Market Overview
The USA Predictive Biomarkers Market is valued at approximately USD ~ billion based on a recent historical assessment. This market growth is primarily driven by advancements in genomic technologies and increased demand for personalized medicine. The ability of predictive biomarkers to offer early detection and prognosis of diseases such as cancer, cardiovascular, and neurodegenerative disorders significantly contributes to this expansion. Increasing adoption of biomarkers in clinical settings and research further boosts their market presence in the healthcare and pharmaceutical industries.
The USA dominates the global predictive biomarkers market due to its strong healthcare infrastructure, high demand for personalized medicine, and significant R&D investments in biotechnology. Major cities like New York, Boston, and San Francisco lead in research activities and clinical trials, fostering the development and adoption of predictive biomarkers. A favorable regulatory environment, combined with the growing number of academic and commercial collaborations, positions the USA as a leader in the market. Additionally, the presence of numerous biotech and pharmaceutical companies accelerates innovation and product development in this sector.
Market Segmentation
By Product Type
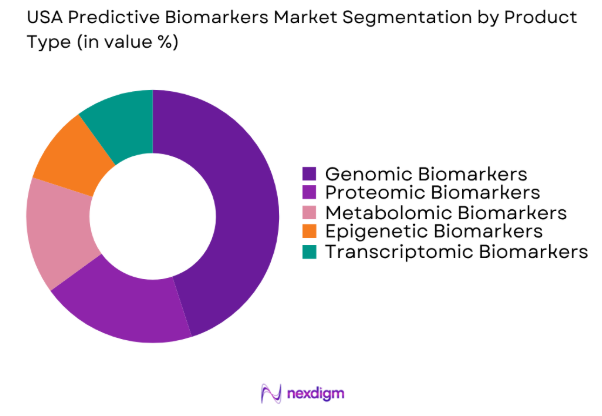
The USA Predictive Biomarkers market is segmented by product type into genomic biomarkers, proteomic biomarkers, metabolomic biomarkers, epigenetic biomarkers, and transcriptomic biomarkers. Recently, genomic biomarkers have dominated the market due to the increasing prevalence of cancer and the role genomic analysis plays in early disease detection and personalized treatments. Advancements in next-generation sequencing (NGS) technologies and the expanding application of genomic biomarkers in clinical trials have bolstered their dominance.
By Platform Type
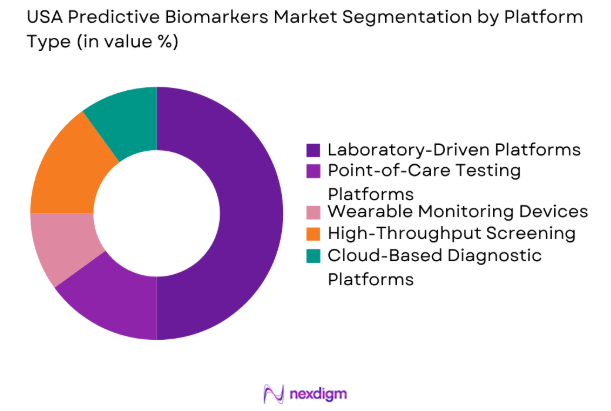
The USA Predictive Biomarkers market is segmented by platform type into laboratory-driven platforms, point-of-care testing platforms, wearable monitoring devices, high-throughput screening platforms, and cloud-based diagnostic platforms. Laboratory-driven platforms currently hold the largest market share, mainly due to their ability to provide high-precision results in clinical research and diagnostics. These platforms offer integrated solutions that support multiple types of biomarkers, enhancing their use in large-scale clinical trials and diagnostics.
Competitive Landscape
The USA Predictive Biomarkers market is highly competitive, with a blend of established companies and emerging biotech firms. These players are involved in the development of new biomarkers and technologies, which is driving market consolidation and innovation. The influence of major players is considerable, as they lead in the advancement of diagnostic and therapeutic biomarker solutions. Companies that can integrate advanced technologies and form strategic partnerships will likely maintain a competitive edge in this growing market.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue |
| Thermo Fisher Scientific | 1956 | Waltham, MA, USA | ~ | ~ | ~ | ~ |
| Illumina | 1998 | San Diego, CA, USA | ~ | ~ | ~ | ~ |
| Bio-Rad Laboratories | 1952 | Hercules, CA, USA | ~ | ~ | ~ | ~ |
| QIAGEN | 1984 | Hilden, Germany | ~ | ~ | ~ | ~ |
| Roche Diagnostics | 1896 | Basel, Switzerland | ~ | ~ | ~ | ~ |
USA Predictive Biomarkers Market Analysis
Growth Drivers
Technological Advancements in Biomarker Development
The continuous advancements in biotechnology and genomic technologies are driving the growth of predictive biomarkers. Innovations such as next-generation sequencing (NGS) and advanced bioinformatics are significantly improving the sensitivity and specificity of biomarkers, which are now crucial for early disease detection and personalized treatment strategies. These technological breakthroughs not only enhance diagnostic accuracy but also broaden the application of biomarkers across various clinical settings. As a result, predictive biomarkers are becoming increasingly essential for managing chronic diseases like cancer, cardiovascular disorders, and neurodegenerative conditions. Their ability to offer tailored treatments and monitor disease progression is revolutionizing healthcare and improving patient outcomes.
Increase in Chronic Disease Incidence
The rising prevalence of chronic diseases like cancer, diabetes, and cardiovascular conditions is a major factor fueling the demand for predictive biomarkers. Early detection and precise monitoring are essential for managing these diseases, and biomarkers play a vital role in this process. By identifying individuals at high risk, predictive biomarkers help healthcare professionals develop personalized treatment plans and monitor disease progression more effectively. This enables optimized interventions, improves patient outcomes, and enhances the efficiency of healthcare systems. As chronic diseases continue to increase, the demand for predictive biomarkers will grow, making them an integral part of modern healthcare and driving the expansion of the market.
Market Challenges
Regulatory Hurdles in Biomarker Approval
A significant challenge for the predictive biomarkers market is the stringent regulatory processes required for the approval of new biomarker-based diagnostic tools. Regulatory bodies, such as the FDA, mandate comprehensive testing and validation before these products can be commercialized. This lengthy approval process can lead to delays in bringing innovative biomarker solutions to market, which hampers the timely adoption of cutting-edge technologies. As a result, companies face obstacles in rapidly responding to market needs, potentially slowing market growth and limiting the availability of new diagnostic tools that could enhance disease detection and personalized treatment strategies. The regulatory burden remains a key barrier to market expansion.
High Cost of Biomarker Development and Implementation
The development and implementation of predictive biomarkers require significant financial investment in research, technology integration, and clinical trials. These high costs can make it challenging for companies to bring new biomarker-based diagnostic tools to market, limiting their affordability and accessibility, particularly in low-resource settings. Smaller institutions and healthcare providers may struggle to invest in the necessary infrastructure and technology to adopt these advancements, slowing down their widespread use. The financial barriers to entry can also limit the pace at which these technologies are integrated into clinical practices, delaying their potential to improve disease detection, monitoring, and personalized treatment. As a result, these cost challenges hinder the broader adoption and commercialization of predictive biomarker solutions.
Opportunities
Rise of Personalized Medicine
Personalized medicine is increasingly popular as it enables tailored treatment plans based on individual genetic profiles and biomarker data. This trend is driving the integration of predictive biomarkers into therapeutic strategies, creating substantial growth opportunities in the market. As genetic research advances and our understanding of disease mechanisms improves, the demand for biomarkers that can guide customized treatment approaches is growing. These biomarkers are essential for developing more effective, individualized therapies, particularly in areas such as oncology, cardiology, and neurology. The shift towards personalized healthcare is enhancing the role of predictive biomarkers, positioning them as a key component of modern treatment regimens and further expanding their market potential.
Expansion of Point-of-Care Diagnostic Solutions
The increasing demand for rapid and cost-effective diagnostics at the point of care presents a significant opportunity for predictive biomarkers. By integrating biomarkers into point-of-care testing platforms, healthcare providers can achieve quicker diagnoses and make faster treatment decisions. This capability is especially valuable in regions with limited access to specialized healthcare facilities, where timely diagnostics can greatly improve patient outcomes. The ability to provide accurate, real-time results in such settings makes predictive biomarkers essential for enhancing healthcare delivery. As point-of-care diagnostics continue to expand, the integration of biomarkers will play a crucial role in transforming healthcare practices, driving market growth, and improving access to advanced diagnostic solutions in underserved areas.
Future Outlook
The USA Predictive Biomarkers Market is expected to experience significant growth over the next five years, driven by continued advancements in technology, expanding healthcare infrastructure, and rising consumer demand for personalized healthcare solutions. As the regulatory environment adapts to support faster approval of innovative biomarker-based products, more opportunities will arise for market players. The increasing integration of predictive biomarkers in clinical settings, particularly in oncology, cardiology, and neurology, will further fuel market growth. Additionally, innovations in AI and machine learning will enhance the predictive power of biomarkers, making them an even more integral part of healthcare systems.
Major Players
- Thermo Fisher Scientific
- Illumina
- Bio-Rad Laboratories
- QIAGEN
- Roche Diagnostics
- PerkinElmer
- Abbott Laboratories
- Agilent Technologies
- Becton Dickinson
- Siemens Healthineers
- GE Healthcare
- Danaher Corporation
- Merck & Co.
- BioMérieux
- Stryker Corporation
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Pharmaceutical companies
- Biotech companies
- Healthcare providers
- Diagnostic laboratories
- Research and development institutions
- Healthcare technology firms
Research Methodology
Step 1: Identification of Key Variables
In this step, key market drivers, challenges, trends, and technological advancements are identified to frame the scope of the market study.
Step 2: Market Analysis and Construction
A thorough analysis is conducted to evaluate the current market conditions, including segmentation, competitive landscape, and market dynamics. Data is collected from primary and secondary sources.
Step 3: Hypothesis Validation and Expert Consultation
Market hypotheses are validated through consultations with industry experts, ensuring the research is grounded in real-world insights and data.
Step 4: Research Synthesis and Final Output
Data and insights gathered are synthesized to provide a comprehensive analysis of the market, concluding with actionable insights and recommendations for stakeholders.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Technological Advancements in Diagnostics
Increase in Chronic Disease Prevalence
Rising Demand for Personalized Medicine - Market Challenges
Regulatory Barriers and Approval Delays
High Costs of Development and Implementation
Data Privacy Concerns - Market Opportunities
Expansion of Biomarker Applications in Drug Development
Growth in Point-of-Care Diagnostics
Partnerships and Collaborations - Trends
Integration of Artificial Intelligence (AI) and Machine Learning (ML)
Shift Toward Non-Invasive Biomarkers
Growing Use of Biomarkers in Early Disease Detection - Government Regulations
- SWOT Analysis of Key Competitors
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Genomic Biomarkers
Proteomic Biomarkers
Metabolomic Biomarkers
Epigenetic Biomarkers
Transcriptomic Biomarkers - By Platform Type (In Value%)
Laboratory-Driven Platforms
Point-of-Care Testing Platforms
Wearable Monitoring Devices
High-Throughput Screening Platforms
Cloud-Based Diagnostic Platforms - By Fitment Type (In Value%)
In-Vivo Fitment
In-Vitro Fitment
Ex-Vivo Fitment
Implantable Biomarker Systems
Non-Invasive Fitment - By End User Segment (In Value%)
Hospitals
Diagnostic Laboratories
Research & Development Institutes
Pharmaceutical Companies
Biotech Companies - By Procurement Channel (In Value%)
Direct Sales
Online Retailers
Distributors and Dealers
Government and Institutional Procurement
Third-Party Resellers
- Market Share Analysis
- Cross Comparison Parameters (Technology Integration, Market Reach, Regulatory Compliance, Product Innovation, Customer Base, Pricing Strategy, Distribution Channels, Partnerships & Collaborations, Brand Reputation, R&D Investment)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Thermo Fisher Scientific
Illumina, Inc.
Bio-Rad Laboratories
Roche Diagnostics
Abbott Laboratories
Agilent Technologies
QIAGEN
PerkinElmer
Becton Dickinson
Merck & Co.
Siemens Healthineers
GE Healthcare
Danaher Corporation
BioMérieux
Stryker Corporation
- Hospitals
- Diagnostic laboratories
- Pharmaceutical and biotech companies
- Research institutions
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035


