Market Overview
The USA Immunoassay Techniques market is valued at approximately USD ~ billion, driven by the increasing demand for early disease detection, particularly in areas such as cancer and infectious diseases. The market is propelled by innovations in technology, including the integration of artificial intelligence and automation in immunoassay systems, as well as the growing shift toward point-of-care testing. These factors are encouraging widespread adoption across hospitals, diagnostic centers, and research institutions.
Dominant players in the USA Immunoassay Techniques market include major cities such as New York, Los Angeles, and Chicago, which are central hubs for healthcare, diagnostics, and medical research. These areas have seen significant infrastructure investments, fostering a favorable environment for the development of immunoassay techniques. The continued expansion of healthcare systems and rising healthcare expenditures, particularly in urban regions, are key factors driving dominance in these regions. Additionally, government initiatives to enhance public health are fueling growth in these areas.
Market Segmentation
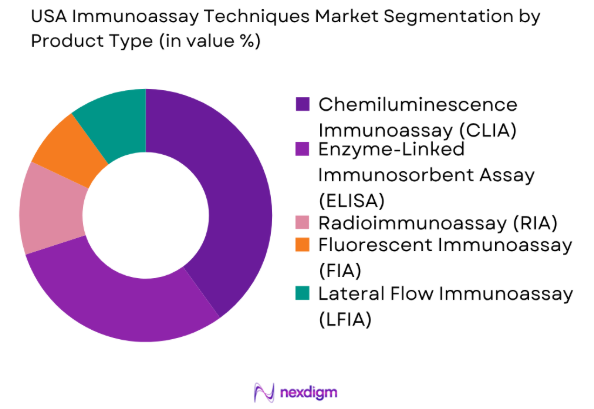
By Product Type
The USA Immunoassay Techniques market is segmented by product type into enzyme-linked immunosorbent assay (ELISA), radioimmunoassay (RIA), chemiluminescence immunoassay (CLIA), fluorescent immunoassay (FIA), and lateral flow immunoassay (LFIA). Recently, the chemiluminescence immunoassay (CLIA) sub-segment has a dominant market share due to its enhanced sensitivity, quicker results, and widespread adoption in hospital settings. The ease of automation and increasing demand for accurate diagnostic solutions contribute to the widespread use of CLIA systems in clinical and research labs.
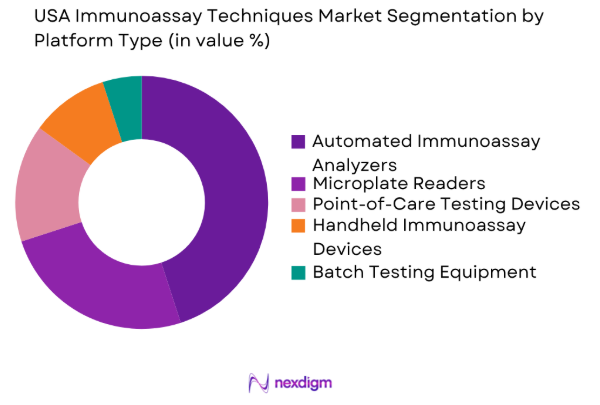
By Platform Type
The USA Immunoassay Techniques market is further segmented by platform type into microplate readers, automated immunoassay analyzers, point-of-care testing devices, handheld immunoassay devices, and batch testing equipment. Among these, automated immunoassay analyzers have seen the highest adoption due to their ability to handle large volumes of tests and provide quicker, more accurate results. Their integration with hospital management systems and automated workflows makes them the preferred choice in clinical laboratories and diagnostic centers.
Competitive Landscape
The USA Immunoassay Techniques market is highly competitive, with major players dominating both technology development and market share. Companies like Thermo Fisher Scientific and Abbott Laboratories are key players driving innovation in immunoassay techniques. These players are focusing on consolidating their market positions through partnerships, acquisitions, and product diversification, particularly in high-growth areas like point-of-care testing and AI-driven solutions. The market is characterized by continuous technological advancements, and the competition is intensifying as companies innovate to meet the growing demand for accurate and quick diagnostic tools.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD) | Market-Specific Parameter |
| Thermo Fisher Scientific | 1956 | Waltham, MA | ~ | ~ | ~ | ~ | ~ |
| Abbott Laboratories | 1888 | Chicago, IL | ~ | ~ | ~ | ~ | ~ |
| Roche Diagnostics | 1896 | Basel, Switzerland | ~ | ~ | ~ | ~ | ~ |
| Danaher Corporation | 1969 | Washington, D.C. | ~ | ~ | ~ | ~ | ~ |
| PerkinElmer | 1937 | Waltham, MA | ~ | ~ | ~ | ~ | ~ |
USA Immunoassay Techniques Market Analysis
Growth Drivers
Technological Advancements in Immunoassay Techniques
The USA Immunoassay Techniques market is largely driven by technological innovations such as the development of chemiluminescence immunoassay (CLIA) systems, point-of-care testing, and AI integration. These advancements have led to faster, more accurate test results, driving increased adoption across clinical and research labs. Furthermore, automation is reducing labor costs and improving the scalability of immunoassays, thus supporting market expansion. The constant push towards more efficient, automated solutions is ensuring that demand continues to grow as healthcare providers seek cost-effective methods to manage patient care. Additionally, these advancements have led to significant improvements in diagnostics for chronic diseases, infections, and cancer, fueling further market growth.
Increased Demand for Early Disease Diagnosis
Early disease detection plays a crucial role in improving patient outcomes and reducing healthcare costs. Immunoassay techniques are pivotal in diagnosing diseases such as cancer, cardiovascular conditions, and infectious diseases in their early stages. The USA Immunoassay Techniques market is experiencing growth due to an increasing demand for early diagnosis, driven by rising awareness, expanding access to healthcare, and advancements in medical research. The growing need for personalized medicine, which relies heavily on accurate diagnostics, is also contributing to the widespread adoption of immunoassays. As healthcare systems focus on proactive care, demand for immunoassays will continue to rise, further driving the market forward.
Market Challenges
High Cost of Advanced Immunoassay Equipment
A significant challenge in the USA Immunoassay Techniques market is the high cost of advanced systems, especially automated analyzers and point-of-care devices. The initial investment required for these technologies is substantial, posing a barrier for smaller diagnostic centers and facilities in rural areas. Beyond the upfront costs, the ongoing maintenance and operational expenses of these sophisticated systems add further financial strain on healthcare providers. While larger hospitals and laboratories can typically absorb these costs, smaller clinics and independent practitioners face difficulty adopting these cutting-edge technologies. This cost disparity limits market penetration in less well-funded regions and among smaller healthcare providers, hindering broader market expansion and the widespread adoption of advanced immunoassay techniques.
Regulatory Barriers and Compliance Issues
Immunoassay devices must meet stringent regulatory standards set by authorities such as the FDA. The regulatory hurdles associated with the approval of new immunoassay technologies can delay product launches and market entry, particularly for smaller companies or new market entrants. These challenges are compounded by the need to comply with various quality control standards, such as ISO certification. The lengthy approval process can limit innovation and slow down the overall market growth, especially when dealing with new product developments or regulatory updates. The complexity of the regulatory environment poses a continuous challenge for companies striving to bring their products to market.
Opportunities
Expansion of Point-of-Care Testing Solutions
Point-of-care (POC) testing presents a major opportunity in the USA Immunoassay Techniques market. The increasing demand for rapid diagnostic results, especially in emergency situations, has led to a rise in the use of portable and user-friendly immunoassay devices. As healthcare systems focus on improving accessibility and efficiency, there is a growing trend toward POC immunoassays. These devices provide quick results, reduce the reliance on complex laboratory infrastructure, and offer cost-effective solutions. With healthcare providers and patients prioritizing convenience and speed, POC immunoassays are expected to gain significant market share in the coming years, revolutionizing the diagnostic landscape by making testing more accessible in diverse settings.
Increasing Adoption in Personalized Medicine
Personalized medicine is transforming healthcare by customizing treatments based on individual genetic profiles, lifestyle, and other factors. Immunoassays are crucial in this process, enabling precise and rapid biomarker measurement essential for creating personalized treatment plans. As the focus on personalized healthcare grows, there is an increasing demand for immunoassay techniques that provide accurate, targeted diagnostic results. With ongoing advancements in biotechnology and personalized medicine, the need for innovative immunoassay solutions will rise, supporting the growing trend of individualized care. These advancements will enhance the ability to deliver tailored healthcare solutions, further driving the demand for advanced immunoassay technologies in the market.
Future Outlook
The future outlook for the USA Immunoassay Techniques market is promising, with significant growth anticipated over the next five years. This growth will be driven by continued technological advancements, particularly in point-of-care testing and automated systems. As the healthcare industry shifts towards more personalized, efficient, and cost-effective care, immunoassays will play a crucial role in diagnosing a variety of diseases. With increasing regulatory support and the continued rise in healthcare spending, the market is expected to witness sustained growth, particularly as demand for faster, more accurate diagnostic tools continues to rise.
Major Players
- ThermoFisher Scientific
- Abbott Laboratories
- Roche Diagnostics
- Danaher Corporation
- PerkinElmer
- Siemens Healthineers
- Bio-Rad Laboratories
- Becton Dickinson
- Quidel Corporation
- Ortho Clinical Diagnostics
- Mindray
- BioMérieux
- Agilent Technologies
- Beckman Coulter
- Sysmex Corporation
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Diagnosticcenters
- Clinical laboratories
- Pharmaceutical companies
- Healthcare providers
- Research institutes
- Biotechnology firms
Research Methodology
Step 1: Identification of Key Variables
The first step involves identifying the core market variables that impact the immunoassay techniques market. This includes understanding key drivers such as technological advancements, consumer behavior, and regulatory factors that influence market growth.
Step 2: Market Analysis and Construction
After identifying key variables, market analysis is conducted using primary and secondary research. This step involves gathering quantitative data through surveys and qualitative data from industry reports and expert consultations to build an accurate market model.
Step 3: Hypothesis Validation and Expert Consultation
The hypotheses derived from initial analysis are tested through consultations with industry experts, including professionals from diagnostics companies, regulatory bodies, and end users. This step ensures the validity and robustness of the market projections.
Step 4: Research Synthesis and Final Output
The final step synthesizes all collected data, validates findings, and prepares the comprehensive market report, which includes market forecasts, competitive landscape, growth drivers, challenges, and opportunities.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Technological Advancements in Immunoassay Techniques
Growing Demand for Early Disease Diagnosis
Expansion of Point-of-Care Testing Solutions - Market Challenges
High Cost of Advanced Immunoassay Equipment
Regulatory Barriers for Immunoassay Devices
Complexity in Sample Collection and Processing - Market Opportunities
Rising Adoption of Immunoassays in Personalized Medicine
Increasing Use of Immunoassays for Infectious Disease Diagnosis
Expanding Applications in Cancer Diagnostics - Trends
Integration of Artificial Intelligence in Immunoassays
Increased Demand for Home-based Immunoassays
Technological Innovations in Point-of-Care Testing - Government Regulations
- SWOT Analysis of Key Competitors
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Enzyme-Linked Immunosorbent Assay (ELISA)
Radioimmunoassay (RIA)
Chemiluminescence Immunoassay (CLIA)
Fluorescent Immunoassay (FIA)
Lateral Flow Immunoassay (LFIA) - By Platform Type (In Value%)
Microplate Readers
Automated Immunoassay Analyzers
Point-of-Care Testing Devices
Handheld Immunoassay Devices
Batch Testing Equipment - By Fitment Type (In Value%)
Laboratory-based Immunoassays
Point-of-Care Immunoassays
Home-based Immunoassays
Automated Immunoassay Systems
Portable Immunoassay Devices - By End User Segment (In Value%)
Hospitals
Clinical Laboratories
Research Institutes
Diagnostic Centers
Pharmaceutical Companies - By Procurement Channel (In Value%)
Direct Procurement
Distributor Networks
Online Procurement
Third-party Vendors
Government Procurement
- Market Share Analysis
- Cross Comparison Parameters (System Type, Platform Type, Procurement Channel, End User Segment, Fitment Type, Diagnostic Type, Testing Method, Reimbursement Model, Technological Integration, Regulatory Compliance)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Thermo Fisher Scientific
Abbott Laboratories
Roche Diagnostics
Siemens Healthineers
Danaher Corporation
Bio-Rad Laboratories
Becton Dickinson
Quidel Corporation
Ortho Clinical Diagnostics
PerkinElmer
Merck Group
F. Hoffmann-La Roche
Waters Corporation
Mindray
BioMérieux
- Increasing Adoption in Clinical Diagnostics
- Focus on Personalized Healthcare Solutions
- Growing Research and Development in Immunoassays
- Increased Investment in Diagnostic Centers
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035