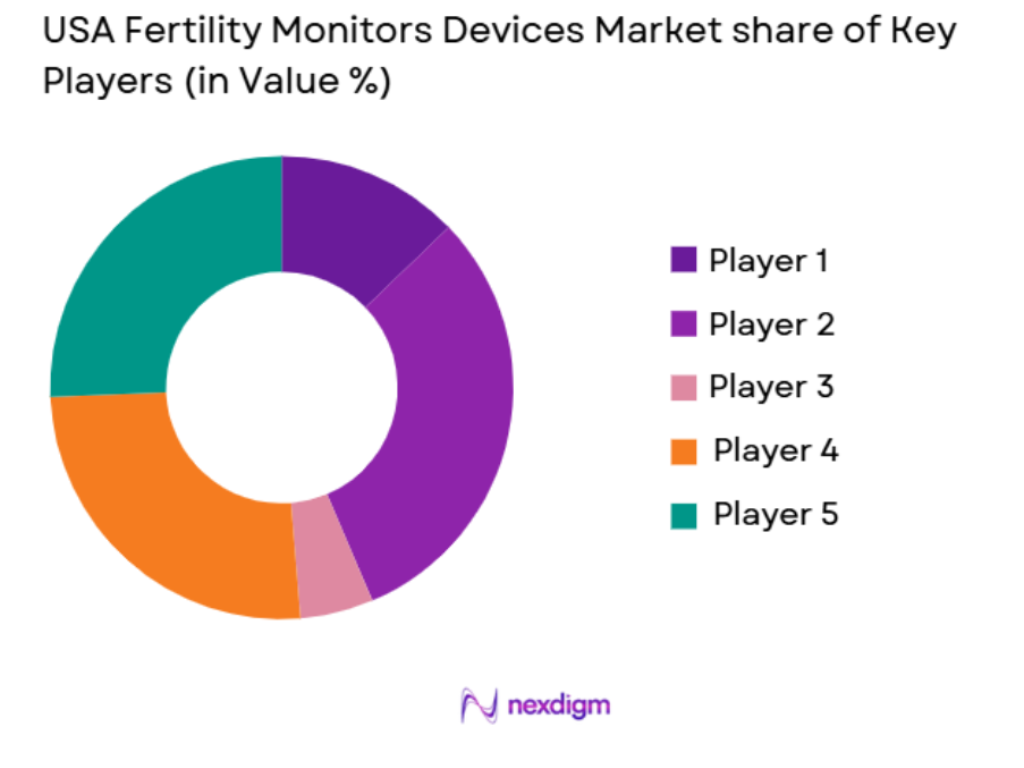
Market Overview
The USA Fertility Monitors Devices market is experiencing strong growth due to an increasing number of women seeking to monitor their fertility for family planning or health tracking purposes. In 2024, the market is valued at approximately USD ~ billion. This growth is primarily driven by advancements in wearable technology and mobile applications, allowing consumers to track their reproductive health with precision. Additionally, rising awareness about fertility issues and the increasing adoption of fertility-monitoring devices in consumer health segments contribute significantly to the market’s expansion.
The USA is the dominant region in the fertility monitor devices market, driven by a large consumer base with a growing focus on health and wellness, especially in metropolitan areas such as New York, Los Angeles, and Chicago. These cities represent a higher concentration of fertility clinics, healthcare providers, and a significant adoption of health technology. Moreover, the regulatory environment in the USA supports innovation in health tech, allowing for the rapid deployment and adoption of fertility monitor devices among consumers seeking accessible and reliable reproductive health solutions.
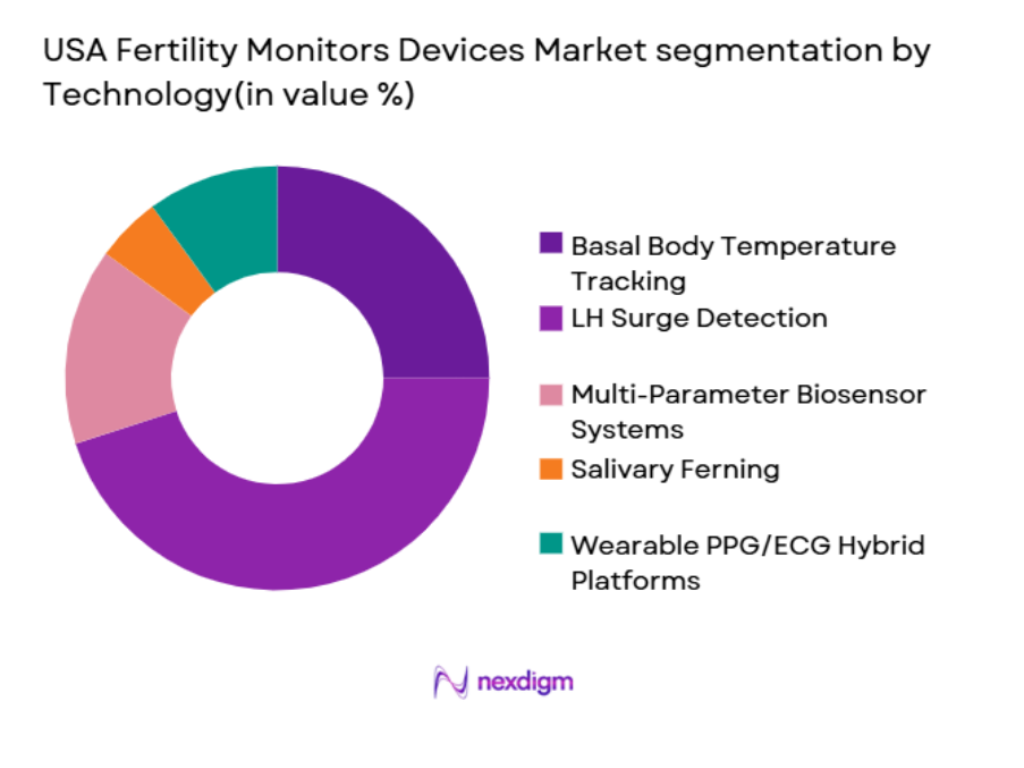
Market Segmentation
By Technology
The USA Fertility Monitors Devices market is segmented by technology into several types, including Basal Body Temperature (BBT) tracking, LH Surge detection, and wearable biosensor systems. Currently, LH Surge detection dominates the market. This is due to its direct relevance to ovulation prediction, which is the most commonly sought-after feature among consumers. Devices using this technology, such as ovulation predictor kits (OPKs), are widely available and trusted for their ease of use and cost-effectiveness. Brands like Clearblue and Ovia have significantly contributed to the market share through their robust product offerings and strong customer trust.
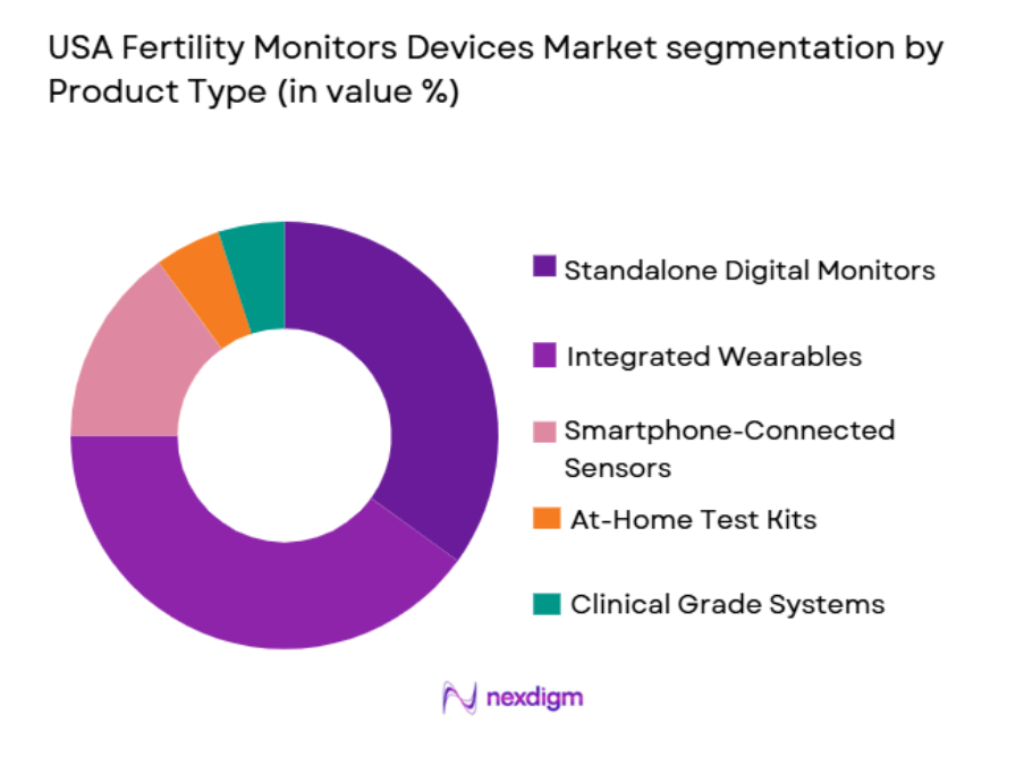
By Product type
The market for fertility monitors is also segmented by product type, including standalone digital monitors, integrated wearables, and smartphone-connected sensors. Integrated wearables, such as the Oura Ring or Fitbit, have gained significant traction due to their seamless integration with consumers’ lifestyles, making them easy to wear daily. These devices offer continuous monitoring and provide a more comprehensive health data overview, which appeals to the growing segment of health-conscious individuals seeking holistic wellness solutions.
Competitive Landscape
The USA Fertility Monitors Devices market is highly competitive, with both established medical device companies and new health tech startups entering the market. The landscape is characterized by the dominance of a few major players who continue to drive innovation and capture significant market share. Companies like Clearblue and Ovia, along with startups such as Mira and Natural Cycles, are among the key players shaping the market’s future. The competition primarily revolves around product innovation, accuracy, customer trust, and pricing strategies.
| Company | Establishment Year | Headquarters | Technology | Product Form Factor | Regulatory Approvals | Distribution Channels |
| Clearblue | 1985 | Swindon, UK | ~ | ~ | ~ | ~ |
| Ovia Health | 2012 | Boston, USA | ~ | ~ | ~ | ~ |
| Natural Cycles | 2013 | Stockholm, Sweden | ~ | ~ | ~ | ~ |
| Mira | 2014 | San Francisco, USA | ~ | ~ | ~ | ~ |
| Femometer | 2013 | Shenzhen, China | ~ | ~ | ~ | ~ |
USA Fertility Monitors Devices Market Analysis
Growth Drivers
Rising Infertility Prevalence
Infertility in the United States is on the rise, with approximately 12% of women of reproductive age facing challenges in getting pregnant or carrying a pregnancy to term, according to the Centers for Disease Control and Prevention (CDC). This increasing prevalence is driving demand for fertility monitoring devices. The trend is further reinforced by a rising number of women delaying childbirth due to personal and professional choices, leading to an increase in age-related infertility. In 2022, around 5.5 million women in the U.S. received infertility treatment, highlighting the growing market need for advanced fertility tracking solutions.
U.S. Women’s Health Awareness
The heightened awareness of women’s health is significantly propelling the market for fertility monitors. The U.S. has witnessed a growing interest in preventative health care, with many women now proactively managing their reproductive health. According to the U.S. Department of Health and Human Services, 61% of women aged 18-44 have reported using digital health tools to track their menstrual cycles and fertility. This trend is supported by the increasing popularity of wellness apps and wearable devices. Additionally, federal funding for women’s health initiatives, totaling over $2.7 billion in 2024, has helped raise awareness and provided a strong foundation for fertility monitor adoption.
Market Restraints
Reimbursement Gaps
While the fertility monitor market is expanding, reimbursement challenges remain a significant barrier. The majority of fertility tracking devices are not covered under federal insurance programs such as Medicaid, and only a small percentage of private insurers offer coverage for fertility monitoring tools. As of 2024, approximately 45% of fertility treatment costs in the U.S. are paid out-of-pocket by consumers, creating a financial barrier for many women. Without broad insurance reimbursement, the accessibility of these devices remains limited, especially for lower-income households. The absence of coverage further restricts the widespread adoption of advanced fertility tracking technologies.
Clinical Accuracy Concerns
Despite technological advancements, concerns about the clinical accuracy of fertility monitors remain a challenge. The U.S. Food and Drug Administration (FDA) has issued warnings regarding the accuracy of some over-the-counter fertility tracking devices, particularly those that claim to predict ovulation and pregnancy. As of 2024, only about 35% of fertility devices on the market have FDA approval, and this lack of regulatory oversight leads to trust issues among consumers. The gap in clinical validation is a significant barrier to greater adoption of fertility monitors, as many consumers and healthcare providers still prefer more traditional and medically validated methods.
Market Opportunities
AI‑Enabled Predictive Fertility Analytics
The market for AI-driven fertility analytics presents significant opportunities for growth, supported by advancements in artificial intelligence and machine learning technologies. Currently, AI-enabled fertility solutions are gaining traction among consumers, especially as they offer more personalized and accurate predictions based on a wider range of data inputs, such as basal body temperature, hormone levels, and real-time lifestyle factors. AI is already being used in applications like Ovia Health’s fertility tracker, which employs machine learning algorithms to predict ovulation cycles with greater accuracy. In the U.S., AI-driven solutions are expected to benefit from a surge in consumer demand for more personalized healthcare, with the U.S. government investing heavily in AI healthcare technologies, contributing over $1.5 billion to AI research in health tech by 2024.
Future Outlook
Over the next few years, the USA Fertility Monitors Devices market is expected to witness significant growth, fueled by the increasing demand for personalized health data and advancements in wearable technology. The adoption of fertility-monitoring devices is anticipated to rise due to the greater awareness of reproductive health and the continuous integration of artificial intelligence (AI) to provide more accurate predictions. Additionally, government initiatives to improve reproductive health accessibility will further bolster the growth of the market.
Major Players
- Clearblue
- Ovia Health
- Natural Cycles
- Mira
- Femometer
- Ava AG
- Kindara
- Clue
- Proov
- Dot Labs
- Pearl Fertility
- I-Health, Inc.
- The Stork by Rinovum
- Fairhaven Health
- FitTrack
Key Target Audience
- Investments and Venture Capitalist Firms
- Government and Regulatory Bodies (FDA, CDC, NCI)
- Fertility Clinics & IVF Centers
- OB/GYN Practitioners
- Medical Device Distributors
- Consumer Health Technology Companies
- E-commerce Platforms & Retailers
- Insurance Companies (Health Plans)
Research Methodology
Step 1: Identification of Key Variables
The first phase involves identifying the key market variables that influence the fertility monitoring devices industry. This includes a comprehensive study of consumer preferences, adoption rates, technological advancements, and regulatory frameworks that shape the sector.
Step 2: Market Analysis and Construction
This step focuses on analyzing historical data from multiple sources to construct accurate market estimates for the USA Fertility Monitors Devices market. The analysis includes looking at sales performance, growth rates, and market penetration for various product categories.
Step 3: Hypothesis Validation and Expert Consultation
We engage with industry experts, including product developers, healthcare professionals, and market analysts, to validate initial hypotheses. This phase involves interviews and surveys with key stakeholders to refine assumptions about the market’s growth trajectory.
Step 4: Research Synthesis and Final Output
In this phase, the data from primary and secondary research sources are synthesized. We validate findings through direct interaction with manufacturers, clinics, and retailers, ensuring a thorough and reliable analysis of the market. This final output serves as the foundation for forecasting and decision-making.
- Executive Summary
- Research Methodology (Market Definitions & Scope of Fertility Monitors Devices, Abbreviations & Nomenclature, Data Collection Framework, Quantitative & Qualitative Research Integration, Primary Research Protocols, Forecasting Models, Market Estimation Algorithms, Confidence Intervals & Error Margins, Research Limitations & Assumptions)
- Definition and Delineation of Fertility Monitors Devices
- Historical Market Genesis & Commercial Evolution
- Market Life Cycle and Adoption Curve
- Ecosystem Mapping — From OEMs to End‑Users
- Supply Chain Structure & Value Chain Alignment (Component Sourcing, Calibration Labs, Distribution Nodes)
- Reimbursement Dynamics & Insurance Coverage Landscape
- Market Growth Drivers
Rising Infertility Prevalence
U.S. Women’s Health Awareness
Employer Sponsored Fertility Benefits
Technological Miniaturization of Biosensors - Market Restraints
Reimbursement Gaps
Clinical Accuracy Concerns
Regulatory Barriers
- Growth Opportunities
AIEnabled Predictive Fertility Analytics
Telemedicine Integration
Personalized Fertility Regimens
- Emerging Market Trends
SubscriptionBased Models
Data Monetization via Health Platforms
Biometric API Ecosystems - Regulatory & Policy Environment
FDA Classification & Compliance Requirements
Standards for Clinical Validation & Label Claims
Health Data Privacy Regulations (HIPAA, State Privacy Laws)
Reimbursement Policies (Medicare, Commercial Insurers)
Post Marketing Surveillance and Reporting
- By Revenue (USD), 2026-2035
- By Unit Shipment Volumes, 2026-2035
- ASP (Average Selling Price) Dynamics, 2026-2035
- Installed Base & Replacement Cycles, 2026-2035
- By Technology (In Revenue %)
Basal Body Temperature Tracking
LH Surge Detection
MultiParameter Biosensor Systems
Salivary Ferning
Wearable PPG/ECG Hybrid Platforms - By Product Form Factor (In Revenue %)
Standalone Digital Monitors
Integrated Wearables
Smartphone‑Connected Sensors
At‑Home Test Kits
Clinical Grade Systems - By Connectivity & Deployment (In Revenue %)
Bluetooth‑Enabled
Wi‑Fi Enabled
USB/Offline Devices
App Ecosystem Integrated - By End‑User (In Revenue %)
Direct‑to‑Consumer
Fertility Clinics & IVF Centers
OB/GYN Specialist Practices
Telehealth Providers - By Distribution Channel (In Revenue %)
Ecommerce Retailers
BrickandMortar Medical Device Stores
Pharmacies/Drug Stores
Clinical Procurement Contracts - By Price Tier (In Revenue %)
Premium Tier
MidTier
Value/Entry Tier
- Market Structure (Consolidation, Fragmentation Indices)
- Market Share Analysis (Revenue & Device Shipments)
- Competitive Positioning Matrix (Price Tier vs Technology Depth)
- Innovation Index and Technology Readiness Levels (TRL)
- Cross Comparison Parameters (Product Accuracy, Algorithm Predictive Validity, Device Energy Efficiency & Battery Life, API/Cloud Integration Capability, Regulatory Clearance Status, Consumer Retention & Churn Rates, Channel Partner Coverage, App Store Rating & Engagement Metrics)
- Company Profiles of Key Players
Ava AG
Clearblue
Ovia Health
Kindara
Clue
Natural Cycles Nordic AB
Mira
Proov
Femometer Tech
Prima-Temp
Oviva Therapeutics
Philips Healthcare
GE Healthcare
Fitbit
- Clinic Buying Behavior and Procurement Cycles
- Prescriber Influence and Medical Guidelines
- Patient Decision Journey and Purchase Drivers
- Clinical Workflow Integration Barriers
- Pain Points and Unmet Needs Mapping
- By Revenue (USD), 2026-2035
- By Unit Shipment Volumes, 2026-2035
- By ASP (Average Selling Price) Dynamics, 2026-2035
- By Installed Base & Replacement Cycles, 2026-2035