Market Overview
The USA pulse oximeters equipment market demonstrates substantial scale, with a recorded valuation of approximately USD ~ billion based on a recent historical assessment, supported by data from sources such as the U.S. Food and Drug Administration (FDA) device registration trends and Centers for Medicare & Medicaid Services procurement records. Growth is driven by rising chronic respiratory conditions, expanding surgical volumes, and widespread integration of monitoring systems in acute and home care settings, alongside heightened patient awareness and continuous monitoring requirements.
Major demand concentration is observed across metropolitan healthcare hubs including New York City, Los Angeles, Chicago, and Houston, where dense hospital networks and advanced clinical infrastructure drive equipment utilization. These regions benefit from high patient throughput, established reimbursement frameworks, and strong adoption of digital health technologies. Additionally, states like California and Texas lead due to large population bases, increasing home healthcare penetration, and strong presence of medical device distributors and manufacturing ecosystems.
Market Segmentation
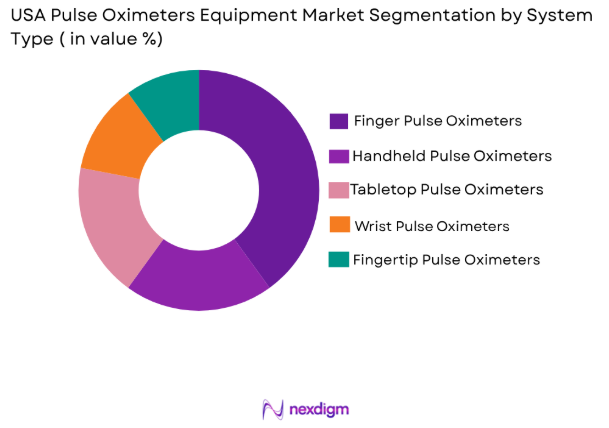
By System Type
USA pulse oximeters equipment market is segmented by system type into fingertip pulse oximeters, handheld pulse oximeters, tabletop/bedside pulse oximeters, wrist-worn pulse oximeters, and pediatric/neonatal pulse oximeters. Recently, fingertip pulse oximeters have a dominant market share due to their affordability, portability, and widespread use in homecare and outpatient settings. These devices are highly preferred for quick spot-check monitoring and are easily accessible through retail and online channels. Their ease of use, minimal training requirement, and growing consumer awareness for self-monitoring further contribute to their strong adoption across diverse user groups.
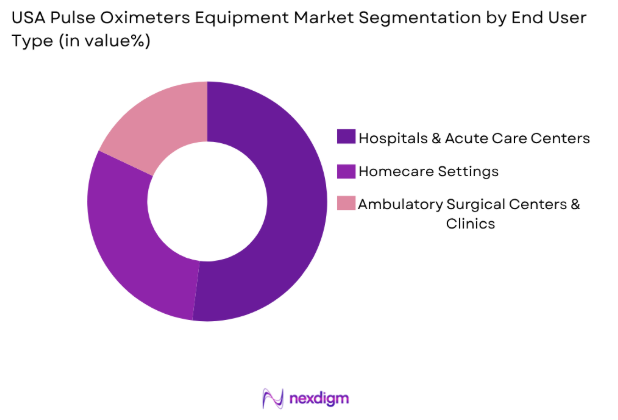
By End User Type
USA pulse oximeters equipment market is segmented by end user into hospitals & acute care centers, homecare settings, and ambulatory surgical centers & clinics. Recently, hospitals & acute care centers hold a dominant market share due to continuous patient monitoring requirements across ICUs, operating rooms, and emergency departments. The high volume of inpatient admissions and surgical procedures necessitates reliable and advanced monitoring equipment. Additionally, regulatory standards mandate oxygen saturation monitoring in critical care, reinforcing consistent procurement, while integration with multi-parameter monitoring systems enhances operational efficiency.
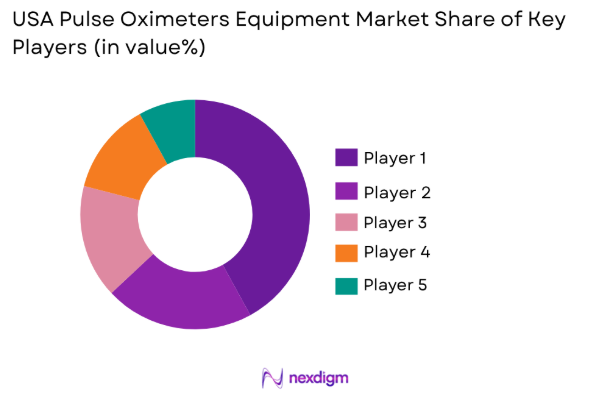
Competitive Landscape
The competitive landscape of the USA pulse oximeters equipment market is highly dynamic, with numerous players vying for market share through technological innovation, strategic partnerships, and acquisitions. Major players in the market are focusing on offering advanced products with improved accuracy, connectivity features, and user-friendly designs. The market is also witnessing consolidation, with companies expanding their product portfolios to meet the increasing demand for remote patient monitoring solutions. Large healthcare providers and medical equipment manufacturers dominate the landscape, influencing market trends through their strong distribution networks and brand presence.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD) | Additional Parameter |
| Medtronic | 1949 | Dublin, Ireland | ~ | ~ | ~ | ~ | ~ |
| Philips Healthcare | 1891 | Amsterdam, Netherlands | ~ | ~ | ~ | ~ | ~ |
| GE Healthcare | 1892 | Chicago, USA | ~ | ~ | ~ | ~ | ~ |
| Masimo Corporation | 1989 | Irvine, USA | ~ | ~ | ~ | ~ | ~ |
| Nonin Medical | 1986 | Plymouth, USA | ~ | ~ | ~ | ~ | ~ |
USA Pulse Oximeters Equipment Market Analysis
Growth Drivers
Technological Advancements in Pulse Oximeters
Technological advancements in pulse oximeters have greatly accelerated the growth of the market. The development of non-invasive devices that deliver real-time and accurate oxygen saturation readings has made them indispensable in healthcare settings, particularly in critical care and home healthcare. Furthermore, the integration of wireless and Bluetooth technologies has enabled continuous, remote monitoring of patients’ oxygen levels, significantly improving patient care by allowing healthcare professionals to intervene swiftly when necessary. The ongoing innovation in wearable devices, which track vital signs such as oxygen levels along with other parameters like heart rate and temperature, is further enhancing patient care. As more people rely on these devices to manage chronic respiratory conditions like asthma, COPD, and even post-COVID care, the demand for pulse oximeters continues to rise. This technological evolution not only improves the accuracy and accessibility of monitoring but also increases its convenience, providing a seamless experience for both healthcare providers and patients.
Increasing Healthcare Awareness
Increasing healthcare awareness, particularly regarding the management and prevention of chronic respiratory diseases, is another major factor contributing to market growth. Public health campaigns and increased focus on health education, especially after the rise of COVID-19, have led to greater awareness of the importance of monitoring oxygen saturation. As people become more conscious of their oxygen levels, they seek affordable and non-invasive solutions to manage their health, further driving the demand for pulse oximeters. These devices are particularly essential for individuals with chronic lung conditions, where monitoring oxygen levels can prevent complications and early diagnosis. Moreover, growing consumer interest in preventive healthcare, coupled with increased healthcare awareness among older adults, is pushing the adoption of pulse oximeters. The widespread availability of information on the benefits of early detection and monitoring has accelerated their adoption, particularly in households with individuals at high risk of respiratory issues.
Market Challenges
High Initial Cost of Pulse Oximeters
A primary challenge for the pulse oximeter market in the USA is the high initial cost of advanced devices, particularly for high-end models with added features. While these devices are critical in improving patient outcomes and enabling early detection of respiratory issues, their cost can be prohibitive, especially for individuals who rely on home healthcare solutions. Many pulse oximeters, particularly those with advanced wireless capabilities or integration with other medical devices, come with a substantial price tag that makes them inaccessible to lower-income populations or those without sufficient insurance coverage. The high upfront cost deters widespread adoption, limiting market penetration in certain demographics. Moreover, the cost barrier impacts both individual consumers and smaller healthcare providers, preventing the full potential of pulse oximeters from being realized. This challenge is compounded by the growing demand for affordable healthcare solutions, creating a gap in the market that remains largely unmet for those unable to afford premium models.
Regulatory and Certification Barriers
Another significant challenge in the USA pulse oximeters market is navigating the regulatory and certification barriers that manufacturers must overcome before introducing new products to the market. Securing approval from regulatory bodies such as the FDA is essential for ensuring product safety and effectiveness, but the process can be lengthy and costly. Manufacturers must meet rigorous standards for accuracy, durability, and safety, which adds complexity and can delay the launch of new models. Smaller companies, in particular, may struggle with the resources required to comply with these stringent regulations. Additionally, the evolving regulatory landscape means that companies must continually adapt to new requirements and guidelines, making it difficult to keep up with the pace of innovation. This complex certification process not only increases the time to market but also raises the overall cost of production, further limiting the accessibility of pulse oximeters, especially in emerging markets.
Opportunities
Expansion in Emerging Markets
The expansion of the USA pulse oximeters market into emerging markets presents a significant growth opportunity. As healthcare infrastructure improves in regions such as Latin America, Asia, and Africa, the demand for reliable and affordable healthcare monitoring devices is increasing. Pulse oximeters, with their simple design and non-invasive functionality, are well-suited for these markets, where there is a growing need for cost-effective healthcare solutions. Manufacturers have the chance to tap into these regions by offering budget-friendly models that meet the demands of local consumers and healthcare providers. Moreover, the expansion of healthcare insurance coverage in these regions, coupled with increasing government investments in healthcare, presents a promising opportunity for market growth. By focusing on product affordability and accessibility, pulse oximeter manufacturers can increase their market share and enhance healthcare delivery in emerging markets.
Integration with Other Wearable Healthcare Devices
Another promising opportunity in the pulse oximeter market is the integration of pulse oximeters with other wearable healthcare devices such as smartwatches and fitness trackers. As consumers increasingly seek holistic health monitoring solutions, combining pulse oximeters with other vital-sign tracking devices presents a unique value proposition. By offering multifunctional wearables that not only measure oxygen levels but also track heart rate, temperature, and other health metrics, companies can cater to the growing demand for comprehensive health data. This integration enhances consumer engagement with healthcare, enabling real-time monitoring of multiple parameters and fostering a more personalized approach to health management. Furthermore, the trend towards connected health ecosystems, with data shared across devices and platforms, allows manufacturers to tap into a broader market of health-conscious consumers. This trend could drive long-term growth, as integrated devices offer convenience and improved health insights, creating a favorable environment for further market expansion.
Future Outlook
Over the next five years, the USA pulse oximeters equipment market is expected to experience steady growth driven by the increasing demand for home healthcare solutions and technological advancements. The integration of pulse oximeters with telemedicine platforms will likely play a significant role in expanding their usage beyond traditional healthcare settings. Regulatory support for remote patient monitoring is anticipated to provide further momentum to the market. The market will also see enhanced demand from emerging applications, such as wellness tracking and fitness monitoring, as consumers increasingly turn to wearable health technologies.
Major Players
- Medtronic
- Philips Healthcare
- GE Healthcare
- Nonin Medical
- Masimo Corporation
- Smiths Medical
- Covidien (Medtronic)
- Sonic Healthcare
- Omron Healthcare
- Innovo Medical
- Zebra Medical Technologies
- Contec Medical Systems
- Roscoe Medical
- Criticare Systems
- Nellcor Puritan Bennett (Medtronic)
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Healthcare providers
- Hospitals and healthcare institutions
- Home healthcare service providers
- Medical equipment distributors
- Emergency medical services
Research Methodology
Step 1: Identification of Key Variables
Key market variables including product categories, end users, pricing trends, and regulatory factors are identified. These variables are mapped based on industry relevance and impact on market performance. Additional factors such as technological advancements, supply chain dynamics, and competitive positioning are also incorporated to ensure a comprehensive variable framework. This step ensures that both macroeconomic and microeconomic influences are captured effectively.
Step 2: Market Analysis and Construction
Comprehensive data is collected from secondary sources and validated through primary research. Market size is constructed using bottom-up and top-down approaches. Multiple data triangulation techniques are applied to enhance reliability and minimize discrepancies. Historical data trends and demand patterns are also analyzed to strengthen the foundation of market modeling and forecasting accuracy.
Step 3: Hypothesis Validation and Expert Consultation
Initial findings are validated through expert interviews with industry professionals. Assumptions are refined based on real-world insights and market dynamics. Feedback from key stakeholders such as manufacturers, distributors, and healthcare providers is incorporated to ensure practical relevance. This process helps in identifying potential gaps and aligning the research with current industry developments.
Step 4: Research Synthesis and Final Output
All data points are synthesized into a structured report. Final outputs are reviewed for accuracy, consistency, and alignment with industry standards. Advanced analytical tools are used to generate insights, trends, and forecasts. Quality checks and peer reviews are conducted to ensure the report meets rigorous research standards and delivers actionable intelligence.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Rising prevalence of respiratory and cardiovascular disorders increasing routine oxygen saturation monitoring demand
Expanding home healthcare and remote patient monitoring adoption across chronic care and post-acute settings
Higher patient safety focus in hospitals, ambulatory centers, and emergency response environments driving pulse oximeter procurement - Market Challenges
Accuracy limitations in low perfusion conditions, motion environments, and diverse skin pigmentation groups
Price pressure from commoditized low-cost imports affecting premium device positioning and margin realization
Regulatory compliance and product recall risks creating barriers for smaller manufacturers and distributors - Market Opportunities
Growing demand for connected pulse oximeters integrated with telehealth and electronic health record platforms
Increasing use of pediatric and neonatal monitoring devices in specialized care units and maternity hospitals
Procurement opportunities from outpatient care expansion, long-term care facilities, and at-home chronic disease management programs - Trends
Shift toward wireless, app-based, and cloud-connected pulse oximetry solutions for continuous monitoring
Product innovation focused on improved sensor accuracy, compact design, and multi-parameter monitoring capability - Government Regulations
- SWOT Analysis
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Fingertip Pulse Oximeters
Handheld Pulse Oximeters
Tabletop / Bedside Pulse Oximeters
Wrist-Worn Pulse Oximeters
Pediatric / Neonatal Pulse Oximeters - By Platform Type (In Value%)
Standalone Monitoring Systems
Wireless / Bluetooth-Enabled Systems
USB / App-Connected Systems
Integrated Multi-Parameter Monitoring Systems
Cloud-Linked Remote Monitoring Systems - By Fitment Type (In Value%)
Portable Systems
Fixed Bedside Systems
Wearable Systems
Clip-On Systems - By End User Segment (In Value%)
Hospitals & Acute Care Centers
Homecare Settings
Ambulatory Surgical Centers & Clinics
- Market Share Analysis
- Cross Comparison Parameters (Product Portfolio Breadth, Sensor Accuracy Performance, Connectivity Features, End User Coverage, Pricing Positioning, Distribution Network Strength, Regulatory Compliance & Certifications, Product Innovation Rate, After-Sales Service Support, Brand Reputation, Manufacturing Capabilities, Supply Chain Efficiency, R&D Investment Intensity, Device Durability & Reliability, Integration with Digital Health Platforms)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Masimo Corporation
Medtronic plc
Nonin Medical, Inc.
Koninklijke Philips N.V.
Nihon Kohden Corporation
GE HealthCar
Smiths Medical
Becton, Dickinson and Company
Baxter International Inc.
Contec Medical Systems Co., Ltd.
Edan Instruments, Inc.
Shenzhen Mindray Bio-Medical Electronics Co., Ltd.
Zacurate
ChoiceMMed
Vyaire Medical, Inc.
- Hospitals prioritizing multi-parameter integrated monitoring systems
- Growth in home healthcare adoption among chronic disease patients
- Ambulatory centers focusing on cost-effective portable devices
- Increased use in emergency and critical care settings
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- By Average System Price, 2026-2035
- By System Complexity Tier, 2026-2035