Market Overview
The USA Epidural Analgesia Systems Market is expected to reach a market size of USD ~ billion, driven by the increasing adoption of epidural analgesia systems in pain management, especially in surgical and labor settings. The market’s growth is attributed to the rise in surgeries and obstetric procedures, along with technological advancements in epidural delivery systems, making them safer and more efficient. This growth is also propelled by the aging population, which demands more effective pain management solutions for chronic conditions.
In terms of geographic dominance, the USA stands out as a leader in the market, largely due to its advanced healthcare infrastructure and high healthcare expenditure. States like California, Texas, and Florida contribute significantly to the market size, with leading hospitals and surgical centers offering high-end epidural analgesia solutions. Urban areas such as New York and Los Angeles, where a large portion of the population undergoes surgeries, are key contributors to the demand for these systems.
Market Segmentation
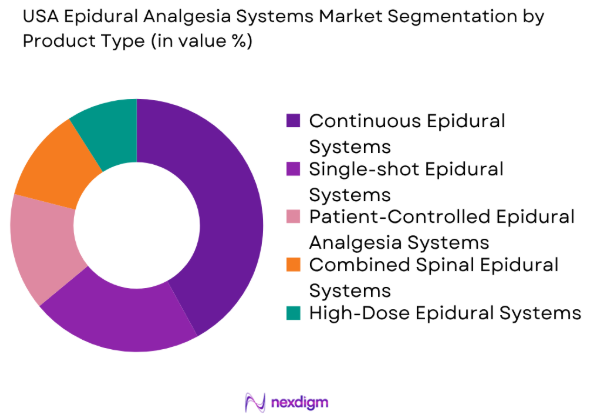
By Product Type
The USA Epidural Analgesia Systems market is segmented by product type into Continuous Epidural Systems, Single-shot Epidural Systems, Patient-Controlled Epidural Analgesia (PCEA) Systems, Combined Spinal Epidural Systems, and High-Dose Epidural Systems. Recently, Continuous Epidural Systems have had a dominant market share due to factors such as their ability to provide long-term pain relief for patients undergoing major surgeries. Continuous systems allow for consistent administration of medication, reducing the need for repeated doses and enhancing patient comfort, making them particularly popular in hospitals and surgical centers.
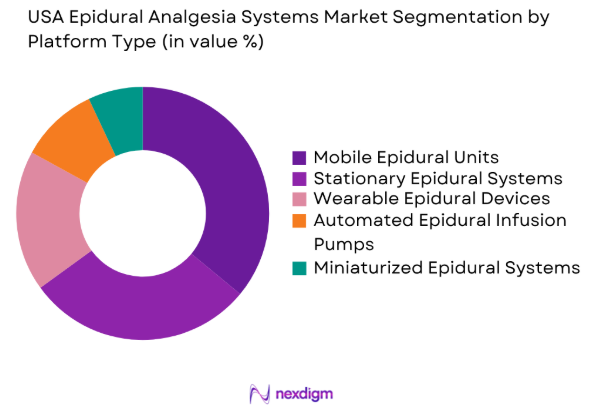
By Platform Type
The USA Epidural Analgesia Systems market is also segmented by platform type into Mobile Epidural Units, Stationary Epidural Systems, Wearable Epidural Devices, Automated Epidural Infusion Pumps, and Miniaturized Epidural Systems. Mobile Epidural Units currently hold the largest market share, as their flexibility and ability to be used in a variety of settings—whether in operating rooms, recovery units, or even home care—make them highly appealing to healthcare providers and patients alike. The growing demand for outpatient procedures and home care solutions has further boosted the popularity of mobile units, which can be easily transported and set up.
Competitive Landscape
he competitive landscape in the USA Epidural Analgesia Systems Market is characterized by both large multinational companies and smaller specialized manufacturers. Market consolidation is evident as major players continue to expand their product offerings and improve technological innovations. The influence of large companies is significant, especially in terms of research and development investments, which has propelled advancements in the accuracy, safety, and efficiency of epidural analgesia systems. Companies are increasingly focusing on collaborations with hospitals and clinics to offer more personalized and integrated pain management solutions.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Additional Parameter |
| Baxter International Inc. | 1931 | Deerfield, Illinois | ~ | ~ | ~ | ~ | ~ |
| Medtronic Plc | 1949 | Dublin, Ireland | ~ | ~ | ~ | ~ | ~ |
| B. Braun Melsungen AG | 1839 | Melsungen, Germany | ~ | ~ | ~ | ~ | ~ |
| Smiths Medical | 1851 | London, UK | ~ | ~ | ~ | ~ | ~ |
| Abbott Laboratories | 1888 | Chicago, Illinois | ~ | ~ | ~ | ~ | ~ |
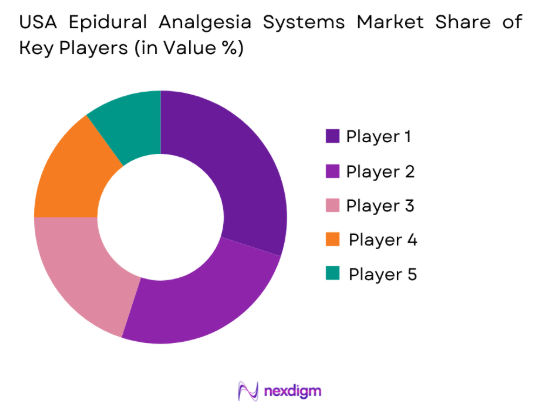
USA Epidural Analgesia Systems Market Analysis
Growth Drivers
Technological Innovation
Technological innovation plays a crucial role in driving the growth of the USA Epidural Analgesia Systems market. Advancements such as automated dose adjustments, enhanced patient-controlled epidural analgesia systems, and miniaturized devices are improving system efficiency and safety. These innovations enable better adaptability to diverse patient needs, making the systems increasingly popular in hospitals and clinics, particularly in surgical and maternity care. Additionally, the rising demand for outpatient and homecare solutions is boosting the adoption of portable and wearable epidural systems. As these systems evolve to become more user-friendly and efficient, they are expected to enhance patient outcomes and further expand their presence in healthcare settings.
Aging Population
The aging population is a significant growth driver for the USA Epidural Analgesia Systems market. As people age, the prevalence of chronic pain conditions, such as osteoarthritis, back pain, and nerve disorders, rises, increasing the demand for effective pain management solutions. Epidural analgesia systems are frequently used for managing pain during surgeries and for long-term relief, particularly for elderly patients who may have limited tolerance for other pain relief methods. As the elderly population requiring surgical procedures and chronic pain management grows, the demand for epidural analgesia systems continues to rise, further accelerating market growth. This trend is expected to persist, particularly in regions with higher concentrations of older populations.
Market Challenges
High Cost of Equipment
A significant challenge for the USA Epidural Analgesia Systems market is the high initial cost of equipment. The upfront investment required for advanced systems, such as infusion pumps, automated devices, and necessary infrastructure, can be prohibitively expensive for smaller hospitals, clinics, and outpatient centers. This cost barrier limits widespread adoption, especially in regions with budget constraints. While the long-term benefits of improved pain management and patient satisfaction are evident, the high capital expenditure required for purchasing, maintaining, and upgrading these systems remains a major hurdle for many healthcare providers. As a result, this financial challenge restricts the growth potential of the market, particularly in more cost-sensitive areas.
Regulatory Hurdles
Another challenge for the USA Epidural Analgesia Systems market is the complex and time-consuming regulatory approval process. Manufacturers must navigate strict FDA regulations to bring new products to market, which can delay the introduction of innovations. The approval process for medical devices in the USA often involves extensive testing, documentation, and clinical trials, all of which can significantly extend product development timelines. Additionally, regulatory changes or stricter safety standards can complicate the approval process, further delaying market entry. For companies in the market, meeting these regulatory requirements is both time-consuming and costly. This creates barriers for quick innovation and limits the availability of newer, more efficient products, slowing overall market growth and product adoption.
Opportunities
Homecare and Outpatient Services
A key opportunity in the USA Epidural Analgesia Systems market is the growing demand for homecare and outpatient services. As patients increasingly prefer cost-effective outpatient procedures and faster recoveries, the need for portable, wearable epidural analgesia systems for non-hospital use is rising. More homecare services are being offered, and patients are being discharged earlier after surgery, often continuing their pain management at home. This shift creates a significant opportunity for manufacturers to develop compact, user-friendly systems that meet the needs of patients requiring post-surgical pain relief outside of hospital settings. Additionally, evolving healthcare reimbursement policies, with more insurance companies covering homecare services, are expected to further support the growth of this market segment.
Emerging Markets
Another opportunity lies in the expansion of epidural analgesia systems into emerging markets. Many developing countries are improving their healthcare infrastructure and investing in advanced medical devices to meet the growing demand for quality healthcare. As awareness of effective pain management solutions like epidural analgesia systems increases in these regions, there is potential for significant market growth. Manufacturers can capitalize on this by offering affordable, high-quality systems that cater to the specific needs of emerging market populations, particularly in Asia-Pacific and Latin American countries. Partnerships with local distributors and healthcare providers could further facilitate the adoption of these systems in underserved regions.
Future Outlook
The USA Epidural Analgesia Systems market is expected to experience steady growth over the next few years, driven by technological advancements, an aging population, and increasing demand for effective pain management solutions. Innovations such as patient-controlled analgesia systems and automated drug delivery devices are expected to play a significant role in improving patient outcomes and boosting market growth. Regulatory support for advanced pain management technologies and rising healthcare expenditure will further contribute to market expansion. As more healthcare facilities embrace modern epidural systems, the market is likely to witness an increase in product demand, particularly in outpatient and homecare settings.
Major Players
- Baxter International Inc.
- Medtronic Plc
- B. Braun Melsungen AG
- Smiths Medical
- Abbott Laboratories
- Fresenius Kabi AG
- Teleflex Incorporated
- Johnson & Johnson
- Halyard Health
- Cerner Corporation
- Boston Scientific
- ICU Medical
- Envista Holdings Corporation
- Terumo Corporation
- Linde Group
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Hospitals and surgical centers
- Medical device distributors
- Pain management specialists
- Ambulatory surgical centers
- Healthcare consultants
- Homecare service providers
Research Methodology
Step 1: Identification of Key Variables
Identifying the key variables affecting the USA Epidural Analgesia Systems market, including product types, geographic regions, and regulatory frameworks.
Step 2: Market Analysis and Construction
Analyzing the market through primary and secondary research, including interviews with industry experts and data collection from reputable sources.
Step 3: Hypothesis Validation and Expert Consultation
Validating market hypotheses through consultations with healthcare professionals, industry leaders, and key stakeholders in the market.
Step 4: Research Synthesis and Final Output
Synthesizing the collected data and preparing the final report, including market projections, segmentation, and analysis.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Growing prevalence of chronic pain conditions
Advances in medical technology
Increasing number of surgeries - Market Challenges
High initial cost of equipment
Risk of complications and side effects
Regulatory hurdles and approval delays - Market Opportunities
Expansion in emerging markets
Integration of artificial intelligence
Demand for personalized pain management solutions - Trends
Adoption of minimally invasive procedures
Growth in outpatient and ambulatory care
Focus on patient-controlled analgesia (PCA) systems - Government Regulations
- SWOT Analysis of Key Competitors
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Continuous Epidural Systems
Single-shot Epidural Systems
Patient-Controlled Epidural Analgesia (PCEA) Systems
Combined Spinal Epidural Systems
High-Dose Epidural Systems - By Platform Type (In Value%)
Mobile Epidural Units
Stationary Epidural Systems
Wearable Epidural Devices
Automated Epidural Infusion Pumps
Miniaturized Epidural Systems - By Fitment Type (In Value%)
Intrathecal Fitment
Epidural Fitment
Combined Spinal-Epidural (CSE) Fitment
Peripheral Nerve Block Fitment
Implantable Epidural Systems - By End User Segment (In Value%)
Hospitals
Ambulatory Surgical Centers
Pain Management Clinics
Maternity & Obstetrics Units
Emergency Care Units - By Procurement Channel (In Value%)
Direct Sales to Hospitals
Distributor-based Procurement
Online Platforms and E-commerce
Government Contracts and Tenders
Private Medical Group Procurement
- Market Share Analysis
- Cross Comparison Parameters (System Type, Platform Type, End-User Segment, Procurement Channel, Growth Drivers)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Baxter International Inc.
Medtronic Plc
B. Braun Melsungen AG
Smiths Medical
Fresenius Kabi AG
Abbott Laboratories
Johnson & Johnson
Halyard Health
Teleflex Incorporated
Grünenthal GmbH
Cerner Corporation
Boston Scientific
ICU Medical
Envista Holdings Corporation
Terumo Corporation
- Hospitals (Acute care settings)
- Ambulatory surgical centers
- Maternity hospitals (Obstetrics units)
- Pain management clinics
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035