Market Overview
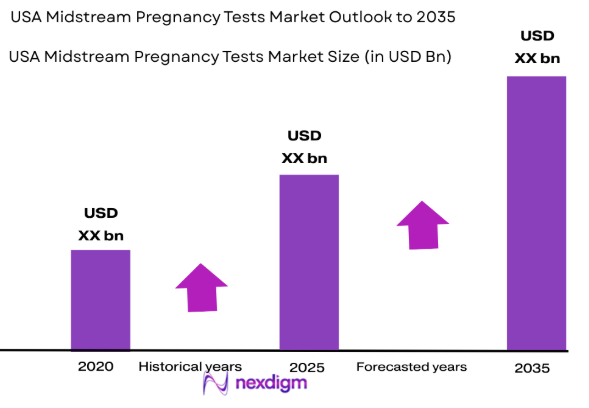
The USA Midstream Pregnancy Tests Market is valued at approximately USD ~ billion in 2024. This growth is attributed to factors such as an increasing preference for home-based diagnostic solutions, advancements in midstream test kit technology, and rising consumer demand for reliable and quick results. The market is driven by the growing awareness of early pregnancy detection and the increasing accessibility of over-the-counter products. The market has also been boosted by continuous innovations that enhance ease of use and product accuracy.
The USA leads the Midstream Pregnancy Tests Market, driven by urbanized regions such as New York, Los Angeles, and Chicago. These cities dominate due to their high population density, higher disposable incomes, and widespread access to retail pharmacies and online channels. Additionally, the rapid adoption of homecare diagnostics in these areas is facilitated by extensive healthcare awareness, including the use of early-stage pregnancy detection kits. Strong retail networks, including major pharmacy chains and online platforms, also contribute to the dominance of these regions in the market.
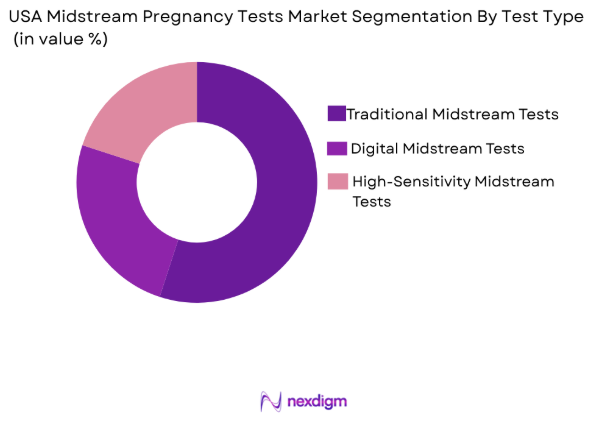
Market Segmentation
By Test Type
The USA Midstream Pregnancy Tests Market is segmented by test type into traditional midstream tests, digital midstream tests, and high-sensitivity midstream tests. Among these, traditional midstream tests have a dominant market share due to their long-standing presence and high consumer trust. These tests offer ease of use and provide reliable results, making them a preferred option for most consumers. The simplicity of design, affordability, and accessibility in retail outlets further drives their dominance in the market. On the other hand, digital tests and high-sensitivity tests are gradually gaining traction due to increased consumer preference for more accurate and technologically advanced options, though they currently account for a smaller market share.
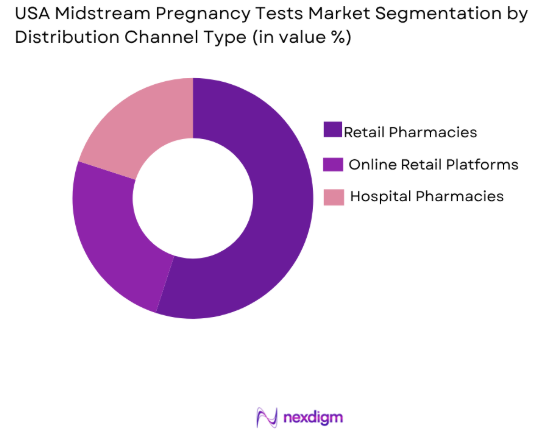
By Distribution Channel Type
The market is segmented by distribution channel into retail pharmacies, online retail platforms, and hospital pharmacies. Retail pharmacies hold the dominant market share as they provide easy access to over-the-counter pregnancy tests. Chains like CVS, Walgreens, and Rite Aid serve as major touchpoints for consumers, while their wide network ensures that pregnancy tests are readily available to a vast population. Online retail platforms are also growing in popularity, offering convenience and the ability to compare brands easily. Hospital pharmacies are a niche segment, often serving clinical environments and offering higher-priced, medically certified tests.
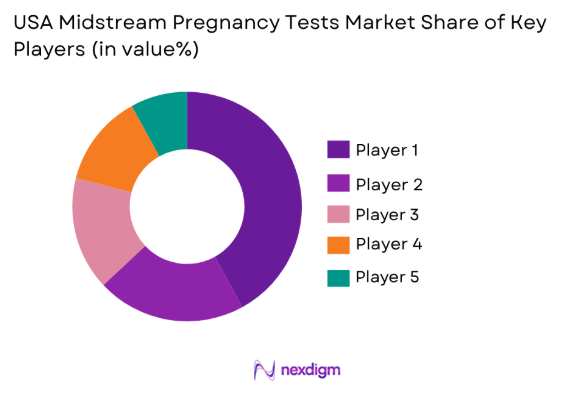
Competitive Landscape
The USA midstream pregnancy tests market is dominated by key players, including Abbott Laboratories, Procter & Gamble (Clearblue), and Church & Dwight Co. These companies hold significant market share due to their strong brand presence, wide distribution networks, and continuous innovation in product offerings.
| Company Name | Establishment Year | Headquarters | Product Range | Revenue (USD) | Distribution Channels | Innovation Index | Market Penetration |
| Abbott Laboratories | 1888 | Chicago, USA | ~ | ~ | ~ | ~ | ~ |
| Procter & Gamble | 1837 | Cincinnati, USA | ~ | ~ | ~ | ~ | ~ |
| Church & Dwight Co. | 1846 | Ewing, USA | ~ | ~ | ~ | ~ | ~ |
| Quidel Corporation | 1979 | San Diego, USA | ~ | ~ | ~ | ~ | ~ |
| Mankind Pharma | 1995 | New York, USA | ~ | ~ | ~ | ~ | ~ |
USA Midstream Pregnancy Tests Market Analysis
Growth Drivers
Rising Preference for Early Self‑Testing
Self-testing has gained immense popularity due to the increased availability of over the counter (OTC) diagnostic products like midstream pregnancy tests. The convenience and privacy offered by self-testing solutions have contributed to the rising consumer preference for such products. According to the U.S. Food and Drug Administration (FDA), OTC diagnostics have grown by 15% annually from 2021 to 2023. This growth has been fueled by a general increase in healthcare awareness and an expanding consumer market seeking more accessible healthcare options. The increasing demand for home-based medical testing is further supported by macroeconomic shifts toward individual healthcare empowerment and a shift in consumer spending toward personal health.
Digitalization of At‑Home Diagnostics
The rise of digital health technologies has greatly impacted the at-home diagnostics market, particularly in pregnancy tests. Digital pregnancy tests, which include mobile app connectivity and digital displays, have seen a surge in adoption. The growth of telehealth and connected healthcare services in the U.S. is also driving digitalization in diagnostics. In 2022, the number of U.S. telehealth consultations reached 45 million, marking a 5% increase from the previous year. This growth reflects the broader shift toward digitization, especially in health diagnostics, which is helping consumers make more informed decisions in real time. Additionally, the ongoing development of digital devices for personal health monitoring plays a role in boosting demand for digital at-home pregnancy tests.
Market Restraints
Price Sensitivity & Reimbursement Gaps
Price sensitivity remains a significant challenge in the midstream pregnancy test market. The increasing number of budget-conscious consumers, particularly in lower-income households, means that affordability becomes a key factor in purchasing decisions. According to data from the U.S. Census Bureau, over 13% of the U.S. population, or approximately 43 million people, lived below the poverty line in 2023. This economic reality impacts the accessibility of certain diagnostic products, especially premium-priced models. Additionally, the lack of insurance coverage for at-home pregnancy tests exacerbates this issue, as consumers are forced to pay out-of-pocket for products that may be considered essential healthcare tools. As the market continues to evolve, addressing these affordability issues will be key to expanding market reach.
False Negatives / Sensitivity Limits
Despite advancements in test accuracy, false negatives remain a concern in the midstream pregnancy test market. This issue is particularly problematic in cases where pregnancy hormones (hCG) are present in lower concentrations, which can occur in very early stages of pregnancy. A study by the National Institutes of Health (NIH) showed that false-negative results still occur in approximately 5-10% of early-stage tests, especially with lower-sensitivity tests. Additionally, certain health conditions, such as ovarian cysts or the use of fertility treatments, can influence test results. The persistence of these challenges raises concerns among consumers, especially as they seek higher reliability from self-diagnostic tools.
Market Opportunities
Connected Diagnostics & Telehealth Integration
The future of the midstream pregnancy test market is poised for significant growth through connected diagnostics and telehealth integration. Currently, around 38 million people in the U.S. use some form of telehealth service, and this number is expected to increase as healthcare becomes more digital and personalized. The integration of pregnancy tests with telehealth platforms is becoming increasingly common, with leading companies developing systems that allow test results to be shared with healthcare providers via mobile apps or telemedicine consultations. By offering an integrated experience, users can receive professional advice following the test results, enhancing the convenience and accuracy of the testing process. Furthermore, innovations in connected diagnostics are expected to reduce testing errors and increase consumer confidence in using these products regularly, potentially boosting market demand.
Retail Pharmacy Expansion
The expansion of retail pharmacies has become a key growth driver for the midstream pregnancy test market, especially with the increasing role of pharmacies as healthcare hubs. According to the National Association of Chain Drug Stores (NACDS), the number of retail pharmacies in the U.S. reached 69,000 in 2023, marking a steady increase over the past few years. This growth provides consumers with greater accessibility to pregnancy tests, as these retail locations are often considered the first point of contact for healthcare needs. As the retail pharmacy sector continues to expand, particularly in rural and underserved areas, it offers a significant opportunity for pregnancy test manufacturers to increase distribution and market presence
Future Outlook
Over the next 5 years, the USA midstream pregnancy tests market is expected to witness significant growth. Key factors contributing to this growth include advancements in diagnostic accuracy, growing consumer preference for home-based tests, and the expansion of digital health technologies. The rise of e-commerce and retail pharmacies as primary distribution channels will also play a crucial role in enhancing market accessibility. Furthermore, the integration of smartphone applications and digital devices with pregnancy testing kits will likely influence consumer preferences and boost market expansion.
Major Players
- Abbott Laboratories
- Church & Dwight Co.
- Procter & Gamble
- Quidel Corporation
- McKesson Corporation
- Roche Diagnostics
- Siemens Healthineers
- Meridian Bioscience
- Thermo Fisher Scientific
- Danaher Corporation
- AccuMed
- HCG Pregnancy Tests Co.
- Alere
- First Response
- Pregnancy Test
Key Target Audience
- Investments and Venture Capitalist Firms
- Government and Regulatory Bodies
— FDA - Private Label Manufacturers
- Retail Pharmacies and Drugstores
- E-commerce Retailers
- Healthcare Providers
- Hospitals and Diagnostic Laboratories
- OB-GYN and Reproductive Health Clinics
Research Methodology
Step 1: Identification of Key Variables
The first step in the research methodology involves the identification of all significant market drivers, barriers, and potential growth factors for the USA midstream pregnancy tests market. This process is guided by a combination of secondary research using historical market data, primary research interviews, and expert consultations.
Step 2: Data Collection and Market Analysis
In this phase, historical data is compiled to understand market trends and growth trajectories. We also perform an in-depth analysis of product types, distribution channels, and regulatory landscape through multiple sources, including government reports and proprietary databases.
Step 3: Expert Consultation and Validation
Through a series of expert consultations with healthcare professionals, manufacturers, and retail experts, we validate our hypotheses and gather insights into the dynamics shaping the market. The consultations ensure the data’s reliability and accuracy, providing a foundation for forecasting.
Step 4: Data Synthesis and Reporting
The final phase involves synthesizing the collected data and insights, and analyzing it through various segmentation models. This phase integrates consumer preferences, market behavior, and emerging trends, which are then presented in a structured report format to provide a comprehensive view of the USA midstream pregnancy tests market.
- Executive Summary
- Research Methodology (Market Definitions & Assumptions, Abbreviations, Data Sources & Sample Sizes, Market Sizing Framework, Forecast Methodology, Primary & Secondary Research Validation, Bias Control, Limitations)
- Market Definition and Scope
- Market Genesis and Evolution
- Adoption Lifecycle
- Supply Chain & Value Chain Dynamics
- Diagnostic Accuracy & Sensitivity Benchmarks
- Growth Drivers
Rising Preference for Early Self‑Testing
Digitalization of At‑Home Diagnostics - Market Restraints
Price Sensitivity & Reimbursement Gaps
False Negatives / Sensitivity Limits - Market Opportunities
Connected Diagnostics & Telehealth Integration
Retail Pharmacy Expansion - Emerging Trends
Smart Strip & Digital Notifications
IoT‑Enabled Diagnostic Apps - SWOT Analysis
- Porter’s Five Forces
- Competition Ecosystem
- By Value, 2020-2025
- By Volume, 2020-2025
- By Average Selling Price, 2020-2025
- By Distribution Channel, 2020-2025
- By Test Type (in Value%)
Traditional Midstream Tests
Digital Midstream Tests
High-Sensitivity Midstream Tests - By Distribution Channel (in Value%)
Retail Pharmacies
Online Retail Platforms
Hospital Pharmacies - By End‑User (in Value%)
Homecare Users
Clinical & Diagnostic Labs
OB‑GYN & Specialty Clinics
Community Health Centers
Public Health Programs - By Sensitivity Threshold (in Value %)
10 mIU/mL Sensitivity Midstream
20 mIU/mL Sensitivity Midstream
25+ mIU/mL Sensitivity Midstream - By Price Tier (in Value%)
Premium Segment
Mid‑Tier
Value/Budget Segment
- USA Market Share by Value & Volume – Top Players
- Cross‑Comparison Parameters (Company Overview, Product Portfolio & hCG Sensitivity Levels, Retail & Channel Coverage, Distribution Network Density, Midstream Product ASP & Variants, Brand Recognition & Marketing Reach, R&D Investment / Innovation Score, Regulatory Compliance & Quality Certifications)
- SWOT Analysis of Major Players
- Pricing Analysis Based on SKUs for Major Players in UAE RF
- Microneedling Devices Market
- Detailed Profiles of Major Companies
Abbott Laboratories
Church & Dwight Co. Inc.
SPD Swiss Precision Diagnostics
Cardinal Health
QuidelOrtho
McKesson Corporation
Danaher Corporation
Sekisui Chemical
Thermo Fisher Scientific
Medline Industries
Meridian Bioscience
Rapigen
Bayer AG
Roche Diagnostics
Siemens Healthineers
- Test Adoption Patterns
- Frequency & Repeat Testing Demand
- Price Elasticity & Brand Loyalty
- Pain Points & User Experience Insights
- Decision‑Making Influencers
- By Value, 2026-2035
- By Volume, 2026-2035
- By Average Selling Price, 2026-2035
- By Distribution Channel, 2026-2035