Market Overview
The USA medical devices market has witnessed significant growth, with its size driven by rising healthcare expenditures and the continuous innovation of new medical technologies. Based on a recent historical assessment, the market size stands at approximately USD ~ billion, with robust demand for diagnostic and therapeutic devices. This growth is propelled by the increasing prevalence of chronic diseases, an aging population, and the expanding need for healthcare services. Additionally, government healthcare funding and the adoption of advanced technologies have played pivotal roles in shaping this upward trajectory.
The USA continues to lead the medical devices market, driven by the concentration of major manufacturers, cutting-edge research, and technological advancements. Cities like San Francisco, Boston, and New York remain at the forefront due to their strong healthcare infrastructure, skilled workforce, and proximity to top research universities and hospitals. These regions are hubs for both the development and commercialization of medical devices, supported by favorable regulatory environments and substantial investments from both public and private sectors.
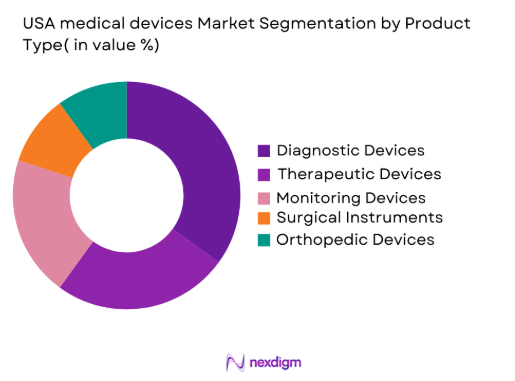
Market Segmentation
By Product Type
The USA medical devices market is segmented by product type into diagnostic devices, therapeutic devices, monitoring devices, surgical instruments, and orthopedic devices. Recently, diagnostic devices have been dominating the market share due to the increasing demand for early disease detection, improved healthcare services, and preventive care. The presence of advanced technologies such as AI-based diagnostics and non-invasive imaging systems has further boosted their adoption, with hospitals, clinics, and diagnostic centers continuously seeking cutting-edge solutions for accurate diagnoses. These products are becoming critical for reducing healthcare costs while improving patient outcomes, which has enhanced their market dominance.
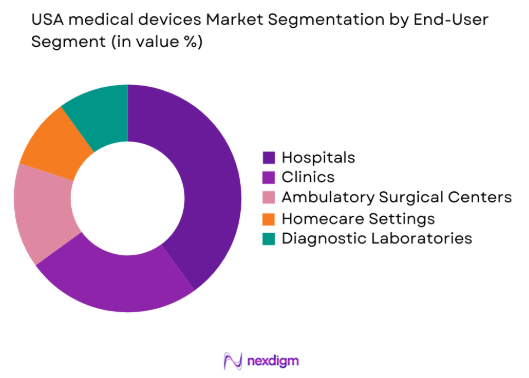
By End-User Segment
The market is segmented by end-user into hospitals, clinics, ambulatory surgical centers, homecare settings, and diagnostic laboratories. Hospitals have a dominant market share, driven by their large-scale medical device usage in diagnostic, therapeutic, and monitoring applications. The growing number of hospitals and healthcare facilities, along with increased investments in healthcare infrastructure, are key drivers of this trend. As the demand for medical devices increases, hospitals are becoming primary consumers of advanced devices, particularly in critical care and surgical departments, ensuring their leadership in the market.
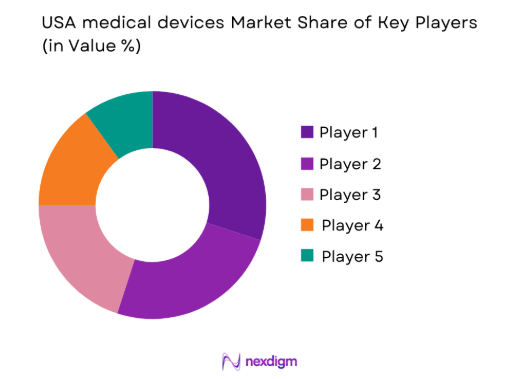
Competitive Landscape
The USA medical devices market is highly competitive, with major players driving innovation and consolidation. Companies such as Medtronic, Abbott Laboratories, and Johnson & Johnson maintain significant influence, continuously investing in product development and strategic acquisitions. These companies leverage advanced technologies and robust distribution networks to expand their market reach. Furthermore, the growing number of mergers and partnerships within the industry highlights the increasing importance of collaboration to stay competitive. The influence of these large corporations continues to shape the market, with both established players and new entrants focusing on innovation and regulatory compliance.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD) | Innovation Focus |
| Medtronic | 1949 | Minneapolis, MN | ~ | ~ | ~ | ~ | ~ |
| Abbott Laboratories | 1888 | Chicago, IL | ~ | ~ | ~ | ~ | ~ |
| Johnson & Johnson | 1886 | New Brunswick, NJ | ~ | ~ | ~ | ~ | ~ |
| Boston Scientific | 1979 | Marlborough, MA | ~ | ~ | ~ | ~ | ~ |
| Stryker | 1941 | Kalamazoo, MI | ~ | ~ | ~ | ~ | ~ |
USA medical devices Market Analysis
Growth Drivers
Rising Geriatric Population
The aging population in the USA is a significant driver of growth in the medical devices market. As the number of elderly individuals increases, so does the demand for devices, especially those related to chronic disease management, orthopedic care, and diagnostics. Older adults are more susceptible to conditions like heart disease, diabetes, and joint disorders, which fuels the need for advanced medical devices. The growing focus on improving the health and quality of life for the elderly, alongside longer life expectancy and better healthcare outcomes, further strengthens this trend. Consequently, demand for diagnostic, monitoring, and treatment devices will continue to rise, supporting market growth.
Technological Advancements in Minimally Invasive Procedures
The continuous development of minimally invasive techniques is another significant driver of the medical devices market. Procedures such as robotic surgeries, laparoscopic surgeries, and endoscopic diagnostics have revolutionized the healthcare landscape, providing patients with quicker recovery times and fewer complications. Medical devices that enable these procedures are in high demand, as they offer significant advantages over traditional surgical techniques. The increasing adoption of robotic surgery systems, imaging technologies, and specialized surgical instruments is expected to drive further growth, as hospitals and healthcare providers look to reduce costs while improving patient care outcomes. This shift toward minimally invasive procedures is fundamentally changing the medical devices landscape, making it one of the key drivers of market growth.
Market Challenges
High Regulatory Barriers
The medical devices market in the USA faces significant regulatory challenges due to the complex and stringent regulations imposed by the Food and Drug Administration (FDA) and other governing bodies. Manufacturers must navigate through rigorous approval processes for new products, including clinical trials, safety assessments, and post-market surveillance. These regulatory requirements not only increase time-to-market but also incur high costs, particularly for smaller firms and new entrants. The evolving regulatory landscape, especially regarding device recalls and adverse event reporting, adds an additional layer of complexity for manufacturers. Compliance with these regulations is critical for market entry and sustainability, but it also presents a significant challenge for companies operating in the medical devices market.
Supply Chain Disruptions
Global supply chain disruptions continue to pose challenges for the medical devices market. The COVID-19 pandemic highlighted the vulnerabilities in supply chains, affecting the availability of raw materials, manufacturing capabilities, and distribution networks. These disruptions have led to delays in product launches, shortages of critical devices, and increased operational costs. Additionally, the reliance on international suppliers for components, especially from Asia, exposes companies to risks associated with geopolitical tensions and trade policies. As the demand for medical devices increases, the strain on supply chains continues to hinder the smooth functioning of the market, posing a considerable challenge for manufacturers to meet consumer expectations and regulatory timelines.
Opportunities
Expansion of Home Healthcare Market
The home healthcare market presents significant growth opportunities for the medical devices sector. With the increasing preference for in-home care, especially among the aging population, medical devices tailored for home use, such as remote monitoring devices, portable diagnostic tools, and telehealth solutions, are gaining traction. The advancements in IoT-enabled devices that allow real-time patient monitoring from home are revolutionizing the healthcare experience. Additionally, the COVID-19 pandemic has accelerated the adoption of home healthcare solutions, further expanding the potential market for these devices. This trend is expected to continue, driven by the rising demand for cost-effective and convenient healthcare solutions, creating opportunities for medical device manufacturers to tap into this growing segment.
Growth in AI and Robotics in Surgery
The integration of AI and robotics in surgical procedures offers substantial opportunities for the medical devices market. Robotic-assisted surgeries, such as those used in orthopedics, urology, and cardiology, are becoming more prevalent due to their ability to enhance precision, reduce recovery times, and minimize surgical risks. AI-powered diagnostic tools are also improving early disease detection, particularly in oncology and cardiology. The continuous advancements in AI and robotic technology are enabling medical professionals to perform complex procedures with greater accuracy, transforming the surgical landscape. As healthcare providers seek to incorporate these cutting-edge technologies to improve patient outcomes, the demand for related medical devices is expected to increase, offering significant market opportunities.
Future Outlook
The future outlook for the USA medical devices market indicates continued growth driven by technological advancements, increasing demand for healthcare services, and a strong focus on chronic disease management. The market is expected to experience further expansion as AI, robotics, and minimally invasive procedures continue to evolve. Additionally, regulatory support for innovative medical devices and the increasing integration of digital health solutions are set to drive market growth over the next few years. This period will see further innovations that will shape the future of healthcare delivery, with significant potential for new product introductions.
Major Players
- Medtronic
- Abbott Laboratories
- Johnson & Johnson
- Boston Scientific
- Stryker
- Siemens Healthineers
- GE Healthcare
- Philips Healthcare
- Zimmer Biomet
- B. Braun Melsungen
- Fresenius Medical Care
- Roche Diagnostics
- 3M
- Danaher Corporation
- Smith & Nephew
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Healthcare providers
- Hospitals and diagnostic centers
- Medical device manufacturers
- Medical device distributors
- Health insurance companies
- Private equity firms
Research Methodology
Step 1: Identification of Key Variables
The first step involves identifying critical variables that influence the medical devices market, including market drivers, regulatory factors, and technological advancements.
Step 2: Market Analysis and Construction
A thorough analysis is conducted to construct a comprehensive view of the market by evaluating trends, competitive dynamics, and consumer behavior.
Step 3: Hypothesis Validation and Expert Consultation
Hypotheses are validated by consulting industry experts, stakeholders, and utilizing primary data sources to refine the findings.
Step 4: Research Synthesis and Final Output
The final step involves synthesizing all research findings into a cohesive report, ensuring accuracy and relevance to the current market conditions.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Increased Healthcare Expenditure
Rising Geriatric Population
Advances in Minimally Invasive Surgeries - Market Challenges
High Regulatory Scrutiny
Rising Healthcare Costs
Cybersecurity Threats in Medical Devices - Market Opportunities
Expanding Home Healthcare Market
Adoption of AI in Diagnostics
Growth of Telemedicine Platforms - Trends
Integration of IoT in Medical Devices
Personalized Medicine Technologies
Robotic-assisted Surgery Advancements - Government Regulations
- SWOT Analysis of Key Competitors
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Diagnostic Devices
Therapeutic Devices
Monitoring Devices
Surgical Instruments
Orthopedic Devices - By Platform Type (In Value%)
Hospital-based Platforms
Homecare Platforms
Mobile Health Platforms
Point-of-Care Platforms
Telemedicine Platforms - By Fitment Type (In Value%)
Wearable Devices
Implantable Devices
Non-invasive Devices
Invasive Devices
Portable Devices - By End User Segment (In Value%)
Hospitals
Clinics
Ambulatory Surgical Centers
Homecare Settings
Diagnostic Laboratories - By Procurement Channel (In Value%)
Direct Procurement
Government Procurement
Private Procurement
Online Procurement Platforms
Third-party Distributors
- Market Share Analysis
- Cross Comparison Parameters (Product Type, End-User Segment, Platform Type, Procurement Channel, Fitment Type, Technology Integration, Regulatory Compliance, Market Reach, Distribution Channels, Consumer Adoption)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Medtronic
Abbott Laboratories
Johnson & Johnson
Boston Scientific
Stryker
Siemens Healthineers
GE Healthcare
Philips Healthcare
Zimmer Biomet
B. Braun Melsungen
Fresenius Medical Care
Roche Diagnostics
3M
Danaher Corporation
Smith & Nephew
- Increasing Demand from Hospitals
- Homecare Market Growth
- Rise in Surgical Interventions
- Technology Adoption in Diagnostic Labs
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035