Market Overview
The USA Urine Analyzers Equipment market demonstrated strong diagnostic equipment demand with a total market size of USD ~ billion based on a recent historical assessment derived from aggregated financial disclosures of major diagnostic equipment manufacturers and regulatory device registration databases. Growth is largely driven by increasing chronic disease screening volumes, rising clinical laboratory automation, and demand for faster urinalysis workflows. Expansion of preventive diagnostics programs and increasing hospital laboratory testing volumes continue to sustain equipment replacement cycles and technology upgrades.
Within the country, major healthcare technology adoption is concentrated in cities such as New York, Boston, San Diego, and Chicago due to dense hospital networks and strong biotechnology ecosystems. California and Massachusetts dominate because of high healthcare expenditure, strong research funding exceeding USD ~ billion, and the presence of diagnostic innovation clusters. Texas also remains a key demand center due to large hospital infrastructure investments exceeding USD ~ billion in recent healthcare modernization programs and expanding private diagnostic chains.
Market Segmentation
By System Type
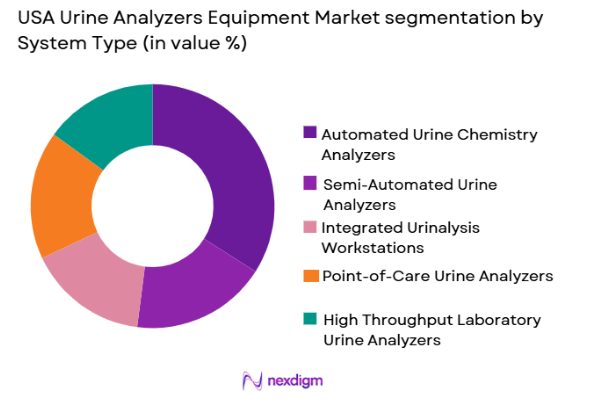
USA Urine Analyzers Equipment market is segmented by system type into Automated Urine Chemistry Analyzers, Semi-Automated Urine Analyzers, Integrated Urinalysis Workstations, Point-of-Care Urine Analyzers, and High Throughput Laboratory Urine Analyzers. Recently, Automated Urine Chemistry Analyzers have a dominant market share due to factors such as increasing laboratory automation needs, higher testing volumes, strong brand presence from major diagnostic companies, and the need for standardized results across large hospital networks. These systems reduce manual errors, improve workflow efficiency, and allow laboratories to process large volumes of tests quickly. Their compatibility with laboratory information systems and electronic health records also makes them preferable for large healthcare providers. Additionally, hospitals continue to upgrade legacy systems to automated platforms to improve diagnostic turnaround time. Growing disease screening programs and preventive testing protocols further strengthen demand for automated analyzers, particularly in high-volume diagnostic environments.
By Platform Type
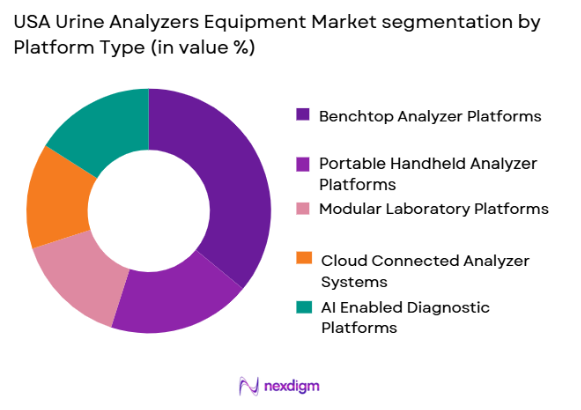
USA Urine Analyzers Equipment market is segmented by platfrom type into Benchtop Analyzer Platforms, Portable Handheld Analyzer Platforms, Modular Laboratory Platforms, Cloud Connected Analyzer Systems, and AI Enabled Diagnostic Platforms. Recently, Benchtop Analyzer Platforms have a dominant market share due to factors such as widespread installation in hospitals and diagnostic laboratories, proven reliability, established infrastructure compatibility, and strong procurement preference among centralized testing facilities. These platforms are considered industry standards for routine urinalysis because they offer consistent performance, multi-parameter testing capability, and integration flexibility. Diagnostic laboratories also prefer benchtop systems due to their scalability and ability to process diverse testing panels. Furthermore, service support availability and technician familiarity with these systems contribute to continued demand. Procurement contracts from hospital groups and laboratory chains further strengthen their leadership position within the market landscape.
Competitive Landscape
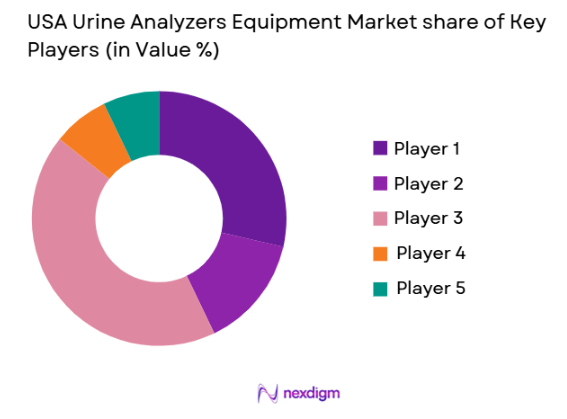
The USA Urine Analyzers Equipment market shows moderate consolidation with global diagnostic leaders controlling innovation pipelines while mid-tier players compete through pricing and niche technological specialization. Major companies leverage automation platforms, AI-based microscopy, and integrated laboratory software to strengthen their competitive positioning. Strategic partnerships with hospital systems and reagent supply agreements further reinforce long-term customer retention. Market competition is also shaped by product reliability, service contracts, regulatory approvals, and integration capabilities with electronic health records.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Additional Parameter – Automation Capability |
| Abbott Laboratories | 1888 | Illinois, USA | ~ | ~ | ~ | ~ | ~ |
| Siemens Healthineers | 1847 | Erlangen, Germany | ~ | ~ | ~ | ~ | ~ |
| Roche Diagnostics | 1896 | Basel, Switzerland | ~ | ~ | ~ | ~ | ~ |
| Beckman Coulter | 1935 | California, USA | ~ | ~ | ~ | ~ | ~ |
| Sysmex Corporation | 1968 | Kobe, Japan | ~ | ~ | ~ | ~ | ~ |
USA Urine Analyzers Equipment Market Analysis
Growth Drivers
Rising prevalence of diabetes and kidney related disorders
The increasing burden of diabetes and chronic kidney disease is a primary driver for urine analyzers, as urinalysis is a standard diagnostic tool for early detection and monitoring. According to the International Diabetes Federation (IDF), 537 million adults were living with diabetes globally in 2022, projected to reach 643 million by 2030, creating sustained diagnostic demand. In India alone, over 101 million individuals were diabetic in 2023 as reported by ICMR, while the World Health Organization (WHO) highlights kidney disease among the top 10 causes of death globally. Additionally, the World Bank reported global healthcare expenditure reaching USD 9.8 trillion in 2023, reflecting increased diagnostic spending. Rising patient volume directly increases demand for urine sediment analysis in hospitals and labs.
Increasing demand for automated laboratory diagnostics
Laboratories are increasingly adopting automation to handle rising test volumes and reduce turnaround times. The World Bank reported global urban population reaching 4.4 billion in 2023, increasing pressure on urban diagnostic labs. Meanwhile, OECD health data (2024) shows diagnostic testing volumes rising across member countries, with clinical lab tests per capita exceeding 20 annually in developed regions. Automation helps manage this scale efficiently. Furthermore, the International Labour Organization (ILO) reported healthcare workforce shortages exceeding 10 million globally in 2023, pushing labs toward automation to maintain productivity. Automated urine analyzers improve throughput, reduce manual errors, and standardize results, making them critical as healthcare systems scale diagnostics infrastructure worldwide.
Market Challenges
High initial investment cost of automated urine analyzers
The adoption of automated urine analyzers is limited by high upfront costs, particularly in developing economies with constrained healthcare budgets. According to the World Bank, low- and middle-income countries accounted for over 75 countries with per capita healthcare spending below USD 500 in 2023, restricting capital investment in advanced diagnostic systems. In India, National Health Accounts (2022-23) reported per capita health expenditure at approximately INR 4,863, indicating limited affordability for high-end automation in smaller labs. Additionally, public healthcare systems prioritize essential infrastructure over advanced diagnostics. This financial barrier slows adoption, especially among standalone diagnostic centers and rural hospitals, where manual microscopy remains prevalent due to lower setup costs.
Maintenance and calibration requirements for advanced systems
Automated urine analyzers require regular calibration, software updates, and skilled handling, which increases operational complexity. The WHO Global Health Workforce Statistics (2023) highlights a shortage of trained laboratory professionals in many regions, with density below 10 lab technicians per 10,000 population in several countries. This lack of skilled personnel impacts maintenance efficiency. Additionally, the International Energy Agency (IEA) reported electricity reliability challenges in multiple emerging economies, affecting uninterrupted operation of automated systems. Maintenance downtime can delay diagnostics, reducing system utilization. These factors increase total cost of ownership and create hesitation among smaller laboratories that lack technical expertise and infrastructure to maintain advanced automated urine analysis systems effectively.
Market Opportunities
Adoption of AI based urine analysis solutions
Artificial intelligence integration in urine analysis presents strong growth opportunities by improving diagnostic accuracy and efficiency. The World Bank (2024) reports global digital adoption expanding rapidly, with over 5.4 billion internet users, enabling AI-enabled healthcare deployment. In healthcare, the WHO Global Strategy on Digital Health highlights increasing use of AI in diagnostics to address workforce shortages. AI-based urine analyzers can process thousands of images per sample, reducing reliance on manual microscopy. Additionally, countries like India under the National Digital Health Mission are expanding digital health infrastructure, supporting AI integration in labs. These trends indicate strong potential for AI-powered urine diagnostics to enhance scalability and standardization in laboratory testing.
Growth of home based diagnostic testing devices
Home-based diagnostics are gaining traction due to increasing healthcare awareness and convenience. According to the World Bank (2023), global life expectancy reached 73 years, increasing the need for regular health monitoring among aging populations. Additionally, the WHO (2024) emphasizes the rise of self-testing and decentralized diagnostics to reduce hospital burden. In India, the Ministry of Health and Family Welfare reports increasing adoption of home healthcare services, particularly in urban areas. Urine test kits and portable analyzers enable early detection of infections and chronic diseases without hospital visits. This shift toward home-based care is creating demand for compact, user-friendly urine diagnostic devices integrated with digital reporting systems.
Future Outlook
The USA Urine Analyzers Equipment market is expected to witness steady expansion driven by increasing laboratory automation, digital diagnostics adoption, and rising chronic disease screening demand. Technological innovation including AI diagnostics and connected laboratory ecosystems will support modernization. Regulatory stability is expected to encourage product approvals and commercialization. Demand will also be supported by preventive healthcare programs and expansion of decentralized diagnostic services. Equipment replacement demand and digital healthcare transformation are expected to remain key growth contributors.
Major Players
- Abbott Laboratories
- SiemensHealthineers
- Roche Diagnostics
- Beckman Coulter
- Sysmex Corporation
- Thermo Fisher Scientific
- Bio-Rad Laboratories
- Arkray Inc.
- ACON Laboratories
- Nova Biomedical
- Mindray Medical
- QuidelOrtho Corporation
- 77 Elektronika
- Cardinal Health
- Danaher Corporation
Key Target Audience
- Hospitals and healthcare systems
- Diagnostic laboratory chains
- Medical device distributors
- Healthcare equipment procurement groups
- Investments and venture capitalist firms
- Government and regulatory bodies
- Private healthcare providers
- Medical technology manufacturers
Research Methodology
Step 1: Identification of Key Variables
Key variables such as equipment pricing, installed diagnostic base, hospital procurement rates, laboratory automation adoption, and chronic disease testing volumes were identified. Regulatory approvals and manufacturer revenue disclosures were also considered.
Step 2: Market Analysis and Construction
Market size was constructed through analysis of company financials, device registrations, procurement data, and healthcare infrastructure investments. Demand modeling was performed based on diagnostic testing volume trends and equipment replacement cycles.
Step 3: Hypothesis Validation and Expert Consultation
Industry assumptions were validated through expert interviews with laboratory managers, biomedical engineers, and procurement specialists. Market estimates were cross verified using industry benchmarks and diagnostic equipment shipment trends.
Step 4: Research Synthesis and Final Output
Data triangulation methods were applied to finalize market estimates. Qualitative and quantitative findings were synthesized into a structured research framework ensuring consistency, reliability, and realistic industry representation.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Rising prevalence of diabetes and kidney related disorders
Increasing demand for automated laboratory diagnostics
Growth in preventive health screening programs
Technological advancements in digital urine sediment analysis
Expansion of diagnostic laboratory networks across the USA - Market Challenges
High initial investment cost of automated urine analyzers
Maintenance and calibration requirements for advanced systems
Shortage of skilled laboratory technicians
Data integration challenges with legacy hospital systems
Pricing pressure due to competitive bidding processes - Market Opportunities
Adoption of AI based urine analysis solutions
Growth of home based diagnostic testing devices
Expansion opportunities in telehealth integrated diagnostics - Trends
Integration of artificial intelligence in urine diagnostics
Shift toward compact point of care testing devices
Increasing adoption of cloud based diagnostic data management
Growth of multiplex urinalysis testing platforms
Development of rapid screening technologies for infection detection - Government Regulations & Defense Policy
FDA regulatory compliance requirements for diagnostic devices
CLIA certification standards for laboratory testing equipment
Data privacy compliance under healthcare information regulations - SWOT Analysis
- Stakeholder and Ecosystem Analysis
- Porter’s Five Forces Analysis
- Competition Intensity and Ecosystem Mapping
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Automated Urine Chemistry Analyzers
Semi-Automated Urine Analyzers
Integrated Urinalysis Workstations
Point-of-Care Urine Analyzers
High Throughput Laboratory Urine Analyzers - By Platform Type (In Value%)
Benchtop Analyzer Platforms
Portable Handheld Analyzer Platforms
Modular Laboratory Platforms
Cloud Connected Analyzer Systems
AI Enabled Diagnostic Platforms - By Fitment Type (In Value%)
Standalone diagnostic systems
Laboratory integrated systems
Hospital information system compatible units
Mobile diagnostic units
Cloud connected diagnostic equipment - By End User Segment (In Value%)
Hospitals and multi specialty clinics
Independent diagnostic laboratories
Physician office laboratories
Academic and research institutes
Home healthcare settings - By Procurement Channel (In Value%)
Direct procurement from manufacturers
Medical equipment distributors
Group purchasing organizations
Online medical equipment platforms
Leasing and rental service providers - By Material / Technology (in Value %)
Flow cytometry based analysis technology
Digital imaging based urine sediment analysis
Reflectance photometry technology
Microfluidic urine testing technology
AI enabled pattern recognition software
- Market structure and competitive positioning
- Market share snapshot of major players
- Cross Comparison Parameters (Product Portfolio Breadth, Automation Capability, Throughput Capacity, Technology Integration Level, Pricing Strategy, Distribution Network Strength, After Sales Service Support, Regulatory Certifications, R&D Investment Intensity, Digital Connectivity Features)
- SWOT Analysis of Key Players
- Pricing & Procurement Analysis
- Key Players
Abbott Laboratories
Becton Dickinson and Company
Siemens Healthineers
Roche Diagnostics Corporation
Beckman Coulter Inc.
Sysmex America Inc.
BioRad Laboratories Inc.
Arkray USA Inc.
77 Elektronika Kft
Mindray North America
QuidelOrtho Corporation
Acon Laboratories Inc.
Clinitek Diagnostics Systems
Nova Biomedical Corporation
Thermo Fisher Scientific Inc.
- Hospitals driving demand through high patient diagnostic volumes
- Diagnostic chains investing in automation for efficiency improvement
- Physician clinics adopting compact point of care analyzers
- Research institutions using advanced analyzers for clinical studies
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025