Market Overview
The USA Respiratory Rate Monitors Equipment Market is valued at USD ~ billion based on a recent historical assessment. This growth is driven by increasing respiratory health concerns, aging populations, and technological advancements in monitoring systems. The market is characterized by a surge in demand for non-invasive monitoring devices, which have been shown to improve patient outcomes while minimizing discomfort. Additionally, hospitals and healthcare facilities are expanding their adoption of advanced equipment, boosting market growth, and contributing to a steady increase in market value.
The dominant regions in the USA Respiratory Rate Monitors Equipment Market are California, Texas, and Florida, with hospitals and healthcare institutions in these areas adopting respiratory monitoring systems at a faster pace. Factors driving this dominance include larger healthcare infrastructure, higher population densities, and increased prevalence of respiratory conditions, especially in the elderly population. Furthermore, these regions have been at the forefront of technological innovation and integration in healthcare, creating an environment conducive to market growth.
Market Segmentation
By Product Type
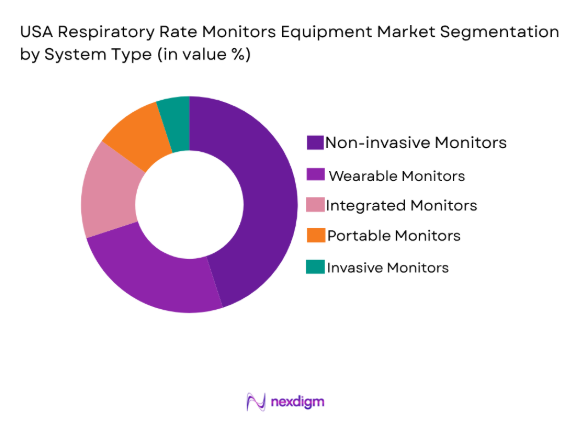
USA Respiratory Rate Monitors Equipment market is segmented by product type into non-invasive monitors, wearable monitors, integrated monitors, portable monitors, and invasive monitors. Recently, non-invasive monitors have a dominant market share due to factors such as demand patterns favoring patient comfort, minimal procedure-related risks, and an increasing focus on outpatient and home care settings. These systems have become increasingly popular because they reduce the need for invasive procedures, offering significant advantages in terms of patient safety and ease of use.
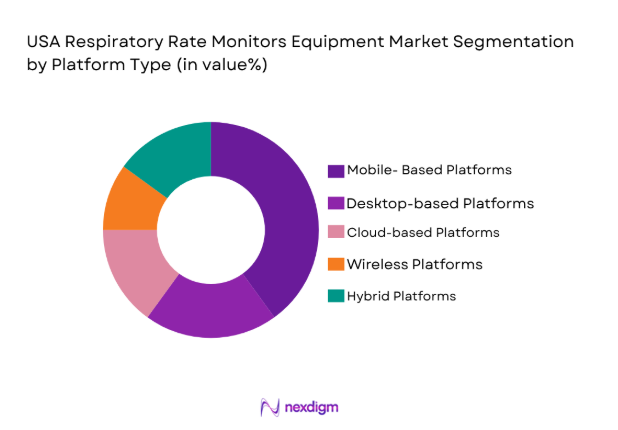
By Platform Type
USA Respiratory Rate Monitors Equipment market is segmented by platform type into mobile-based platforms, desktop-based platforms, cloud-based platforms, wireless platforms, and hybrid platforms. Recently, mobile-based platforms have a dominant market share due to the growing reliance on smartphones and tablets in the healthcare industry. These devices are versatile, cost-effective, and allow healthcare providers to monitor patients remotely, making them an ideal choice for both hospitals and home care environments. Their integration with wearable devices further fuels their adoption.
Competitive Landscape
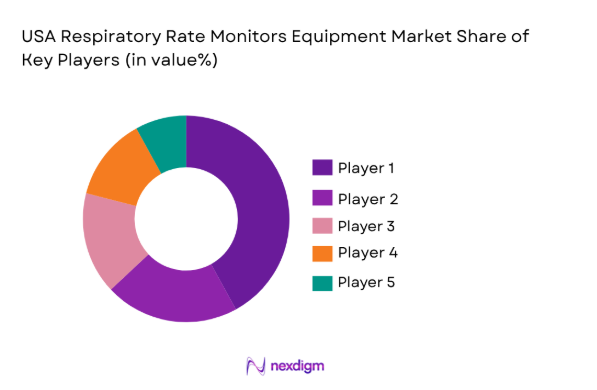
The competitive landscape of the USA Respiratory Rate Monitors Equipment Market is highly fragmented, with numerous players operating in both the technological and healthcare service sectors. Major players in the market are focusing on mergers, acquisitions, and strategic partnerships to consolidate their position. In addition, companies are investing heavily in R&D to advance the technology behind respiratory rate monitors, allowing them to offer more accurate and user-friendly devices. These efforts are influencing the market dynamics and shaping the competitive environment.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue (USD) | Additional Parameter |
| Medtronic | 1949 | Dublin, Ireland | |||||
| Philips Healthcare | 1891 | Amsterdam, Netherlands | |||||
| GE Healthcare | 1892 | Chicago, USA | |||||
| Dräger | 1889 | Lübeck, Germany | |||||
| Masimo Corporation | 1989 | Irvine, USA |
USA Respiratory Rate Monitors Equiptment Market Analysis
Growth Drivers
Technological Advancements in Non-Invasive Devices
Technological advancements in non-invasive monitoring devices are significantly driving market growth. These devices offer improved patient comfort, accurate monitoring, and faster results, making them ideal for both in-hospital and home care settings. Furthermore, innovations in sensor technologies, wireless communication, and artificial intelligence are enhancing the functionality of respiratory rate monitors. These advancements have made non-invasive monitors more accessible, reliable, and cost-effective, driving their increased adoption across healthcare facilities. The demand for these devices is expected to rise as healthcare providers focus on minimizing invasive procedures while ensuring high-quality patient care. With these improvements, non-invasive monitors are expected to play a crucial role in monitoring respiratory conditions, particularly in the elderly population and patients with chronic respiratory diseases.
Increased Focus on Remote Patient Monitoring
The growing emphasis on remote patient monitoring is another key driver for the USA Respiratory Rate Monitors Equipment Market. The shift toward telemedicine and remote healthcare services, accelerated by the COVID-19 pandemic, has led to a greater reliance on digital health technologies, including respiratory rate monitors. Remote monitoring enables healthcare providers to track patient progress without requiring in-person visits, reducing strain on healthcare facilities and enhancing patient outcomes. The ability to monitor patients from the comfort of their homes also improves patient compliance and reduces hospitalization rates. As the demand for remote patient monitoring continues to increase, it is expected that more healthcare providers will invest in respiratory monitoring devices to improve their service offerings.
Market Challenges
High Initial Investment Costs
The high initial investment costs associated with respiratory rate monitors pose a challenge to widespread adoption, particularly among smaller healthcare facilities and homecare providers. Although the long-term benefits of these devices, such as improved patient outcomes and reduced hospital admissions, are substantial, the upfront costs can be prohibitive. Many healthcare institutions, particularly in rural or underfunded areas, face budget constraints that hinder their ability to adopt advanced respiratory monitoring systems. These cost-related challenges slow the market’s overall growth, despite the clear advantages these devices provide.
Regulatory Barriers and Compliance Issues
Regulatory barriers and compliance issues remain a significant challenge in the USA Respiratory Rate Monitors Equipment Market. Manufacturers must adhere to stringent FDA regulations and certification processes, which can delay the market entry of new devices. Additionally, evolving regulations surrounding data privacy and security in digital health platforms add complexity to product development. For companies, the need to navigate these regulatory landscapes can increase costs, extend product development timelines, and create barriers to market entry. Consequently, regulatory compliance issues represent a major obstacle for companies looking to innovate and expand in this space.
Opportunities
Expanding Demand for Homecare Monitoring Solutions
The increasing demand for homecare monitoring solutions presents a significant opportunity in the respiratory rate monitors market. With an aging population and a rise in chronic respiratory conditions such as COPD and asthma, more patients are requiring regular monitoring of their health outside of clinical settings. Homecare respiratory rate monitors offer an effective solution for these patients, allowing them to manage their conditions independently. Healthcare providers are increasingly recommending these devices to improve patient quality of life and reduce hospital visits. As more people opt for homecare monitoring solutions, companies in the respiratory rate monitoring market will find increased demand for their products.
Technological Advancements in Artificial Intelligence for Monitoring Devices
The integration of artificial intelligence (AI) into respiratory rate monitoring devices is poised to unlock new opportunities in the market. AI-powered devices can provide real-time analytics, allowing healthcare professionals to monitor patient data more efficiently and make quicker decisions regarding patient care. AI can also enable predictive analytics, identifying early warning signs of respiratory distress and helping prevent emergency situations. As the healthcare industry continues to embrace AI for improving diagnostics and patient management, the demand for AI-integrated respiratory rate monitors is expected to grow, presenting a lucrative opportunity for market players.
Future Outlook
The future outlook for the USA Respiratory Rate Monitors Equipment Market is positive, with steady growth expected over the next five years. Technological advancements, particularly in non-invasive and AI-integrated devices, will drive innovation and increased adoption. Furthermore, supportive government regulations and the expansion of healthcare services in remote areas will facilitate market growth. As healthcare providers continue to prioritize patient comfort and efficient monitoring, the demand for advanced respiratory rate monitors is anticipated to rise, particularly in homecare settings and ambulatory services.
Major Players
- Medtronic
- Philips Healthcare
- GE Healthcare
- Dräger
- Masimo Corporation
- Nonin Medical
- Nihon Kohden
- Smiths Medical
- Mindray Medical
- Fisher & Paykel Healthcare
- Shenzhen Mindray Bio-Medical Electronics
- ResMed
- Invivo Healthcare
- OxyGo
- Welch Allyn
Key Target Audience
- Investments and venture capitalist firms
- Government and regulatory bodies
- Healthcare providers (Hospitals, Clinics, and Ambulatory Centers)
- Homecare providers
- Medical device distributors
- Insurance companies and healthcare payers
- Research institutions and health tech developers
Research Methodology
Step 1: Identification of Key Variables
Identification of key variables involves defining critical parameters that influence the market, such as product types, platform types, end-users, and technological advancements. Additional factors such as regulatory environment, pricing structures, reimbursement policies, and supply chain dynamics are also incorporated. This step ensures a comprehensive foundation for further analysis by aligning all relevant market determinants.
Step 2: Market Analysis and Construction
This step involves analyzing market data and constructing a detailed framework to understand the market dynamics and identify trends in product usage, pricing, and consumer behavior. Historical data patterns, competitive positioning, and demand-supply gaps are evaluated to build a structured market model. This phase also integrates macroeconomic indicators and healthcare infrastructure developments to enhance analytical accuracy.
Step 3: Hypothesis Validation and Expert Consultation
Validation of hypotheses through consultations with industry experts, healthcare professionals, and market analysts ensures the accuracy of the findings and recommendations. Primary interviews and expert opinions are used to refine assumptions and validate data points. This process minimizes bias and strengthens the reliability of research conclusions through real-world insights.
Step 4: Research Synthesis and Final Output
Synthesizing collected data into comprehensive reports and producing final outputs that provide actionable insights into the market’s future trajectory. Data triangulation methods are applied to reconcile variations across sources and ensure consistency. The final deliverable is structured to support strategic decision-making with clear, data-backed insights and forecasts.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Rising Prevalence of Respiratory Diseases
Technological Advancements in Monitoring Systems
Increasing Healthcare Awareness and Access - Market Challenges
High Initial Cost of Equipment
Complexity in Integration with Existing Systems
Regulatory Barriers and Compliance Issues - Market Opportunities
Expanding Homecare Monitoring Solutions
Technological Innovations for Wearable Devices
Strategic Partnerships with Healthcare Providers - Trends
Growth in Remote Monitoring Services
Increased Use of Artificial Intelligence for Respiratory Monitoring - Government Regulations
- SWOT Analysis
- Porter’s Five Forces
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Non-invasive Monitors
Wearable Monitors
Integrated Monitors
Portable Monitors
Invasive Monitors - By Platform Type (In Value%)
Mobile-based Platforms
Desktop-based Platforms
Cloud-based Platforms
Wireless Platforms
Hybrid Platforms - By Fitment Type (In Value%)
Bedside Monitors
Wearable Devices
Handheld Monitors
Wall-mounted Monitors - By End User Segment (In Value%)
Hospitals
Homecare Providers
Ambulatory Surgical Centers
- Market Share Analysis
- Cross Comparison Parameters (Product Quality, Market Reach, Technology Adoption, Cost Efficiency, Regulatory Compliance, Customer Satisfaction, Distribution Channels, Pricing Competitiveness, Product Portfolio Breadth, After-Sales Service Support, Brand Recognition, R&D Investment Strength, Device Accuracy and Reliability, Integration with Hospital Systems, Wireless Connectivity Capabilities, Portability and Ease of Use, Manufacturing Capacity, Supply Chain Efficiency, Clinical Validation Support, Service Network Availability)
- SWOT Analysis of Key Competitors
- Pricing & Procurement Analysis
- Key Players
Medtronic
Philips Healthcare
GE Healthcare
Dräger
Masimo Corporation
Nonin Medical
Nihon Kohden
Smiths Medical
Mindray Medical
Fisher & Paykel Healthcare
Shenzhen Mindray Bio-Medical Electronics
ResMed
Invivo Healthcare
OxyGo
Welch Allyn
- Hospitals and Healthcare Facilities
- Homecare and Patient Monitoring Solutions
- Ambulatory Surgical Centers and Clinics
- Insurance Providers and Reimbursement Policies
- Forecast Market Value, 2026-2035
- Forecast Installed Units, 2026-2035
- Price Forecast by System Tier, 2026-2035
- Future Demand by Platform, 2026-2035