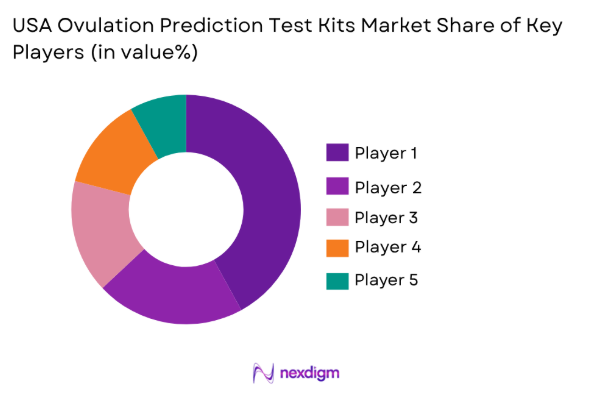
Market Overview
The USA Ovulation Prediction Test Kits market is valued at approximately USD ~ billion in 2025, driven by the increasing awareness of fertility issues and the growing demand for at-home fertility solutions. The market has been propelled by technological advancements in ovulation test kits, such as the integration of mobile apps, digital monitoring, and higher accuracy of test results. Additionally, consumer preference for non-invasive and privacy-focused methods of fertility tracking is contributing to the market’s growth. The expansion of retail and online sales channels also plays a significant role in making these products more accessible to a broader audience.
The USA dominates the Ovulation Prediction Test Kits market due to its large and diverse consumer base, supported by high healthcare awareness and the prevalence of fertility-related concerns among women. Cities such as New York, Los Angeles, and Chicago serve as key hubs due to their high population densities, access to healthcare services, and higher disposable income. Additionally, the presence of global healthcare and consumer goods companies in these regions contributes to market growth, with strong distribution networks and retail presence enhancing product availability and accessibility.
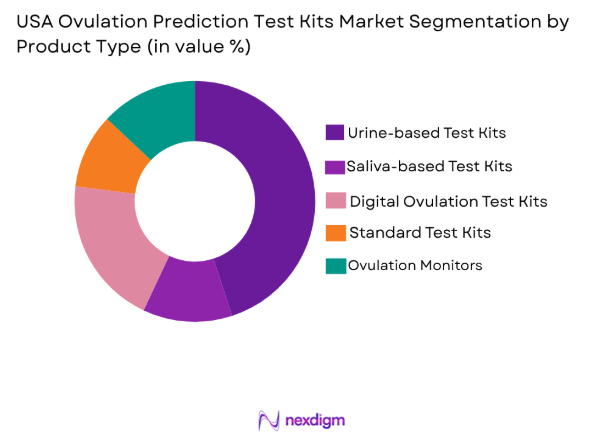
Market Segmentation
By Product Type
The USA Ovulation Prediction Test Kits market is segmented by product type into urine-based test kits, saliva-based test kits, digital ovulation test kits, standard test kits, and ovulation monitors. Among these, urine-based test kits currently hold the dominant market share due to their widespread use and ease of availability. These kits offer consumers a simple and cost-effective method for ovulation detection, which is key in driving their popularity. Brands like Clearblue and First Response have built strong brand loyalty and established trust with consumers through their reliable and accurate test results, further contributing to the dominance of this segment.
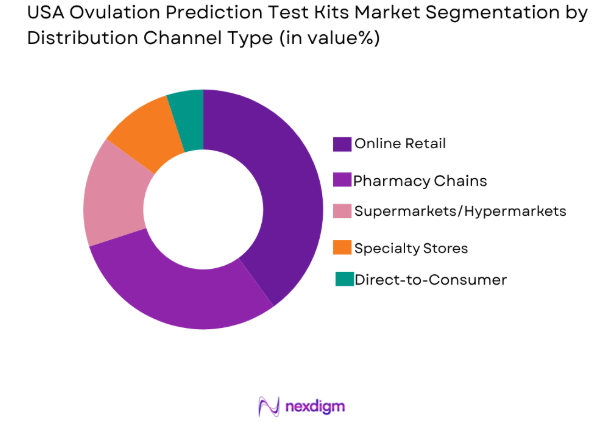
By Distribution Channel Type
The distribution of ovulation prediction test kits in the USA is primarily through online retail, pharmacy chains, supermarkets/hypermarkets, specialty stores, and direct-to-consumer models. Online retail has become a dominant channel, capturing a significant share due to its convenience, wider product variety, and the increasing trend of consumers purchasing health products online. Major players such as Amazon, Walgreens, and CVS have invested in e-commerce platforms to cater to the growing demand for at-home healthcare solutions, which has further fueled the growth of this segment.
Competitive Landscape
Over the next 5 years, the USA Ovulation Prediction Test Kits market is expected to show significant growth driven by increasing demand for fertility tracking solutions, technological advancements in ovulation test accuracy, and rising consumer awareness about reproductive health. The market will also benefit from the expansion of e-commerce platforms, facilitating wider accessibility to consumers. Additionally, the integration of artificial intelligence and machine learning in ovulation test kits, along with the rising preference for non-invasive methods, will further shape the future landscape of the market.
| Company | Establishment Year | Headquarters | Revenue | Product Range | Distribution Channels | Technology Used | Market Reach |
| Procter & Gamble (Clearblue) | 1837 | Cincinnati, Ohio, USA | ~ | ~ | ~ | ~ | ~ |
| Church & Dwight (First Response) | 1846 | Ewing, New Jersey, USA | ~ | ~ | ~ | ~ | ~ |
| EMD Serono (Merck) | 1668 | Darmstadt, Germany | ~ | ~ | ~ | ~ | ~ |
| AccuMed | 2000 | USA | ~ | ~ | ~ | ~ | ~ |
| Wondfo | 1994 | Guangzhou, China | ~ | ~ | ~ | ~ | ~ |
USA Ovulation Prediction Test Kits Market Analysis
Growth Drivers
Increasing Awareness of Fertility Issues
The increasing awareness of fertility issues is driving significant growth in the USA Ovulation Prediction Test Kits market. In recent years, there has been a surge in awareness about reproductive health, particularly among women aged 25-35. According to the U.S. Department of Health and Human Services, 1 in 8 couples in the U.S. experience fertility problems, with the National Center for Health Statistics reporting that approximately 12% of women of reproductive age have trouble getting or staying pregnant. As more women recognize the importance of early fertility monitoring, demand for ovulation prediction test kits continues to rise. Government initiatives promoting reproductive health, such as the Healthy People 2030 program, aim to reduce infertility rates, further amplifying the need for affordable and accessible fertility solutions.
Rising Adoption of Fertility Monitoring Technologies
The adoption of fertility monitoring technologies has been steadily increasing as more individuals turn to high-tech solutions for reproductive health management. As of 2022, over 15% of women in the U.S. actively use fertility tracking apps or devices, driven by advances in technology and the growing availability of non-invasive devices. According to a report from the National Institute of Health, the use of digital fertility monitoring devices has seen an uptick, with women increasingly relying on ovulation prediction kits for home-based testing. This trend is supported by the broader macroeconomic shift toward health-tech integration in daily life. Over 60% of consumers have adopted some form of digital health tool, further contributing to the growth of ovulation prediction test kits.
Market Challenges
High Cost of Advanced Test Kits
The high cost of advanced test kits poses a significant challenge to the growth of the Ovulation Prediction Test Kits market. While entry-level test kits remain affordable, the more advanced options, particularly those integrating digital monitoring and app connectivity, tend to have higher upfront costs. As of 2024, the average cost of a digital ovulation monitor can range from USD 100 to USD 250, which may deter lower-income consumers. According to the World Bank, approximately 30% of households in the U.S. earn less than USD 50,000 annually, which limits their ability to invest in such premium fertility products. This pricing discrepancy creates a barrier for many potential users, despite the increased demand for advanced ovulation tracking methods.
Lack of Awareness in Certain Demographics
Despite growing awareness, there is still a significant knowledge gap regarding ovulation prediction test kits in certain demographic groups. The U.S. Census Bureau highlights that many women, particularly those from lower socioeconomic backgrounds and rural areas, have limited access to fertility health education. Studies show that nearly 40% of women aged 20-39 in rural areas are unaware of ovulation prediction tests, which impacts their decision to use these products. Additionally, cultural factors and lack of healthcare resources contribute to lower adoption rates in specific communities. Government programs targeting reproductive health education are essential in bridging these gaps.
Market Opportunities
Growing Demand for At-Home Fertility Solutions
There is a growing demand for at-home fertility solutions, driven by consumer preference for privacy, convenience, and cost-effectiveness. According to a 2024 survey by the U.S. Centers for Disease Control and Prevention (CDC), 65% of women aged 25-40 are now opting for at-home health products, including fertility tracking tools, to manage reproductive health outside clinical settings. This shift is reflective of broader trends toward self-care, as evidenced by the growing popularity of other home health tests like blood glucose monitors and pregnancy tests. As home fertility solutions become more integrated with digital health apps, the demand for accurate, non-invasive, and easy-to-use ovulation prediction kits continues to rise. This presents a clear opportunity for market players to innovate and cater to the increasing consumer preference for at-home monitoring tools.
Technological Advancements in Test Accuracy
Technological advancements in test accuracy are revolutionizing the ovulation prediction test kits market. In 2024, several brands have introduced innovations, such as integrating artificial intelligence (AI) and machine learning into their ovulation monitors to offer more precise and personalized results. This trend aligns with the increasing consumer demand for highly accurate and easy-to-use fertility tracking devices. According to a study by the National Institutes of Health, the accuracy of digital ovulation prediction kits has improved by more than 20% in the past two years, making them more reliable for women seeking to optimize their chances of conception. As technology continues to evolve, these advancements create an opportunity to further penetrate the market by offering products that meet the growing demand for precision in reproductive health.
Future Outlook
Over the next 5 years, the USA Ovulation Prediction Test Kits market is expected to show significant growth driven by increasing demand for fertility tracking solutions, technological advancements in ovulation test accuracy, and rising consumer awareness about reproductive health. The market will also benefit from the expansion of e-commerce platforms, facilitating wider accessibility to consumers. Additionally, the integration of artificial intelligence and machine learning in ovulation test kits, along with the rising preference for non-invasive methods, will further shape the future landscape of the market.
Major Players
- Procter & Gamble (Clearblue)
- Church & Dwight (First Response)
- EMD Serono (Merck)
- AccuMed
- Wondfo
- Fairhaven Health
- Easy@Home (Zotek)
- Pregmate
- OV-Watch (Natural Cycles)
- BabyHopes
- Test Country
- CVS Health
- Rite Aid
- Amazon (Private Label)
- Walgreens
Key Target Audience
- Investments and Venture Capitalist Firms
- Healthcare Providers and Fertility Clinics
- Fertility Treatment Centers
- Pharmaceutical Companies
- Online Retail Platforms
- Government and Regulatory Bodies (FDA, CDC)
- Major Retail Chains
- Private Label Manufacturers (Amazon, Walmart
Research Methodology
Step 1: Identification of Key Variables
The first step maps the full U.S. ovulation prediction test kit ecosystem, including OTC manufacturers, digital fertility platforms, retailers, fertility clinics, and end users. Secondary research is used to identify the core market variables: product format, hormone detection method, digital connectivity, price architecture, and route-to-market. This stage also defines the market perimeter and excludes adjacent but non-core products.
Step 2: Market Analysis and Construction
Historical market construction is built from published U.S. revenue benchmarks, product-level category structures, and retailer-brand visibility. At this stage, the study analyzes how revenue is distributed between urine-based and advanced connected formats, and between personal-use and clinically linked pathways. Pricing ladders, replenishment patterns, and channel intensity are then used to refine category sizing.
Step 3: Hypothesis Validation and Expert Consultation
Working hypotheses are validated through interviews with brand managers, pharmacy buyers, e-commerce sellers, fertility specialists, and women’s health channel experts. These consultations test assumptions around usage frequency, repeat-purchase behavior, interpretation pain points, and adoption barriers for premium digital kits. The feedback is used to adjust segment weighting and demand assumptions.
Step 4: Research Synthesis and Final Output
The last phase synthesizes secondary evidence and interview findings into a final market narrative. Company positioning, product architecture, demand drivers, and channel priorities are cross-checked to ensure consistency between top-down and bottom-up views. The result is a market model designed for business decision-makers evaluating investment, expansion, partnerships, product launches, or category entry.
- Executive Summary
- Research Methodology (Market Definitions, Product Inclusion/Exclusion Criteria, OTC Diagnostic Scope, Historical Data Triangulation, Forecasting Framework, Top-Down Market Estimation, Bottom-Up Company Revenue Mapping, Channel Mapping, Pricing Benchmarking by SKU, Primary Interviews with Manufacturers/Distributors/Retailers/Clinics, Consumer Adoption Assessment, Limitations and Assumptions)
- Definition and Scope
- Product Genesis and Evolution of Home Ovulation Testing
- Industry Value Chain and Stakeholder Ecosystem
- U.S. Market Structure and Operating Model
- Product Lifecycle and Replacement Cycle
- Supply Chain Mapping from Manufacturing to Consumer
- Regulatory Landscape Snapshot
- Innovation Roadmap and Technology Transition
- Growth Drivers
Increasing Awareness of Fertility Issues
Rising Adoption of Fertility Monitoring Technologies - Market Challenges
High Cost of Advanced Test Kits
Lack of Awareness in Certain Demographics - Market Opportunities
Growing Demand for At-Home Fertility Solutions
Technological Advancements in Test Accuracy - Market Trends
Integration of Mobile Apps with Ovulation Kits
Increased Focus on Non-Invasive Fertility Tracking Solutions - Government Regulation
FDA Approval and Regulatory Standards
Reimbursement Policies - SWOT Analysis
- Stakeholder Ecosystem
- Porter’s Five Forces
- Competitive Landscape
- By Value,2020-2025
- By Volume,2020-2025
- By Average Price,2020-2025
- By Product Format (in Value%)
Urine-based Test Kits
Saliva-based Test Kits
Digital Ovulation Test Kits
Standard Test Kits
Ovulation Monitors - By Distribution Channel (In Value %)
Online Retail
Pharmacy Chains
Supermarkets/Hypermarkets
Specialty Stores
Direct-to-Consumer via Manufacturers - By Application (In Value %)
Women’s Health Clinics
Home Use
Fertility Treatment Centers
Healthcare Professionals - By Test Accuracy/ Sensitivity (In Value %)
High-Sensitivity Tests ( >99% Accuracy)
Medium-Sensitivity Tests (85-98% Accuracy)
Low-Sensitivity Tests (<85% Accuracy) - By End-User Demographics (In Value %)
Women Aged 18-30
Women Aged 31-40
Women Aged 40+
- Market Share of Major Players (by Value and Volume)
- Cross Comparison Parameters (Company Overview, Business Strategies, Recent Developments, Strength, Weakness, Organizational Structure, Revenues, Number of Distribution Channels, Pricing Models, Customer Reach, Brand Loyalty)
- SWOT Analysis of Major Players
- Pricing Analysis by SKU for Major Players in Ovulation Test Kits Market
- Detailed Profiles of Major Companies
Clearblue
First Response
Premom
Easy@Home
Proov
Pregmate
Natalist
Wondfo
ClinicalGuard
AccuMed
CVS Health
Walgreens
Rite Aid
up&up
Frida Fertility
- Consumer Journey Mapping
- TTC Behavior and Testing Protocols
- First-Time vs Repeat User Behavior
- Pain Points and Unmet Needs
- Decision-Making Factors in Brand Selection
- Role of Reviews, Communities, and Influencers
- App Usage, Data Logging, and Retention Behavior
- FSA/HSA and Affordability Considerations
- By Value, 2026-2035
- By Volume, 2026-2035
- By Average Price,2026-2035