Market Overview
The USA Neurostimulators Devices Market demonstrates a strong valuation of approximately USD ~ billion, based on a recent historical assessment supported by data from healthcare device tracking authorities such as the U.S. FDA database and CMS reimbursement records. The market is driven by rising incidences of neurological disorders, including Parkinson’s disease, epilepsy, and chronic pain syndromes, along with increasing adoption of minimally invasive neurostimulation therapies. Advancements in implantable technologies and favorable reimbursement structures further reinforce market expansion.
The market is primarily concentrated in major healthcare hubs such as California, Massachusetts, Texas, and New York, where advanced medical infrastructure and specialized neurology centers support adoption. Cities like Boston, San Francisco, and Houston lead due to the presence of leading hospitals, research institutions, and medtech companies. Strong clinical trial ecosystems and early adoption of innovative neuromodulation therapies contribute significantly to regional dominance within the United States.
Market Segmentation
By Product Type
USA Neurostimulators Devices Market is segmented by product type into spinal cord stimulators, deep brain stimulators, vagus nerve stimulators, sacral nerve stimulators, and transcutaneous electrical nerve stimulators. Recently, spinal cord stimulators have a dominant market share due to factors such as high prevalence of chronic pain conditions, strong clinical validation, and widespread reimbursement support. Their effectiveness in treating failed back surgery syndrome and neuropathic pain has significantly increased adoption across hospitals and specialty clinics, supported by continuous technological improvements.

By EndUser
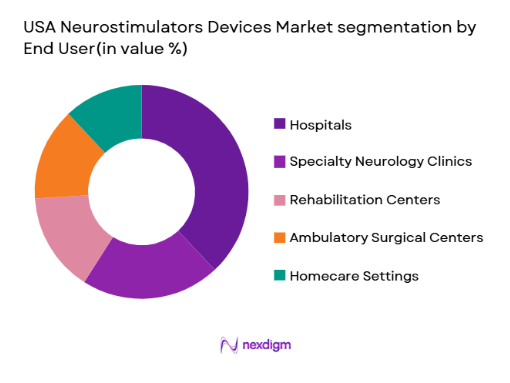
USA Neurostimulators Devices Market is segmented by end users into hospitals, specialty neurology clinics, rehabilitation centers, ambulatory surgical centers, and homecare settings. Recently, hospitals have a dominant market share due to factors such as availability of advanced surgical infrastructure, skilled neurosurgeons, and higher patient inflow for complex procedures. Hospitals also benefit from integrated diagnostic capabilities and access to reimbursement systems, which supports large-scale adoption of implantable neurostimulators.
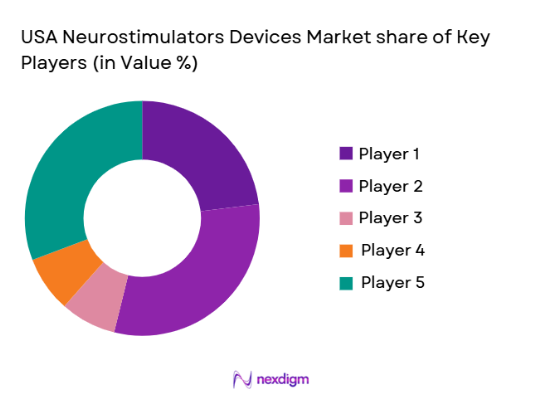
Competitive Landscape
The USA Neurostimulators Devices Market is moderately consolidated, with a few dominant players controlling a significant portion of innovation and distribution channels. Large medical device companies leverage extensive R&D capabilities, regulatory expertise, and global supply chains to maintain competitive advantage. Strategic partnerships, acquisitions, and product portfolio expansion remain key strategies shaping competition.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Innovation Focus |
| Medtronic Plc | 1949 | Dublin, Ireland | ~ | ~ | ~ | ~ | ~ |
| Abbott Laboratories | 1888 | Illinois, USA | ~ | ~ | ~ | ~ | ~ |
| Boston Scientific | 1979 | Massachusetts, USA | ~ | ~ | ~ | ~ | ~ |
| LivaNova Plc | 1987 | London, UK | ~ | ~ | ~ | ~ | ~ |
| Nevro Corp | 2006 | California, USA | ~ | ~ | ~ | ~ | ~ |
USA Neurostimulators Devices Market Analysis
Growth Drivers
Increasing Prevalence of Neurological Disorders and Chronic Pain Conditions:
The growing incidence of neurological disorders such as Parkinson’s disease, epilepsy, and chronic pain syndromes is a primary driver of the USA Neurostimulators Devices Market. Aging populations contribute significantly to the rise in neurodegenerative conditions, thereby increasing demand for effective long-term treatment options. Neurostimulators provide minimally invasive solutions that offer sustained relief compared to conventional therapies. Patients with refractory conditions increasingly prefer neuromodulation due to reduced dependency on pharmacological treatments. Chronic pain management remains a major healthcare burden, driving healthcare providers to adopt advanced neurostimulation devices. The integration of real-time monitoring capabilities enhances treatment outcomes, further boosting adoption. Healthcare systems are actively investing in innovative therapies to reduce hospitalization costs associated with long-term neurological conditions. Additionally, increasing awareness among patients and physicians about neurostimulation benefits supports market expansion. Technological advancements are making these devices more efficient and patient-friendly, accelerating their uptake across healthcare facilities.
Technological Advancements in Implantable Neurostimulation Devices:
Rapid advancements in neurostimulation technology are significantly enhancing the capabilities and adoption of these devices. Innovations such as closed-loop systems, wireless programming, and extended battery life have improved device efficiency and patient outcomes. Manufacturers are focusing on miniaturization and improved electrode design to enhance patient comfort and surgical precision. AI-driven neurostimulation systems enable personalized therapy, allowing real-time adjustments based on patient responses. These advancements reduce complications and increase the success rate of procedures. Integration with digital health platforms facilitates remote monitoring and follow-up care, improving overall treatment effectiveness. Continuous R&D investments by leading players are accelerating the introduction of next-generation devices. Regulatory support for innovative technologies also plays a crucial role in facilitating market growth. As a result, healthcare providers are increasingly adopting advanced neurostimulators, contributing to sustained market expansion.
Market Challenges
High Cost of Neurostimulator Devices and Surgical Procedures:
The high cost associated with neurostimulator devices and implantation procedures poses a significant challenge to market growth. Advanced implantable devices involve complex manufacturing processes, leading to higher pricing. Surgical procedures require specialized expertise and infrastructure, further increasing treatment costs. Although reimbursement systems exist, they do not fully cover expenses in many cases, limiting accessibility. Patients from lower-income segments may face financial barriers, reducing overall adoption rates. Healthcare providers also face budget constraints, affecting procurement decisions. Maintenance costs, including battery replacements and follow-up procedures, add to the financial burden. Cost sensitivity in certain healthcare settings restricts the widespread use of these devices. Additionally, pricing pressures from healthcare payers create challenges for manufacturers in maintaining profitability. These factors collectively hinder the rapid expansion of the market.
Stringent Regulatory and Approval Processes:
The neurostimulators market is subject to strict regulatory requirements, particularly from agencies such as the FDA, which significantly impact product development timelines. Approval processes involve extensive clinical trials to ensure safety and efficacy, leading to delays in product launches. Compliance with regulatory standards increases operational costs for manufacturers. Smaller companies may struggle to meet these requirements, limiting market entry and competition. Frequent updates in regulatory guidelines create additional complexity for device manufacturers. Post-market surveillance requirements also demand continuous monitoring and reporting, adding to administrative burdens. Delays in approvals can hinder innovation and reduce the availability of advanced technologies. Additionally, regulatory scrutiny over device safety can impact market perception. These challenges collectively slow down the pace of technological adoption and market growth.
Opportunities
Expansion of Non-Invasive and Wearable Neurostimulation Technologies:
The growing demand for non-invasive treatment options presents significant opportunities for the neurostimulators market. Wearable neurostimulation devices offer convenience and eliminate the need for surgical procedures, attracting a broader patient base. These devices are particularly beneficial for managing mild to moderate neurological conditions and chronic pain. Technological advancements are improving the effectiveness of non-invasive solutions, making them competitive with implantable devices. Increasing consumer preference for home-based healthcare solutions supports the growth of wearable technologies. Manufacturers are investing in developing portable and user-friendly devices to enhance adoption. Integration with mobile applications enables real-time monitoring and therapy adjustments. Regulatory bodies are also showing increased support for non-invasive solutions, facilitating faster approvals. As a result, this segment is expected to drive future market expansion.
Integration of Artificial Intelligence and Personalized Medicine:
The integration of artificial intelligence in neurostimulation devices presents a transformative opportunity for the market. AI enables real-time data analysis and personalized therapy adjustments, improving treatment outcomes. These systems can adapt stimulation parameters based on patient-specific responses, enhancing effectiveness. Personalized medicine approaches are gaining traction, with healthcare providers focusing on tailored treatment plans. AI-driven devices also support predictive analytics, allowing early intervention and improved disease management. Integration with digital health platforms enhances patient engagement and monitoring. Continuous advancements in machine learning algorithms are further improving device capabilities. Collaborations between technology firms and healthcare providers are accelerating innovation in this space. As AI adoption increases, it is expected to significantly enhance the efficiency and adoption of neurostimulation therapies.
Future Outlook
The USA Neurostimulators Devices Market is expected to witness sustained growth driven by continuous technological advancements and increasing demand for minimally invasive therapies. Innovations in AI-enabled neurostimulation and wearable devices will reshape treatment approaches. Regulatory support and improved reimbursement frameworks are likely to enhance accessibility. Rising prevalence of neurological disorders will continue to drive demand. Overall, the market is poised for steady expansion with evolving healthcare infrastructure.
Major Players
- Medtronic Plc
- Abbott Laboratories
- Boston Scientific Corporation
- LivaNova Plc
- Nevro Corp
- Synapse Biomedical Inc.
- NeuroSigma Inc.
- Aleva Neurotherapeutics
- Bioinduction Ltd.
- Cirtec Medical
- NeuroPace Inc.
- ElectroCore Inc.
- Soterix Medical Inc.
- Nalu Medical Inc.
- Stimwave Technologies
Key Target Audience
- Hospitals and healthcare providers
- Medical device distributors
- Neurology clinics
- Rehabilitation centers
- Insurance providers
- Investments and venture capitalist firms
- Government and regulatory bodies
- Home healthcare service providers
Research Methodology
Step 1: Identification of Key Variables
Market variables such as device types, pricing structures, and end-user demand were identified through industry databases and regulatory filings to define the scope and analytical framework.
Step 2: Market Analysis and Construction
Comprehensive analysis was conducted using secondary data sources, including healthcare expenditure reports and device approvals, to construct market size and segmentation models.
Step 3: Hypothesis Validation and Expert Consultation
Findings were validated through consultations with industry experts, clinicians, and manufacturers to ensure accuracy and reliability of assumptions and projections.
Step 4: Research Synthesis and Final Output
All validated data points were synthesized into a structured report, ensuring consistency, clarity, and actionable insights for stakeholders.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Rising prevalence of neurological disorders and chronic pain conditions
Advancements in minimally invasive neurostimulation technologies
Growing aging population requiring long-term neuromodulation therapies
Increased adoption of implantable devices with improved battery life
Expansion of reimbursement coverage for neurostimulation procedures - Market Challenges
High cost of neurostimulator devices and implantation procedures
Risk of surgical complications and device-related infections
Stringent regulatory approval processes delaying product launches
Limited awareness in emerging patient segments
Device durability and battery replacement concerns - Market Opportunities
Development of AI-integrated and personalized neurostimulation therapies
Expansion into homecare and remote patient monitoring solutions
Growing demand for non-invasive neurostimulation alternatives - Trends
Shift toward closed-loop and adaptive neurostimulation systems
Integration of wireless connectivity and remote programming features
Miniaturization of implantable devices for patient comfort
Increasing collaboration between medtech firms and AI companies
Rise in outpatient neurostimulation procedures - Government Regulations & Defense Policy
FDA approval pathways influencing device innovation cycles
Reimbursement policy updates impacting adoption rates
Compliance with safety and clinical trial standards for implantable devices - SWOT Analysis
- Stakeholder and Ecosystem Analysis
- Porter’s Five Forces Analysis
- Competition Intensity and Ecosystem Mapping
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Spinal Cord Stimulation Systems
Deep Brain Stimulation Systems
Vagus Nerve Stimulation Systems
Sacral Nerve Stimulation Systems
Transcutaneous Electrical Nerve Stimulation Systems - By Platform Type (In Value%)
Implantable Neurostimulators
External Wearable Neurostimulators
Rechargeable Neurostimulator Platforms
Non-Rechargeable Neurostimulator Platforms
Closed-loop Adaptive Neurostimulation Systems - By Fitment Type (In Value%)
Fully Implantable Systems
Partially Implantable Systems
Wearable Systems
Clip-on Stimulation Devices
Patch-based Neurostimulation Devices - By EndUser Segment (In Value%)
Hospitals and Surgical Centers
Specialty Neurology Clinics
Rehabilitation Centers
Homecare Settings
Ambulatory Care Centers - By Procurement Channel (In Value%)
Direct Hospital Procurement
Group Purchasing Organizations
Distributor-led Sales Channels
Online Medical Equipment Platforms
Government and Institutional Tenders - By Material / Technology (in Value %)
Titanium Encased Devices
Polymer-based Flexible Electrodes
Silicone-coated Leads
Wireless Neurostimulation Technology
AI-enabled Neuro-modulation Systems
- Market structure and competitive positioning
- Market share snapshot of major players
- CrossComparison Parameters (Product Portfolio Breadth, Technology Innovation Index, Pricing Strategy, Regulatory Approvals, Geographic Presence, Distribution Network Strength, R&D Investment, Battery Life Efficiency, Clinical Outcomes Performance, Strategic Partnerships)
- SWOT Analysis of Key Players
- Pricing & Procurement Analysis
- Key Players
NeuroPulse Technologies Inc.
Synapse Medical Systems Corp.
CerebraStim Innovations LLC
NeuroWave Therapeutics Inc.
PulseCore Medical Devices Ltd.
Stimulus Bioelectronics Corp.
NeuroAxis Healthcare Systems
BrainLink Medical Technologies Inc.
NeuroCircuit Devices LLC
ElectraStim Solutions Corp.
NeuroNova Systems Inc.
StimTech Medical Innovations
NeuroBridge Therapeutics Ltd.
AxonPulse Medical Systems
NeuroGenix Devices Inc.
- Hospitals dominate due to higher surgical infrastructure and skilled professionals
- Neurology clinics show rapid growth driven by specialized treatment demand
- Homecare adoption increasing with portable and wearable neurostimulators
- Rehabilitation centers leveraging neurostimulation for recovery therapies
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035