Market Overview
The USA PCR Machines Equipment Market is a rapidly evolving sector driven by advancements in diagnostic technologies and increased demand for precise genetic testing. Based on a recent historical assessment, the market size reached USD ~ billion, with growth fueled by innovations in PCR technology and a surge in demand for disease diagnosis. A rising focus on personalized medicine, the COVID-19 pandemic’s legacy, and growing healthcare spending have been central to the market’s momentum. The market has seen substantial investment from healthcare providers, research institutions, and diagnostic companies, ensuring continuous improvements and innovations in PCR systems.
In terms of regional dominance, the USA PCR Machines Equipment Market is largely driven by key urban centers such as New York, Los Angeles, and Chicago. These cities serve as major healthcare hubs with extensive infrastructure and strong healthcare spending, propelling demand for PCR technology. Furthermore, states like California and Massachusetts are known for their robust healthcare research industries, facilitating the growth of the market. In addition to urban dominance, favorable government policies, such as reimbursement for diagnostic procedures, continue to shape the market dynamics in these regions.
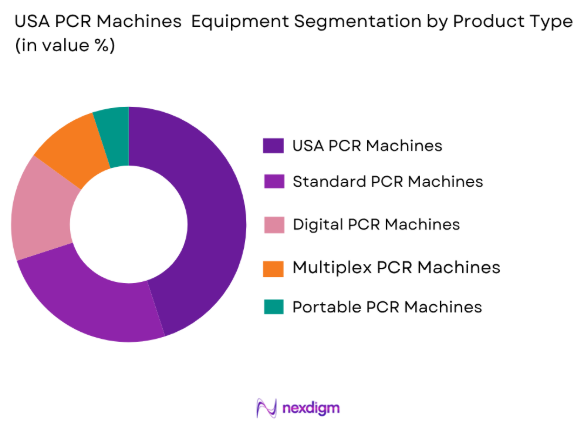
Market Segmentation
By Product Type
USA PCR Machines Equipment Market is segmented by product type into real-time PCR machines, standard PCR machines, digital PCR machines, multiplex PCR machines, and portable PCR machines. Recently, real-time PCR machines have a dominant market share due to their ability to offer rapid results and greater precision in molecular diagnostics. With increasing demand for point-of-care testing, real-time PCR systems have become the preferred choice in clinical diagnostics, research, and infectious disease detection. Their ability to deliver high sensitivity and specificity, along with reduced turnaround time, makes them suitable for both small and large-scale testing. Additionally, real-time PCR machines have gained popularity due to their versatility in detecting a wide range of pathogens, including viruses and bacteria, making them a valuable tool during outbreaks such as the COVID-19 pandemic.
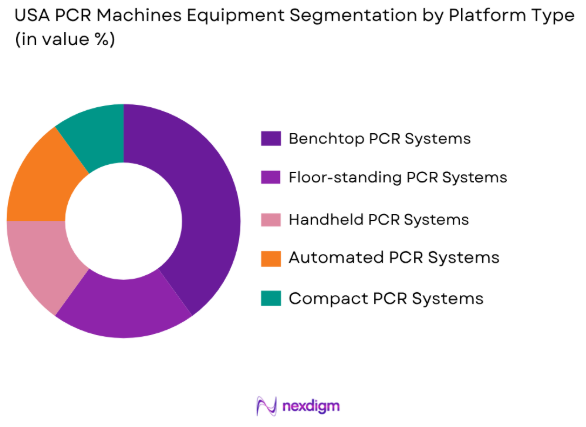
By Platform Type
USA PCR Machines Equipment Market is segmented by platform type into benchtop PCR systems, floor-standing PCR systems, handheld PCR systems, automated PCR systems, and compact PCR systems. Recently, benchtop PCR systems have dominated the market due to their balance of cost-effectiveness, performance, and space efficiency, making them highly suitable for laboratory settings. Benchtop systems are designed for use in both clinical and research laboratories, offering high throughput and ease of use. Their popularity can be attributed to their adaptability in various fields, including diagnostics, research, and testing in laboratories with limited space. Additionally, the growing trend toward more user-friendly, automated systems has also fueled the adoption of benchtop PCR machines in a wide range of laboratory environments.
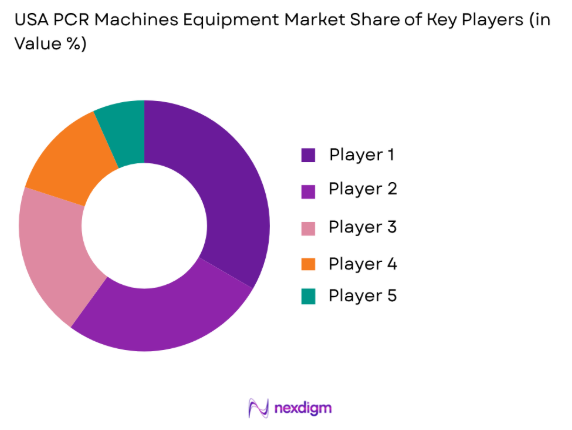
Competitive Landscape
The competitive landscape of the USA PCR Machines Equipment Market is characterized by significant consolidation, with leading players driving technological advancements and market penetration. Major players have expanded their portfolios by acquiring smaller companies or forming strategic alliances, ensuring continuous innovation and customer loyalty. Established companies leverage their R&D capabilities, extensive distribution networks, and broad customer bases to maintain dominance. Competitive pressure is also seen from smaller firms specializing in niche markets, particularly in mobile and portable PCR systems. These companies challenge the status quo by offering cost-effective, cutting-edge solutions suited for small-scale operations and point-of-care testing environments.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD) | Technology Integration |
| Thermo Fisher Scientific | 1956 | Waltham, Massachusetts | ~ | ~ | ~ | ~ | ~ |
| Bio-Rad Laboratories | 1952 | Hercules, California | ~ | ~ | ~ | ~ | ~ |
| Agilent Technologies | 1999 | Santa Clara, California | ~ | ~ | ~ | ~ | ~ |
| Qiagen | 1984 | Hilden, Germany | ~ | ~ | ~ | ~ | ~ |
| Roche Diagnostics | 1896 | Basel, Switzerland | ~ | ~ | ~ | ~ | ~ |
USA PCR Machines Equipment Market Analysis
Growth Drivers
Increasing Diagnostic Demand in Healthcare
The demand for diagnostic testing in healthcare is increasing rapidly due to the rising global burden of diseases and the growing need for early diagnosis. In the U.S., healthcare spending on diagnostic services exceeded $90 billion in 2022, with a significant portion driven by the need for accurate and efficient diagnostic tools like PCR machines. The U.S. Centers for Disease Control and Prevention (CDC) reported a surge in diagnostic testing during the COVID-19 pandemic, which further highlights the demand for PCR-based diagnostics, a trend expected to continue in response to ongoing health challenges.
Advancements in PCR Technology
The continuous advancements in PCR technology are driving its adoption in various healthcare applications. Real-time PCR (qPCR) machines, which provide faster and more accurate results, are increasingly used in both clinical and research settings. According to the U.S. Food and Drug Administration (FDA), in 2022, there was an increase in the approval of PCR-based diagnostic tests due to their enhanced accuracy and ability to detect genetic material of pathogens with greater sensitivity. This technological evolution has made PCR a critical tool in the diagnosis of a wide range of diseases, such as cancer and infectious diseases.
Challenges
High Capital Investment and Maintenance Costs
The high upfront cost and ongoing maintenance expenses for PCR machines pose a challenge for widespread adoption, especially in low-resource settings. In 2023, the average cost for a high-end PCR machine in the U.S. was around $40,000, and the cost of consumables and reagents adds to the financial burden. The U.S. National Institutes of Health (NIH) reports that laboratory equipment costs, including PCR machines, account for a significant portion of research funding, often limiting access to these critical tools in academic and diagnostic settings. This is a major barrier to the broader use of PCR technology, particularly in underfunded healthcare systems.
Regulatory and Compliance Barriers
Regulatory and compliance barriers for PCR machines are a key challenge, particularly regarding the approval and standardization of diagnostic tests. The U.S. FDA regulates PCR-based tests, which requires manufacturers to adhere to strict guidelines before market approval. As of 2023, more than 100 PCR test kits were waiting for FDA emergency use authorization during the ongoing infectious disease threats, causing delays in product availability. These regulatory hurdles increase costs and extend the time to market, slowing the adoption of innovative PCR machines and tests.
Opportunities
Expanding Demand for Point-of-Care Testing
The demand for point-of-care (POC) testing is growing, particularly in remote areas where access to centralized labs is limited. In 2022, the U.S. Department of Health and Human Services (HHS) reported a significant rise in the use of POC PCR devices during the COVID-19 pandemic, with millions of tests being conducted at the point of care. This growth is supported by the ongoing need for quick, on-site diagnostics in both healthcare settings and for public health surveillance, creating a substantial opportunity for the expansion of PCR technologies in the POC market.
Increasing Use of PCR in Precision Medicine and Personalized Healthcare
PCR technology is becoming increasingly important in precision medicine, where genetic information is used to tailor treatments to individual patients. The U.S. National Cancer Institute (NCI) has highlighted the role of PCR in detecting genetic mutations in cancer cells, enabling targeted therapies. As of 2023, more than 100,000 cancer patients in the U.S. received personalized treatments based on genetic testing, a practice that increasingly relies on PCR. The rising trend of personalized medicine presents a growing opportunity for PCR machines in clinical settings, driving demand for accurate and reliable diagnostics.
Future Outlook
The USA PCR Machines Equipment Market is expected to continue its growth trajectory over the next five years, driven by ongoing technological advancements, increasing demand for diagnostic accuracy, and a growing focus on genetic testing. New innovations, including the integration of artificial intelligence with PCR systems, will enhance their capabilities and streamline the testing process. As regulatory support continues to evolve, especially in areas such as point-of-care testing and genetic diagnostics, the market will see increased opportunities for expansion. Furthermore, the increasing use of PCR systems for various applications, including infectious disease detection, oncology, and personalized medicine, will further fuel market growth.
Major Players
- ThermoFisher Scientific
- Bio-Rad Laboratories
- Agilent Technologies
- Qiagen
- Roche Diagnostics
- Abbott Laboratories
- PerkinElmer
- Becton Dickinson
- SABiosciences
- Fisher Scientific
- Takara Bio
- Stilla Technologies
- GenetixBiotech
- Microsynth
- Illumina
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Hospitals and healthcare facilities
- Medical device distributors
- Pharmaceutical companies
- Diagnostic laboratories
- Research institutions
- Private healthcare providers
Research Methodology
Step 1: Identification of Key Variables
The first step involves identifying critical market variables, including technological trends, regulatory frameworks, and consumer preferences that affect market growth.
Step 2: Market Analysis and Construction
In this phase, an in-depth analysis of the PCR machines market is conducted, focusing on factors such as current market size, growth trends, and segmentation.
Step 3: Hypothesis Validation and Expert Consultation
Expert consultations are carried out to validate assumptions and hypotheses about market trends, customer preferences, and competitive dynamics.
Step 4: Research Synthesis and Final Output
The findings from analysis and expert consultation are synthesized into actionable insights, forming the final output for strategic recommendations.
- Executive Summary
- Research Methodology (Market Definitions and Assumptions, Abbreviations, Market Sizing Approach, Data Collection Framework, Industry Interviews, Primary Research Insights, Limitations, and Forecast Validation)
- Market Definition and Scope
- PCR Technology Evolution and Industry Genesis
- Overview of PCR Applications in Diagnostics
- Regulatory Landscape in the USA
- Market Value Chain and Supply Chain Insights
- Growth Drivers
Increasing Diagnostic Demand in Healthcare
Advancements in PCR Technology
Rising Prevalence of Infectious Diseases
Government and Private Sector Investments in Diagnostics - Market Challenges
High Capital Investment and Maintenance Costs
Regulatory and Compliance Barriers
Supply Chain Constraints and Equipment Availability
Intense Competition in the PCR Market - Market Opportunities
Expanding Demand for Point-of-Care Testing
Increasing Use of PCR in Precision Medicine and Personalized Healthcare
Growth in Agricultural and Environmental PCR Applications
Technological Innovations and Product Developments - Trends
Adoption of Automated PCR Systems
Miniaturization and Portability of PCR Machines
Increasing Integration with AI for Diagnostics and Monitoring
Rise of Multiplex PCR for Simultaneous Pathogen Detection - Government Regulations
- SWOT Analysis of Major Players
- Pricing and Profitability Analysis for Major Players
- Porter’s Five Forces
- By Value, 2020-2025
- By Volume, 2020-2025
- By Average Selling Price Trends, 2020-2025
- By Market Share, 2020-2025
- By Product (In Value%)
Real-time PCR Machines
Standard PCR Machines
Digital PCR Machines
Multiplex PCR Machines
Portable PCR Machines - By Platform (In Value%)
Benchtop PCR Systems
Floor-standing PCR Systems
Handheld PCR Systems
Automated PCR Systems
Compact PCR Systems - By End-User (In Value%)
Hospitals and Clinics
Diagnostic Laboratories
Biotech and Pharmaceutical Companies
Academic and Research Institutions - By Distribution Channel (In Value%)
Direct Sales
Healthcare Distribution Networks
Online Sales Platforms - By Region (In Value%)
Northeast USA
Midwest USA
Southern USA
Western USA
- Market Share of Major Players
- Market Share by Value & Volume
- Share Distribution by Product Type
- Cross-Comparison Parameters(Company Overview, Business Strategies, Product Portfolios, Geographic Reach, R&D Investment, Pricing Strategies, Distribution Networks, Manufacturing Capacities)
- Profiles of Major Companies
Thermo Fisher Scientific
Bio-Rad Laboratories
Agilent Technologies
QIAGEN
Abbott Laboratories
Illumina
Promega Corporation
Merck KGaA
PerkinElmer
Takara Bio
Becton, Dickinson and Company
Roche Diagnostics
F. Hoffmann-La Roche AG
Applied Biosystems
Hudson Robotics
- Hospitals and Healthcare Institutions
- Diagnostic Laboratories
- Biotech and Pharmaceutical Companies
- Academic and Research Institutions
- Public Health and Government Agencies
- By Value, 2026-2035
- By Volume, 2026-2035
- By Average Selling Price Trends, 2026-2035
- By Market Share, 2026–2035