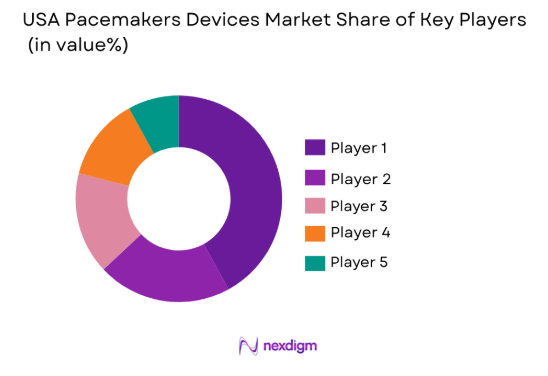
Market Overview
The USA Pacemakers Devices Market has shown significant growth, with a market size of USD ~ billion in recent assessments, driven by an aging population and increasing prevalence of cardiac diseases. Technological innovations, such as leadless pacemakers and wireless devices, are further propelling the market. Increased healthcare access and improved diagnostics have also contributed to the market’s expansion. Factors such as these are expected to continue fostering growth, supporting the adoption of advanced pacemaker technologies.
The United States holds dominance in the market due to advanced healthcare infrastructure, high adoption rates of new technologies, and well-established reimbursement policies for pacemaker procedures. Major cities like New York, Los Angeles, and Chicago, along with a strong presence of leading medical device manufacturers, contribute significantly to market growth. The country’s well-developed healthcare system, coupled with governmental support for cardiovascular care, further strengthens its position as the leader in the pacemaker device market.

Market Segmentation
By Product Type
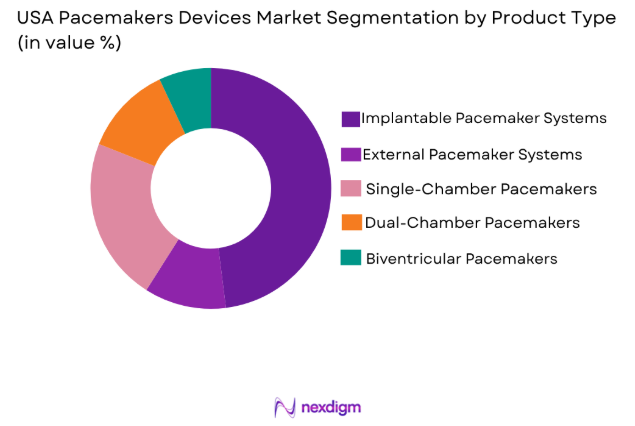
USA Pacemakers Devices Market is segmented by product type into Implantable Pacemaker Systems, External Pacemaker Systems, Single-Chamber Pacemakers, Dual-Chamber Pacemakers, and Biventricular Pacemakers. Recently, Implantable Pacemaker Systems have dominated the market due to their widespread adoption for long-term management of heart conditions, offering greater convenience for patients compared to external systems. Additionally, advances in battery life and device miniaturization have enhanced their appeal, leading to increased demand in hospitals and clinics.
By Platform Type
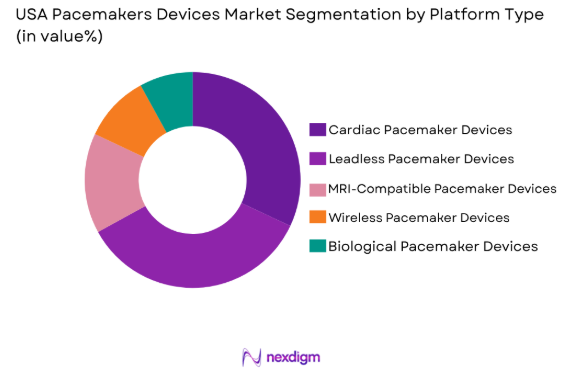
USA Pacemakers Devices Market is segmented by platform type into Cardiac Pacemaker Devices, Leadless Pacemaker Devices, MRI-Compatible Pacemaker Devices, Wireless Pacemaker Devices, and Biological Pacemaker Devices. Leadless Pacemaker Devices have gained substantial market share due to their compact size, reduced risk of infection, and the growing preference for minimally invasive procedures. These devices are particularly appealing to patients seeking alternatives to traditional pacemaker systems, which require leads and larger implantation sites.
Competitive Landscape
The competitive landscape of the USA Pacemakers Devices Market is marked by the presence of global leaders in medical device manufacturing, resulting in consolidation within the market. Major players in this industry drive innovation through strategic acquisitions, partnerships, and the development of next-generation pacemaker technologies. This has created a competitive environment, where differentiation in product features, regulatory approvals, and distribution networks plays a key role in capturing market share.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD) | Additional Parameter (R&D Investment) |
| Medtronic | 1949 | Minneapolis, MN | ~ | ~ | ~ | ~ | ~ |
| Boston Scientific | 1979 | Marlborough, MA | ~ | ~ | ~ | ~ | ~ |
| Abbott Laboratories | 1888 | Chicago, IL | ~ | ~ | ~ | ~ | ~ |
| Biotronik | 1963 | Berlin, Germany | ~ | ~ | ~ | ~ | ~ |
| MicroPort Scientific Corp | 2001 | Shanghai, China | ~ | ~ | ~ | ~ | ~ |
USA Pacemakers Devices Market Analysis
Growth Drivers
Aging Population and Increasing Cardiac Disease Incidence
The aging population is a significant growth driver for the USA Pacemakers Devices Market, as older adults are more likely to experience cardiac conditions such as arrhythmias and heart failure, which require pacemaker implantation. The increasing prevalence of cardiovascular diseases has also amplified the demand for pacemaker devices. Cardiovascular diseases are the leading cause of death in the U.S., and this health crisis has driven up the need for advanced pacemaker technologies to manage heart rhythm disorders. This growing demand for heart disease management solutions is expected to continue in the future, further accelerating the adoption of pacemaker systems. The increase in awareness about the benefits of pacemaker implantation also plays a critical role in driving market growth. Moreover, as the healthcare system becomes more equipped to handle heart-related illnesses, the reliance on pacemakers will continue to expand, driving the demand for these life-saving devices across the U.S.
Technological Advancements in Pacemaker Devices
Technological advancements in pacemaker devices have been a major contributor to the growth of the USA market. Innovations such as leadless pacemakers, which do not require leads for implantation, have gained significant attention due to their smaller size and reduced risk of complications. Additionally, the development of MRI-compatible pacemakers allows patients to undergo magnetic resonance imaging without removing the device, a key factor in their growing popularity. Furthermore, advances in battery technology and wireless pacemakers are driving the growth of the market by offering longer device life and enhanced patient comfort. These advancements in product features and functionality continue to make pacemakers a more viable option for patients, thus increasing their market penetration. As pacemaker technology continues to evolve, it is expected that the market will experience further growth and the development of even more efficient and minimally invasive devices.
Market Challenges
High Costs of Pacemaker Implantation
One of the major challenges facing the USA Pacemakers Devices Market is the high cost of pacemaker implantation. The initial cost of pacemaker systems, combined with the cost of surgery and post-implantation care, can be prohibitively expensive for many patients. This is a significant barrier for individuals without adequate insurance coverage or financial means, limiting market access. Even with insurance, high co-pays or deductibles can deter patients from opting for this procedure. Furthermore, the continued rise in healthcare costs in the U.S. has put additional pressure on both healthcare providers and patients, complicating access to pacemaker implantation. Despite the advancements in technology and the life-saving benefits of pacemakers, the cost remains a significant hurdle that could potentially slow market growth. Overcoming this challenge will require more cost-effective solutions and improved healthcare policies that address affordability.
Regulatory Hurdles and Long Approval Processes:
Regulatory hurdles and long approval processes present another significant challenge for the pacemaker market. The U.S. Food and Drug Administration (FDA) sets strict standards for the approval of pacemaker devices, and obtaining FDA clearance can take several years. This extended timeline delays the availability of new and innovative products to market, which in turn hinders competition and innovation. Furthermore, the complex approval process creates a barrier for smaller companies and startups, making it difficult for them to compete with established industry giants. As regulatory requirements continue to evolve, companies must navigate a complex landscape of testing, documentation, and clinical trials to bring new pacemaker devices to market. The lengthy approval process can delay the introduction of potentially life-saving devices and deter companies from investing in the pacemaker market, thus hindering overall market growth.
Opportunities
Technological Innovations in Leadless Pacemakers
Technological innovations in leadless pacemakers present a significant opportunity for growth in the USA Pacemakers Devices Market. Leadless pacemakers, which eliminate the need for leads and their associated complications, offer significant advantages such as reduced infection risk, smaller incision sites, and quicker recovery times. These devices also offer better patient comfort and have been shown to deliver comparable or superior outcomes to traditional pacemakers. The increasing demand for minimally invasive procedures and the growing preference for smaller, more discreet medical devices provide a strong foundation for the expansion of the leadless pacemaker market. With continued advancements in this area, leadless pacemakers are expected to capture a larger share of the pacemaker market in the U.S. Additionally, as more clinical evidence supports their safety and effectiveness, the adoption of leadless pacemakers is likely to accelerate, presenting a major opportunity for manufacturers.
Expansion into Emerging Markets
Another key opportunity for growth lies in the expansion of pacemaker technology into emerging markets. While the U.S. remains the largest market for pacemakers, developing countries with large populations, such as China, India, and Brazil, are witnessing an increase in the prevalence of cardiovascular diseases. As these markets expand their healthcare infrastructure, the demand for pacemaker devices is expected to rise significantly. Manufacturers can capitalize on this trend by entering these emerging markets with affordable and technologically advanced pacemaker solutions. Additionally, healthcare improvements, such as the expansion of government-backed healthcare programs and better access to cardiovascular treatments, are expected to drive demand for pacemakers in these regions. This presents a significant growth opportunity for companies looking to tap into new revenue streams and establish a strong presence in emerging markets.
Future Outlook
The USA Pacemakers Devices Market is poised for steady growth in the next five years, with technological innovations and the increasing prevalence of heart diseases driving demand. As healthcare systems continue to adopt advanced cardiac care technologies, including leadless and MRI-compatible pacemakers, the market will expand further. Additionally, regulatory support and improved reimbursement policies will contribute to the market’s growth. These factors, combined with growing awareness of heart disease management, are expected to ensure a positive outlook for the pacemaker device industry in the U.S.
Major Players
- Medtronic
- Boston Scientific
- Abbott Laboratories
- Biotronik
- MicroPort Scientific Corp
- Philips Healthcare
- Vitatron
- Medico
- LivaNova
- Eden Medical
- Implantable Device Technologies
- St. Jude Medical (acquired by Abbott)
- Biotronic
- Sorin Group (now LivaNova)
- Nihon Kohden
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Hospitals and healthcare providers
- Medical device distributors
- Healthcare insurance companies
- Cardiovascular research institutions
- Medical technology investors
Research Methodology
Step 1: Identification of Key Variables
The first step involves identifying the key variables that will drive market growth, including technological advancements, market trends, and regulatory factors.
Step 2: Market Analysis and Construction
We conduct a thorough market analysis, including segmenting the market by product type, platform type, and key geographical regions, to build a comprehensive market model.
Step 3: Hypothesis Validation and Expert Consultation
Our market model is validated through consultations with industry experts, including key players, researchers, and regulatory bodies, to ensure accuracy.
Step 4: Research Synthesis and Final Output
The final output synthesizes all findings from the market analysis, expert feedback, and data collection into a comprehensive market report.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Aging Population and Increased Incidence of Cardiac Diseases
Technological Advancements in Pacemaker Devices
Rising Awareness and Access to Healthcare - Market Challenges
High Cost of Pacemaker Implantation
Regulatory and Certification Barriers
Complications and Risks Associated with Pacemaker Systems - Market Opportunities
Technological Innovations in Pacemaker Devices
Emerging Markets and Untapped Regions
Government Initiatives and Healthcare Investments - Trends
Increasing Adoption of Leadless Pacemakers
Growth in Minimally Invasive Procedures - Government Regulations
- SWOT Analysis
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Implantable Pacemaker Systems
External Pacemaker Systems
Single-Chamber Pacemakers
Dual-Chamber Pacemakers
Biventricular Pacemakers - By Platform Type (In Value%)
Cardiac Pacemaker Devices
Leadless Pacemaker Devices
MRI-Compatible Pacemaker Devices
Wireless Pacemaker Devices
Biological Pacemaker Devices - By Fitment Type (In Value%)
Transvenous Pacemaker Systems
Epicardial Pacemaker Systems
Leadless Pacemaker Systems
Temporary Pacemaker Systems - By End User Segment (In Value%)
Hospitals
Ambulatory Surgical Centers
Cardiac Care Clinics
- Growth Drivers
Aging Population and Increased Incidence of Cardiac Diseases
Technological Advancements in Pacemaker Devices
Rising Awareness and Access to Healthcare - Market Challenges
High Cost of Pacemaker Implantation
Regulatory and Certification Barriers
Complications and Risks Associated with Pacemaker Systems - Market Opportunities
Technological Innovations in Pacemaker Devices
Emerging Markets and Untapped Regions
Government Initiatives and Healthcare Investments - Trends
Increasing Adoption of Leadless Pacemakers
Growth in Minimally Invasive Procedures - Government Regulations
- SWOT Analysis
- Porter’s Five Forces
- Hospitals and Cardiac Centers
- Ambulatory Surgical Centers
- Long-Term Care Facilities
- Rehabilitation Centers
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035