement is no longer a general medtech narrative in U.S. EEG; it is visible in actual device clearances and in federal neurotechnology funding. In 2024, FDA cleared the REMI-AI Rapid Detection Module for automated seizure-event analysis within the REMI Remote EEG Monitoring System, and the clearance states that the module performs near-real-time analysis of EEG data in non-ICU care settings. In September 2024, FDA cleared encevis for the review, monitoring, and analysis of scalp-electrode EEG recordings to aid neurologists. In April 2025, FDA cleared autoSCORE 2.0, a decision-support software that assists trained professionals in reviewing human scalp EEG recordings from patients aged 3 months or older for EEG studies of under 4 hours. These developments show a clear move from simple acquisition hardware toward integrated review, detection, and decision-support stacks, which expands replacement demand and raises the value of software-enabled EEG platforms. Federal research infrastructure reinforces this direction: NIH’s BRAIN Initiative had $402 million appropriated in FY2024, and NIH continued issuing 2024 funding opportunities specifically for neural recording and modulation tools. The macro backdrop also remains supportive. CMS recorded $5.3 trillion in national health spending in 2024, while the World Bank lists U.S. GDP at $28.75 trillion in the same year. That combination of regulated product progress, decision-support capability, and sustained federal neurotechnology funding supports higher clinical confidence in modern EEG systems and strengthens the installed-base upgrade cycle across hospitals, neurology practices, epilepsy centers, and acute monitoring environments.
Market Challenges
High Equipment Costs
High equipment costs remain a real market constraint in the U.S. EEG machines segment even without citing device price tags, because EEG procurement sits inside a broader hospital cost structure that is already heavy in labor, benefits, compliance, and capital allocation. BLS reported that total employer compensation costs in U.S. hospitals averaged $67.64 per hour worked in June 2024, including $44.73 in wages and salaries and $22.90 in benefits. At the same time, CMS reported total national health expenditure of $5.3 trillion in 2024, with private health insurance spending at $1,644.6 billion, Medicare at $1,118.0 billion, Medicaid at $931.7 billion, and out-of-pocket spending at $556.6 billion. Those numbers show that providers and payers are already operating inside a large but highly committed expenditure base, so capital purchases such as EEG consoles, amplifiers, software modules, cloud archiving, disposable components, and service contracts compete against staffing, inpatient throughput, and general hospital operating needs. FDA’s product classification database also shows that a full-montage standard electroencephalograph acquires, displays, stores, and archives EEG signals using 16 or more electrodes, which reflects the system complexity involved in standard clinical setups. The macro context does not remove that pressure: the World Bank lists U.S. GDP per capita at $84,534 in 2024, but that same high-income environment also carries high labor and operating baselines for providers. For smaller hospitals, rural systems, independent neurology centers, and centers with low EEG volumes, the challenge is therefore not demand absence but capital prioritization under a cost-heavy care-delivery model.
Regulatory Barriers
Regulatory burden is another structural restraint because EEG equipment in the U.S. is not a lightly supervised category. FDA classifies electroencephalographs under 21 CFR 882.1400 as Class II devices, and the same classification page distinguishes multiple regulated subtypes, including amplitude-integrated electroencephalographs and reduced-montage standard electroencephalographs. FDA also defines a full-montage standard electroencephalograph as a system using 16 or more electrodes, showing that device claims are tied to precise hardware and signal-acquisition specifications. For manufacturers, the hurdle is not only submission itself but evidence preparation, software validation, cybersecurity documentation, performance testing, and predicate comparisons. Under MDUFA V, the shared FDA-industry goal for 510(k) submissions received in FY2024 is 124 calendar days to decision. That timeline is meaningful for EEG vendors because newer systems increasingly combine hardware, cloud connectivity, AI analysis, and event-detection software, all of which raise the documentation burden. The FDA letters and summaries issued in 2024 and 2025 for products such as REMI-AI RDM, QUEX ED / QUEX S, encevis, and auto SCORE 2.0 show that modern EEG products are being cleared, but they are being cleared inside a tightly specified Class II framework. For the market, that slows smaller entrants, lengthens commercialization cycles, and raises the threshold for software-led innovation. Even in a macro environment where the World Bank records U.S. GDP of $28.75 trillion and CMS records $5.3 trillion in health expenditure in 2024, regulatory time and validation cost still shape how fast new EEG equipment can reach scale.
Opportunities
Technological Advancements in Wireless EEG Machines
Wireless EEG is one of the clearest opportunities because the U.S. market is moving from cart-based, wired neurodiagnostics toward mobile, easier-to-deploy systems that fit emergency, bedside, ward, and distributed-care workflows. FDA activity in 2024 and 2025 supports this directly. In November 2024, FDA cleared the Natus BrainWatch system, describing it as a mobile EEG device with a disposable headpiece containing 10 passive electrodes and a touchscreen tablet interface. In November 2024, FDA also cleared a Beacon Biosignals device that includes 6 EEG electrodes and 1 reference electrode. In October 2024, FDA cleared QUEX ED / QUEX S, whose summary describes a portable EEG monitor that can display EEG data in real time and transfer recording files to the cloud. These are commercially relevant signals for the U.S. EEG machines market because wireless and reduced-montage systems lower setup friction, expand bedside use, and support point-of-care neurology outside the traditional EEG lab. They also align with broader care decentralization. CMS reported 365 approved facilities across 138 health systems in 37 states for Acute Hospital Care at Home resources, and CMS’s CY 2024 Physician Fee Schedule finalized broader care-management support around remote physiologic monitoring in RHCs and FQHCs beginning January 1, 2024. With the World Bank placing U.S. GDP per capita at $84,534 in 2024 and CMS recording $5.3 trillion in national health expenditure, the operating environment can support premium clinical workflows when those workflows reduce nurse time, speed triage, or extend monitoring reach. That makes wireless EEG a strong forward-looking opportunity grounded in current adoption infrastructure rather than speculative future numbers.
Increasing Demand for Portable and Non-invasive Devices
Demand for portable and non-invasive EEG devices is likely to remain structurally strong because U.S. neurology demand is spreading across aging care, emergency response, ambulatory diagnostics, sleep-related pathways, and home-oriented care models. The aging base alone supports that direction: the U.S. Census Bureau reported that the population aged 65 years and older rose to 61.2 million from 2023 to 2024. NIH also reported 6.9 million Americans living with Alzheimer’s in 2024, while CDC reported 2.9 million adults with active epilepsy and 456,000 children with active epilepsy. These patient groups benefit from technologies that reduce preparation burden, avoid invasive procedures, and allow faster brain-function capture in non-specialist settings. Current delivery models are already shifting in that direction. CMS reported that its Acute Hospital Care at Home study covered 332 participating hospitals across 38 states from November 2020 through July 2024, and CMS finalized policy in the CY 2024 Medicare Physician Fee Schedule to include remote physiologic monitoring in general care management for RHCs and FQHCs. Portable EEG fits this infrastructure better than fixed-room systems because it is easier to deploy in community sites, step-down units, memory-care pathways, and bedside observation environments. FDA clearances during 2024 reinforce this demand pattern: BrainWatch, QUEX, REMI-AI, and related systems all point toward smaller-footprint, clinically actionable EEG. The macro base remains supportive, with World Bank data showing a 2024 U.S. population of 340,110,988 and GDP of $28.75 trillion. In that context, non-invasive and portable EEG is not just a product preference; it matches the way U.S. neurological care delivery is being reorganized.
Future Outlook
Over the next five years, the USA EEG Machines Equipment market is expected to show significant growth. Key drivers include continuous advancements in EEG technology, including wireless and portable devices, and the increasing demand for early diagnosis and continuous monitoring of neurological disorders. The rise in the aging population, alongside the growing awareness of brain health and neurological conditions, will further contribute to market expansion. Furthermore, as healthcare shifts towards homecare services and telemedicine, the need for more flexible, portable, and remote monitoring EEG devices will intensify, creating opportunities for companies to innovate and cater to new consumer needs.
Major Players
- Philips Healthcare
- Medtronic
- Nihon Kohden Corporation
- Cadwell Laboratories
- BioSemi
- Natus Medical Incorporated
- Compumedics
- Brain Products
- NeuroWave Systems
- Emotiv Inc.
- Advanced Brain Monitoring
- EEG Devices Inc.
- GE Healthcare
- General Electric Healthcare
- Brain Science International
Key Target Audience
- Investments and Venture Capitalist Firms
- Government and Regulatory Bodies (FDA, NIH)
- Healthcare Providers
- Neurodiagnostic Centers
- Medical Equipment Distributors
- Private Healthcare Networks
- Hospitals and Clinics
- Medical Research Institutions
Research Methodology
Step 1: Identification of Key Variables
The research begins by identifying the key variables that influence the USA EEG Machines Equipment Market. This includes identifying the main stakeholders such as healthcare providers, EEG manufacturers, and end users. Through desk research and analysis of secondary data sources, we define the parameters critical to understanding market dynamics.
Step 2: Market Analysis and Construction
In this step, historical data from credible sources such as industry reports, financial documents, and government publications is analyzed to estimate the market size and identify growth trends. The focus is on evaluating the market penetration of EEG devices in various healthcare settings and determining how this influences future market demand.
Step 3: Hypothesis Validation and Expert Consultation
Developed hypotheses regarding market trends and growth drivers are tested through consultations with industry experts. Telephone interviews, surveys, and expert panels will provide insights into the challenges and opportunities that industry participants foresee, validating the data gathered in previous steps.
Step 4: Research Synthesis and Final Output
The final phase synthesizes the findings into a comprehensive analysis. Engaging with key industry players such as manufacturers and distributors provides deeper insights into operational data, customer preferences, and emerging trends. This ensures the report is accurate and reflects the most up-to-date market dynamics.
- Executive Summary
- Research Methodology (Market Definitions and Assumptions, Abbreviations, Market Sizing Approach, Consolidated Research Approach, Understanding Market Potential Through In-Depth Industry Interviews, Primary Research Approach, Limitations and Future Conclusions)
- Definition and Scope
- Market Genesis and Evolution
- Historical Market Timeline of Key Players
- Business Cycle and Key Factors Impacting Growth
- Supply Chain and Value Chain Analysis
- Technological Advancements in EEG Equipment
- Growth Drivers
Increasing Prevalence of Neurological Disorders
Advancements in EEG Technology
Market Challenges
High Equipment Costs
Regulatory Barriers
Opportunities
Technological Advancements in Wireless EEG Machines
Increasing Demand for Portable and Non-invasive Devices
Trends
Integration of EEG with Artificial Intelligence (AI)
Growing Use of Cloud-Based EEG Data Solutions
Government Regulations
FDA Approval Process for EEG Equipment
Quality Assurance Standards for Medical Devices
Import Tariffs and Healthcare Policies - SWOT Analysis
- Porter’s Five Forces
- Competition Ecosystem
- By Value, 2020-2025
- By Volume, 2020-2025
- By Average Price, 2020-2025
- By Product Type (In Value %)
Portable EEG Machines
Bedside EEG Systems
Digital EEG Equipment
Traditional Analog EEG Equipment
Wireless EEG Systems - By Application (In Value %)
Hospitals
Diagnostic Centers
Research Institutions
Ambulatory Surgical Centers
Homecare and Remote Monitoring - By End-User (In Value %)
Healthcare Providers
Research and Educational Institutions
Clinical Research Organizations
Patients/Individuals
Neurodiagnostic Centers - By Region (In Value %)
Northeast USA Midwest USA
South USA
West USA - By Distribution Channel (In Value %)
Direct Sales
Online Retail
Healthcare Distributors
Retail Stores
Medical Equipment Resellers
- Market Share of Major Players by Value/Volume
- Cross Comparison Parameters (Company Overview, Business Strategies, Recent Developments, Strength, Weakness, Organizational Structure, Revenues, Revenue by Product Type, Number of Distribution Channels, Number of Dealers, Distribution Network, Production Plant, Manufacturing Capacity, Unique Value Offerings, and others)
- SWOT Analysis of Major Players
- Pricing Analysis Basis Product Categories for Major Players
- Detailed Profiles of Major Companies
Philips Healthcare
Nihon Kohden Corporation
General Electric Healthcare
Medtronic PLC
Siemens Healthineers
Cadwell Laboratories
Compumedics Limited
EEG Devices Inc.
Brain Science International
BioSemi
NeuroWave Systems
Natus Medical Incorporated
Brain Products
Emotiv Inc.
Advanced Brain Monitoring
- Market Demand and Utilization
- Purchasing Power and Budget Allocations
- Regulatory and Compliance Requirements
- Needs, Desires, and Pain Point Analysis
- Decision-Making Process
- By Value, 2026-2035
- By Volume, 2026-2035
- By Average Price, 2026-2035