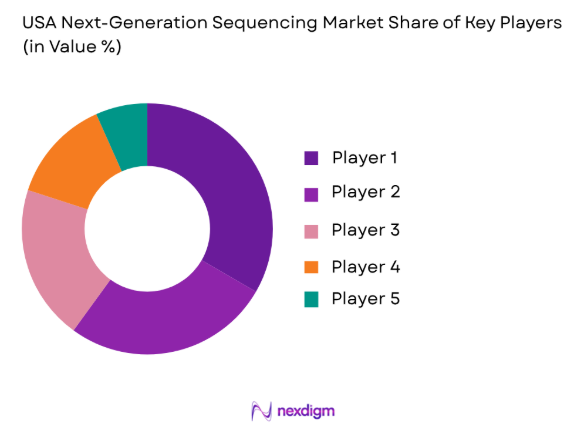
Market Overview
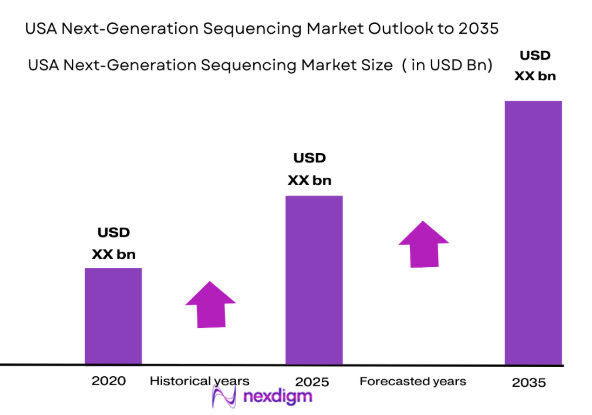
The USA Next-Generation Sequencing (NGS) market is valued at USD ~ billion in 2024, reflecting strong growth due to technological advancements and increasing demand for personalized medicine. The market is driven by declining sequencing costs, advances in bioinformatics, and the growing prevalence of genetic disorders and cancer. Key factors such as the expansion of genomic research, public funding initiatives, and the adoption of NGS in clinical diagnostics contribute to the market’s robust trajectory. The evolution of sequencing technologies, such as long-read and single-molecule sequencing, is further stimulating the market’s expansion.
The USA continues to dominate the NGS market, driven by major research hubs like Boston, San Francisco, and New York, which host some of the largest genomic research institutions and biotech companies. Government investments in genomic studies, such as the National Institutes of Health (NIH) funding for precision medicine, further solidify the country’s leadership. The extensive presence of tech giants like Illumina, Thermo Fisher Scientific, and Pacific Biosciences in these regions also contributes to the dominance, with strong infrastructure supporting R&D and clinical applications.
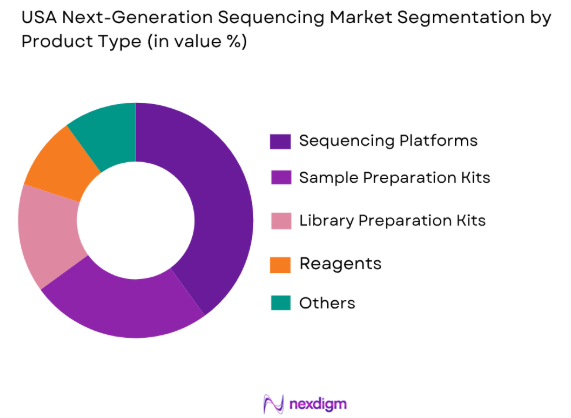
Market Segmentation
By Product Type
The USA Next-Generation Sequencing (NGS) market is segmented by product type into several categories, each serving a specific need in clinical and research applications. The most prominent product types in the market include sequencing reagents, sequencing platforms, consumables, and software solutions. Sequencing reagents are in high demand due to their pivotal role in sequencing workflows, facilitating high-quality DNA and RNA sequencing. Additionally, software solutions for bioinformatics are essential for data analysis, variant calling, and result interpretation. Sequencing platforms, particularly those based on Illumina’s sequencing by synthesis (SBS) technology, continue to dominate the market due to their reliability and scalability.
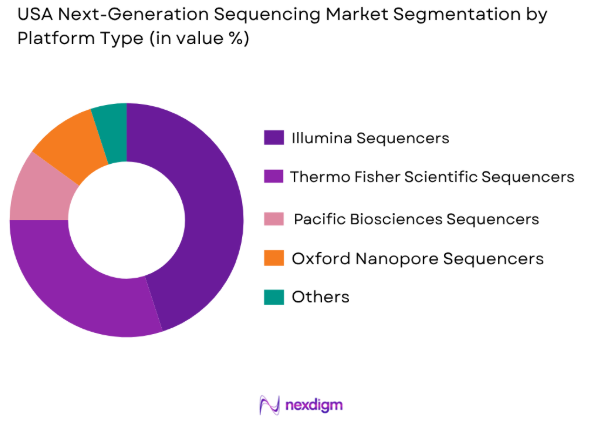
By Platform Type
USA Next-Generation Sequencing market is segmented by platform type into Illumina sequencers, Thermo Fisher Scientific sequencers, Pacific Biosciences sequencers, Oxford Nanopore Technologies sequencers, and others. Among these, Illumina sequencers hold a dominant market share due to their well-established reputation, widespread adoption, and comprehensive range of solutions for genomic analysis. Their scalability, high throughput capabilities, and continuous innovation in sequencing technologies have made Illumina sequencers the preferred choice for both research and clinical applications, thereby driving their leading market position.
Competitive Landscape
The USA NGS market is highly competitive, with a few major players leading the way in technology development, market share, and innovation. Dominant players like Illumina and Thermo Fisher Scientific provide a wide range of NGS platforms that cater to clinical, research, and diagnostic applications. Other significant players include Pacific Biosciences, BGI Genomics, and Oxford Nanopore Technologies. These companies hold a strong presence due to their significant investments in R&D, broad product portfolios, and strategic partnerships with healthcare providers and academic institutions.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD) |
| Illumina | 1998 | San Diego, CA, USA | ~ | ~ | ~ | ~ |
| Thermo Fisher Scientific | 2006 | Waltham, MA, USA | ~ | ~ | ~ | ~ |
| Pacific Biosciences | 2004 | Menlo Park, CA, USA | ~ | ~ | ~ | ~ |
| Oxford Nanopore Technologies | 2005 | Oxford, UK | ~ | ~ | ~ | ~ |
| BGI Genomics | 1999 | Shenzhen, China | ~ | ~ | ~ | ~ |
USA Next-Generation Sequencing Market Analysis
Growth Drivers
Clinical Adoption Acceleration
The clinical adoption of Next-Generation Sequencing (NGS) technologies has been significantly fueled by the rising prevalence of genetic disorders and cancers in the USA. In 2022, the Centers for Disease Control and Prevention (CDC) reported that approximately 1.9 million new cancer cases were diagnosed, driving demand for genomic sequencing in oncology. Furthermore, the American Cancer Society estimates that 1 in 2 men and 1 in 3 women will be diagnosed with cancer in their lifetime, further accelerating NGS adoption. This rising incidence underpins the growing importance of NGS for precision medicine, thus supporting its widespread clinical use.
Declining Cost-per-Genome & Read Accuracy Improvements
The cost of sequencing has decreased significantly over the years. As of 2023, the cost of sequencing a human genome had fallen to less than USD 1,000, a dramatic reduction from approximately USD 10 million in 2001. This cost reduction has been enabled by technological innovations and competition among NGS players. Additionally, improvements in read accuracy, including advancements in error correction algorithms, have enhanced the overall quality of sequencing results. This enables broader applications of NGS in clinical diagnostics and research, further driving market adoption. According to the National Institutes of Health (NIH), the improvements in accuracy have made NGS a cornerstone in cancer genomics.
Challenges
FDA Breakthrough Device Designations
The U.S. Food and Drug Administration (FDA) has granted breakthrough device designations to several NGS-based diagnostic technologies, which is catalyzing innovation and regulatory approval timelines. As of 2023, the FDA granted breakthrough status to a next-generation sequencing-based liquid biopsy technology for detecting cancer mutations. This designation facilitates faster review and approval processes for NGS products, ensuring that innovative diagnostic tools reach the market quicker. Such designations further validate the clinical utility of NGS technologies and expand their potential applications in diagnostics. This trend underscores the increasing regulatory acceptance of NGS and its growing role in healthcare.
CPT Code Evolution
The evolution of Current Procedural Terminology (CPT) codes for NGS testing is a critical trend in facilitating reimbursement for genomic tests. In 2023, new CPT codes were introduced by the American Medical Association (AMA) to account for multi-gene panels and whole-genome sequencing tests, making it easier for healthcare providers to get reimbursed for these advanced diagnostic services. The incorporation of NGS-specific codes ensures that the technology is more accessible and affordable to healthcare providers, driving broader clinical integration. This regulatory update is expected to boost the adoption of NGS testing in clinical settings, especially in oncology and genetic disease diagnostics.
Opportunities
Real-Time & Long-Read Sequencing Growth
Real-time sequencing technologies, particularly those utilizing nanopore sequencing, have gained traction in the market, offering significant potential for rapid genomic analysis. In 2023, the FDA provided regulatory clearance for the first real-time sequencing device, aimed at expanding its clinical diagnostic applications. This growth is further bolstered by long-read sequencing technologies, which allow for more accurate and comprehensive genome mapping. In comparison to traditional short-read sequencing, long-read technology can resolve complex genomic regions, which has been critical in applications such as structural variation detection. With increasing interest from academic research institutions, this technology holds promise for addressing key limitations in current sequencing methods.
Integration of AI/ML in Variant Calling & Interpretation
Artificial Intelligence (AI) and Machine Learning (ML) have become integral in the field of genomics, particularly for variant calling and interpretation. These technologies enable the analysis of vast amounts of genomic data, making it possible to identify genetic variations with higher precision and speed. In 2023, the NIH funded multiple projects focused on integrating AI and ML algorithms to improve the accuracy of NGS-based diagnostics. The success of these AI-powered tools in identifying clinically relevant variants has already seen adoption in various sectors, including oncology and rare disease diagnostics. AI’s potential to significantly reduce manual interpretation and improve clinical decision-making continues to drive NGS market growth.
Future Outlook
The USA Next-Generation Sequencing market is set to experience continued growth over the next several years, driven by technological innovations in sequencing platforms, increased demand for personalized medicine, and greater adoption of NGS in clinical diagnostics. As healthcare providers and research institutions continue to implement genomic sequencing technologies to improve patient outcomes and research efficacy, the market will likely expand further. Continued reductions in sequencing costs, improvements in sequencing accuracy, and emerging applications in various sectors, including agriculture and forensics, will ensure the USA’s dominance in the global NGS landscape.
Major Players
- Illumina, Inc.
- Thermo Fisher Scientific, Inc.
- Pacific Biosciences of California
- Oxford Nanopore Technologies
- BGI Genomics
- Qiagen N.V.
- PerkinElmer, Inc.
- Agilent Technologies, Inc.
- Takara Bio Inc.
- Genuity Science
- Invitae Corporation
- GeneDx (Subsidiary of BioReference)
- Fulgent Genetics
- Personalis, Inc.
- Roche Sequencing Solutions
Key Target Audience
- Investment and venture capital firms
- Government and regulatory bodies (FDA, CMS, NIH)
- Pharmaceutical and biotech companies
- Hospitals and clinical laboratories
- Research institutions and genomic laboratories
- Medical device manufacturers
- Health insurance companies and payer organizations
- Clinical diagnostics providers
Research Methodology
Step 1: Identification of Key Variables
In the first phase, the key variables driving the USA Next-Generation Sequencing market are identified. These include factors like advancements in sequencing technologies, regulatory policies, and application-specific drivers (oncology, genetic testing, etc.). Primary and secondary data collection methods are utilized to map out the key stakeholders and market dynamics.
Step 2: Market Analysis and Construction
We analyze historical and current data to construct an accurate market model. This involves segment-wise analysis, demand forecasting, and the evaluation of key trends within the clinical and research applications of NGS.
Step 3: Hypothesis Validation and Expert Consultation
In this phase, data hypotheses are validated by consulting industry experts. These consultations provide valuable insights into the operational and financial aspects of major players, ensuring that the data is both realistic and aligned with the market’s current and future trajectory.
Step 4: Research Synthesis and Final Output
The final phase involves synthesizing insights from the top players, market reports, and industry trends to develop a comprehensive market outlook. This includes validation through interviews with key market participants and analyzing consumer preferences, product usage, and technological innovations.
- Executive Summary
- Research Methodology (Definitions, Scope, and Standardizations Assumptions and Nomenclature, Data Collection Matrix, Market Sizing Approach, Triangulation and Forecast Validation, Limitations and Future Scope)
- Industry Structure & Evolution
- Historical Development and Innovation Curve
- Clinical vs Research Application Spectrum
- Workflow Value Chain
- Regulatory Landscape Overview
- Growth Drivers
Clinical Adoption Acceleration
Declining Cost-per-Genome & Read Accuracy Improvements
Increase in Public & Private Genomic Initiatives
Expansion of Cloud Bioinformatics Infrastructure - Challenges
CPT Code Evolution FDA
Breakthrough Device Designations - Opportunities
Real-Time & Long-Read Sequencing Growth
Integration of AI/ML in Variant Calling & Interpretation - Trends
FDA Breakthrough Device Designations
CPT Code Evolution
CLIA/CAP Compliance Impact - Government Regulations
- SWOT Analysis of Key Competitors
- Porter’s Five Forces
- By Total Addressable Market Analysis, 2020-2025
- By Serviceable Available Obtainable Market, 2020-2025
- By Market Dynamics: Adoption Rate Index, Utilization Density, 2020-2025
- By Product Type (In Value %)
Illumina Sequencers
Thermo Fisher Scientific Sequencers
Pacific Biosciences Sequencers
Oxford Nanopore Sequencers - By Platform Type (In Value %)
Sequencing Platforms
Sample Preparation Kits
Library Preparation Kits
Reagent - By End-User Type (In Value %)
Hospitals and Clinical Labs
Academic & Government Research Centers
Contract Research Organizations
Pharmaceutical & Biotech Companies
Direct-to-Consumer Testing Providers - By Service Model Type (In Value %)
In-house Sequencing Services
Outsourced NGS Services (CRO/CLIA Labs)
Hybrid Model (CapEx + Service Contracts) - By Sequencer Throughput Type (In Value %)
Low-Throughput
Mid-Throughput
High-Throughput
Ultra-High-Throughput - By Bioinformatics Solution Type (In Value %)
On-Premise Data Analysis
Cloud-Based Genomics Analytics
AI/ML Based Interpretation Software
Data Storage & Archival Services
- Market Share Analysis
Market Share by Technology Segment
Market Share by End-User Segment
Clinical vs Research Segment Share - Cross-Comparison Parameters (Corporate Profile & Business Model, Platform Portfolio Breadth, Installed Base & Replacement Cycle, R&D Intensity & Innovation Pipeline, Strategic Alliances & Channel Partnerships, Service & Support Network Coverage, Bioinformatics Ecosystem Integration, Pricing Strategy & Consumable Revenue Model, Time to Result Metrics, Regulatory Clearances & Clinical Claims, Patent Holdings & IP Position, Data Security & Compliance Capabilities, Customer Retention & Loyalty Metrics, Quality Certifications & Standards)
- Company Profiles
Illumina, Inc.
Thermo Fisher Scientific, Inc.
Pacific Biosciences of California, Inc.
Oxford Nanopore Technologies
BGI Genomics
Roche Sequencing Solutions
Qiagen N.V.
PerkinElmer, Inc.
Agilent Technologies, Inc.
Takara Bio Inc.
Genuity Science
Invitae Corporation
GeneDx
Fulgent Genetics
Personalis, Inc.
- Utilization Patterns by Institution Type
- Budget Allocation & Capital Investment Cycles
- Procurement Decision Drivers
- Pain Points
- Enterprise vs Academic Purchasing Behavior
- By Total Addressable Market Analysis, 2026-2035
- By Serviceable Available Obtainable Market, 2026-2035
- By Market Dynamics: Adoption Rate Index, Utilization Density, 2026-2035