Market Overview
The USA urine sediment microscopes market is part of the broader urinalysis instruments industry, which recorded a global value of USD ~ billion for automated urine sediment analyzers and USD ~ billion for total urinalysis systems based on a recent historical assessment, according to Grand View Research. The market is driven by increasing diagnostic testing volumes, automation of clinical laboratories, and rising detection of kidney disorders and urinary infections, which continue to increase the use of microscopic urinalysis technologies across healthcare facilities.
Major demand originates from cities such as New York, Boston, San Diego, and Chicago due to strong healthcare infrastructure, large diagnostic laboratory networks, and the presence of leading medical device manufacturers. The United States dominates adoption due to advanced clinical laboratory systems, high healthcare spending exceeding USD ~ trillion, and strong regulatory oversight encouraging high-quality diagnostics, which supports continued installation of automated microscopy solutions in hospital networks and reference laboratories.
Market Segmentation
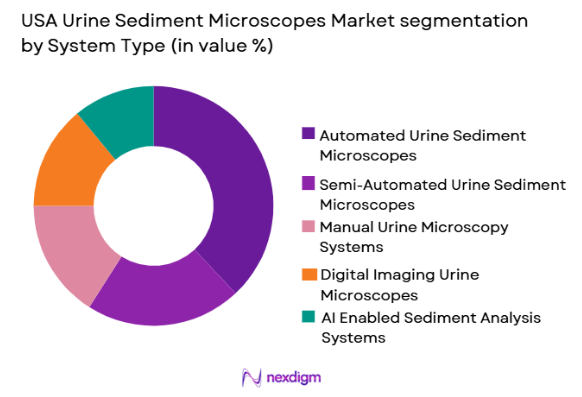
By System Type
USA Urine Sediment Microscopes market is segmented by system type into automated urine sediment microscopes, semi-automated urine sediment microscopes, manual urine microscopy systems, digital imaging urine microscopes, and AI-enabled sediment analysis systems. Recently, automated urine sediment microscopes have a dominant market share due to factors such as demand patterns, brand presence, infrastructure availability, or consumer preference. Clinical laboratories increasingly prefer these systems because they reduce manual workload and improve diagnostic consistency. Automation also allows high sample throughput in centralized labs. Integration with laboratory information systems has also increased their preference. Strong product availability from major diagnostic manufacturers and demand for standardization across multi-location diagnostic chains also contribute to this segment’s leadership.
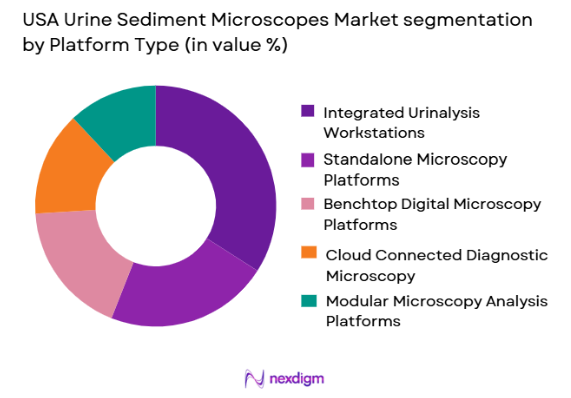
By Platform Type
USA Urine Sediment Microscopes market is segmented by platform type into standalone microscopy platforms, integrated urinalysis workstations, cloud-connected diagnostic microscopy systems, benchtop digital microscopy platforms, and modular microscopy analysis platforms. Recently, integrated urinalysis workstations have a dominant market share due to factors such as demand patterns, brand presence, infrastructure availability, or consumer preference. Hospitals increasingly prefer integrated platforms because they combine biochemical and microscopic analysis in a single workflow. This reduces sample handling errors and improves turnaround time. Growing consolidation among diagnostic providers has increased demand for multi-functional systems. Vendor support services and interoperability with hospital IT infrastructure also contribute to segment growth.
Competitive Landscape
The USA urine sediment microscopes market shows moderate consolidation with multinational diagnostic companies dominating technology development and distribution. Major players benefit from established distribution networks, strong product portfolios, and integration capabilities with laboratory software platforms. Competition is largely based on automation capability, imaging accuracy, workflow integration, and service support. Strategic partnerships, product upgrades, and regulatory approvals play a significant role in strengthening the competitive positioning of leading companies while smaller players focus on niche automation and digital microscopy innovations.
| Company Name | Establishment Year | Headquarters | Technology Focus | Market Reach | Key Products | Revenue | Automation Integration Capability |
| Sysmex Corporation | 1968 | Kobe, Japan | ~ | ~ | ~ | ~ | ~ |
| Siemens Healthineers | 1847 | Erlangen, Germany | ~ | ~ | ~ | ~ | ~ |
| Roche Diagnostics | 1896 | Basel, Switzerland | ~ | ~ | ~ | ~ | ~ |
| Beckman Coulter | 1935 | California, USA | ~ | ~ | ~ | ~ | ~ |
| Abbott Laboratories | 1888 | Illinois, USA | ~ | ~ | ~ | ~ | ~ |
USA Urine Sediment Microscopes Market Analysis
Growth Drivers
Rising prevalence of kidney disorders and urinary tract infections in the United States
The demand for urine sediment microscopes in the United States is strongly driven by the increasing burden of kidney-related diseases and urinary tract infections (UTIs). According to the Centers for Disease Control and Prevention, around 37,000,000 adults in the U.S. were living with chronic kidney disease between 2022 and 2024, while kidney diseases ranked among the top causes of mortality with over 54,000 deaths recorded annually. Additionally, UTIs account for approximately 8,000,000 healthcare visits each year, increasing diagnostic workload in clinical laboratories. The World Bank reported U.S. population at 333,000,000 in 2024, supporting high testing volumes, while healthcare expenditure exceeded 4,500,000,000,000 USD, reinforcing diagnostic infrastructure expansion.
Increasing laboratory automation adoption to reduce diagnostic turnaround time
Laboratory automation is accelerating the adoption of urine sediment microscopy systems across the United States, driven by the need to handle rising diagnostic volumes efficiently. The U.S. Bureau of Labor Statistics reported over 335,000 clinical laboratory technologists and technicians employed in 2024, yet demand continues to outpace workforce availability. Automated microscopy helps laboratories process thousands of urine samples daily, reducing turnaround time significantly. The International Monetary Fund highlighted U.S. GDP at 28,780,000,000,000 USD in 2024, reflecting strong healthcare investment capacity. Additionally, U.S. healthcare spending per capita exceeded 13,000 USD, indicating significant allocation toward advanced diagnostic technologies, including automated urine sediment analyzers in hospitals and diagnostic labs.
Market Challenges
High acquisition and maintenance costs of automated microscopy systems
The adoption of advanced urine sediment microscopes is constrained by high capital and operational costs, particularly for small and mid-sized laboratories. According to the U.S. Census Bureau, there were over 35,000 medical laboratories operating in the U.S. between 2022 and 2024, many of which function with limited budgets. While the World Bank reported U.S. inflation levels fluctuating between 4 and 8 index points during 2022–2023, rising costs of imported optical components and maintenance services have increased financial pressure on healthcare providers. Furthermore, national health expenditure exceeding 4,500,000,000,000 USD reflects overall spending, but cost distribution remains uneven, limiting adoption of high-end automated microscopy in smaller facilities.
Shortage of skilled laboratory technicians trained in advanced microscopy
A shortage of trained professionals capable of operating automated urine sediment microscopes presents a significant market barrier. The U.S. Bureau of Labor Statistics reported that the U.S. healthcare system had around 335,000 laboratory technicians in 2024, while job openings continue to rise annually due to aging workforce and increased diagnostic demand. Training in advanced microscopy and automation remains limited, restricting effective utilization of sophisticated systems. Additionally, the World Bank noted that tertiary education enrollment in the U.S. exceeded 19,000,000 students, yet specialized medical laboratory training remains a small subset. This skill gap directly impacts operational efficiency and slows adoption of advanced urine sediment analysis technologies.
Market Opportunities
Expansion of AI powered automated urine sediment analysis solutions
The integration of artificial intelligence into urine sediment analysis presents a strong growth opportunity in the U.S. market. The National Science Foundation reported federal R&D spending exceeding 190,000,000,000 USD in 2024, with a growing portion allocated to AI and healthcare technologies. AI-powered microscopy enables automated detection of cells, bacteria, and crystals, improving diagnostic accuracy and efficiency. Additionally, the World Bank highlighted that over 92 out of 100 people in the U.S. had internet access in 2024, supporting integration of cloud-based diagnostic platforms. Combined with healthcare digitalization, these factors create a strong foundation for scaling AI-enabled urine sediment microscopy solutions across hospitals and laboratories.
Growing adoption of point of care urinalysis diagnostics
Point-of-care (POC) diagnostics are gaining traction in the United States, creating demand for compact urine sediment microscopy solutions. The Centers for Medicare & Medicaid Services reported that total national health expenditure reached over 4,500,000,000,000 USD in 2024, with increasing focus on decentralized care delivery. Additionally, the World Bank recorded U.S. urban population at over 276,000,000 in 2024, supporting widespread deployment of outpatient and urgent care centers. Rising patient preference for faster diagnostics, combined with over 1,000,000 physician office visits annually involving urinalysis, strengthens the case for portable and rapid urine sediment testing technologies at the point of care.
Future Outlook
The USA urine sediment microscopes market is expected to experience steady growth supported by rising automation investments and continued demand for diagnostic efficiency. Advancements in AI-enabled microscopy and digital imaging will likely improve diagnostic accuracy and workflow integration. Regulatory support for advanced in vitro diagnostic technologies is expected to encourage product innovation. Growing focus on preventive diagnostics and chronic disease monitoring will also support testing demand. Expansion of laboratory networks and digital healthcare infrastructure will remain important growth contributors.
Major Players
- Sysmex Corporation
- Siemens Healthineers
- Roche Diagnostics
- Abbott Laboratories
- Beckman Coulter
- ARKRAY Inc
- Mindray Bio Medical Electronics
- HORIBA Medical
- 77 Elektronika
- Menarini Diagnostics
- Bio Rad Laboratories
- Thermo Fisher Scientific
- Danaher Corporation
- Agilent Technologies
- Dirui Industrial
Key Target Audience
- Hospital procurement departments
- Diagnostic laboratory chains
- Medical device distributors
- Healthcare equipment investors
- Private equity healthcare investors
- Venture capital healthcare investors
- Government and regulatory bodies
- Healthcare technology procurement groups
Research Methodology
Step 1: Identification of Key Variables
Primary variables including diagnostic testing demand, laboratory automation adoption, disease prevalence, and healthcare infrastructure investment were identified. Secondary data sources included clinical diagnostics publications and healthcare statistics databases. Market boundaries were defined based on product scope and end user adoption patterns.
Step 2: Market Analysis and Construction
Market structure was constructed using supply side manufacturer data and demand side laboratory procurement trends. Historical equipment sales and healthcare testing volumes were analyzed. Comparative technology benchmarking was also conducted to understand competitive positioning.
Step 3: Hypothesis Validation and Expert Consultation
Industry assumptions were validated through interviews with laboratory professionals and diagnostic equipment specialists. Technology adoption trends were verified using manufacturer product releases and regulatory clearances. Market estimates were tested against clinical testing growth indicators.
Step 4: Research Synthesis and Final Output
All findings were consolidated through triangulation of supply, demand, and technology indicators. Quantitative and qualitative insights were integrated into a structured framework. Final conclusions were derived after consistency checks and validation reviews.
- Executive Summary
- Research Methodology (Definitions, Scope, Industry Assumptions, Market Sizing Approach, Primary & Secondary Research Framework, Data Collection & Verification Protocol, Analytic Models & Forecast Methodology, Limitations & Research Validity Checks)
- Market Definition and Scope
- Value Chain & Stakeholder Ecosystem
- Regulatory / Certification Landscape
- Sector Dynamics Affecting Demand
- Strategic Initiatives & Infrastructure Growth
- Growth Drivers
Rising prevalence of kidney disorders and urinary tract infections in the United States
Increasing laboratory automation adoption to reduce diagnostic turnaround time
Growing demand for accurate microscopic urinalysis in preventive healthcare
Technological advancements in digital imaging and AI based diagnostics
Expansion of diagnostic laboratory chains across the country - Market Challenges
High acquisition and maintenance costs of automated microscopy systems
Shortage of skilled laboratory technicians trained in advanced microscopy
Integration complexity with legacy laboratory information systems
Reimbursement limitations for advanced urinalysis diagnostics
Data management challenges due to large diagnostic imaging volumes - Market Opportunities
Expansion of AI powered automated urine sediment analysis solutions
Growing adoption of point of care urinalysis diagnostics
Partnership opportunities between diagnostic labs and technology providers - Trends
Shift toward fully automated digital urine sediment microscopy
Adoption of AI based particle recognition algorithms
Increasing use of remote diagnostics and telepathology integration
Growth of compact microscopy solutions for small laboratories
Rising focus on workflow integrated urinalysis platforms - Government Regulations & Defense Policy
FDA regulatory approvals governing diagnostic microscopy devices
CLIA compliance requirements for clinical laboratory testing systems
Quality standards defined by the College of American Pathologists - SWOT Analysis
- Stakeholder and Ecosystem Analysis
- Porter’s Five Forces Analysis
- Competition Intensity and Ecosystem Mapping
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025
- By System Type (In Value%)
Automated Urine Sediment Microscopes
Semi-Automated Urine Sediment Microscopes
Manual Laboratory Microscopes for Urinalysis
Digital Imaging Urine Sediment Microscopes
AI Enabled Urine Sediment Analysis Systems - By Platform Type (In Value%)
Standalone Laboratory Microscopy Platforms
Integrated Urinalysis Workstation Platforms
Cloud Connected Diagnostic Microscopy Systems
Benchtop Digital Microscopy Platforms
Modular Microscopy Analysis Platforms - By Fitment Type (In Value%)
Standalone diagnostic microscopy units
Laboratory information system integrated systems
Modular add on microscopy units
Workstation integrated sediment analyzers
Mobile diagnostic cart based systems - By End User Segment (In Value%)
Hospital diagnostic laboratories
Independent diagnostic laboratories
Academic and research institutions
Specialty nephrology clinics
Point of care diagnostic centers - By Procurement Channel (In Value%)
Direct manufacturer procurement
Medical device distributors
Group purchasing organizations
Online medical equipment procurement platforms
Leasing and rental service providers - By Material / Technology (in Value %)
Brightfield optical microscopy technology
Phase contrast microscopy technology
Digital imaging sensor based microscopy
Artificial intelligence image recognition software
Automated slide scanning technology
- Market structure and competitive positioning
- Market share snapshot of major players
- Cross Comparison Parameters (Product Portfolio Breadth, Automation Capability, Imaging Accuracy, AI Integration Level, Pricing Strategy, Distribution Network Strength, After Sales Support, Regulatory Approvals, R&D Investment, Software Integration Capability)
- SWOT Analysis of Key Players
- Pricing & Procurement Analysis
- Key Players
Sysmex Corporation
Beckman Coulter Diagnostics
Roche Diagnostics
Siemens Healthineers
Abbott Laboratories
HORIBA Medical
77 Elektronika Kft
Menarini Diagnostics
Arkray Inc
Dirui Industrial Co Ltd
Mindray Bio Medical Electronics
BioRad Laboratories
Thermo Fisher Scientific
Danaher Corporation
Agilent Technologies
- Hospitals dominate demand due to high patient testing volumes
- Diagnostic laboratory chains drive adoption of high throughput systems
- Research institutions focus on advanced imaging capabilities
- Specialty clinics prefer compact and cost efficient microscopy solutions
- By Market Value, 2020-2025
- By Installed Units, 2020-2025
- By Average System Price, 2020-2025
- By System Complexity Tier, 2020-2025