Market Overview
The USA urine test strips market is valued at approximately USD ~ billion in 2024. This growth is primarily driven by an increased demand for diagnostic solutions, especially in the areas of chronic disease management, home healthcare, and point-of-care testing. The market is influenced by advancements in test strip technologies, including digital and multi-parameter strips that offer enhanced diagnostic capabilities. Furthermore, the growing prevalence of diabetes, urinary tract infections, and kidney disorders, along with rising awareness of self-monitoring health tools, has bolstered the adoption of urine test strips in both clinical and home settings.
The USA remains the dominant market for urine test strips due to its advanced healthcare infrastructure, high prevalence of chronic conditions, and well-established regulatory frameworks. States like California, New York, and Texas lead the market due to their large population base, extensive healthcare networks, and high adoption of point-of-care diagnostic devices. Additionally, urban areas with dense medical facilities, such as Boston and Chicago, also contribute significantly to market growth due to the demand for accurate and timely diagnostic testing solutions in both healthcare settings and consumer markets.
Market Segmentation
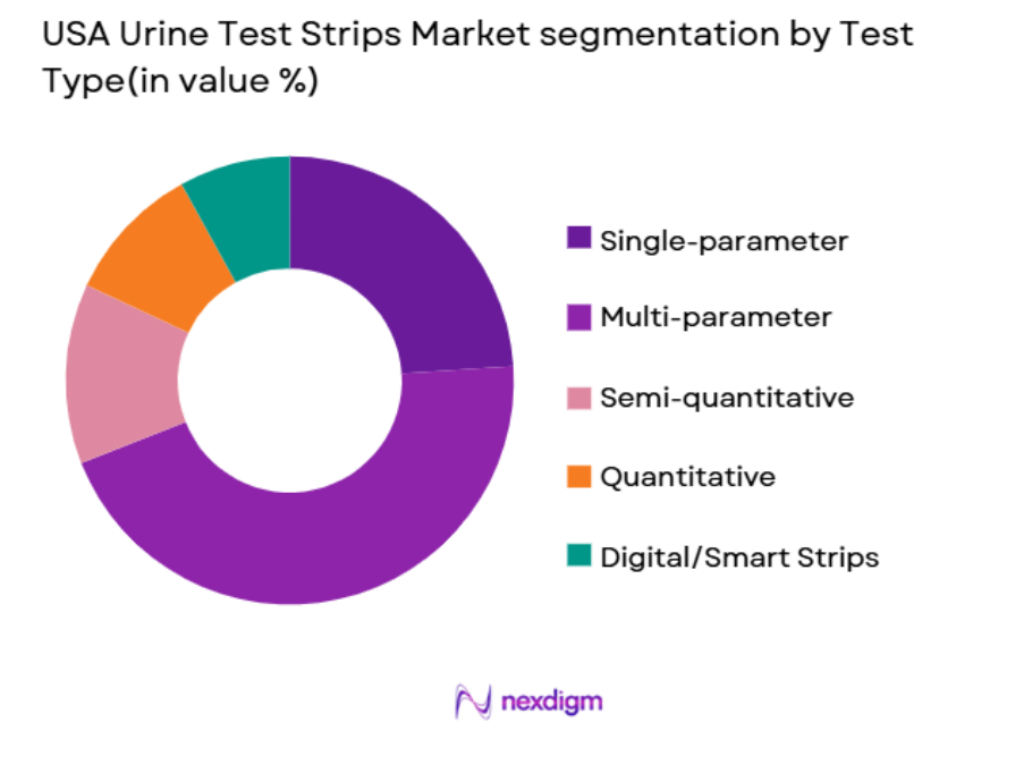
By Test Type
The USA urine test strips market is segmented by test type into single-parameter, multi-parameter, semi-quantitative, quantitative, and digital/smart strips. Among these, multi-parameter test strips hold the dominant share due to their ability to detect multiple health conditions at once, offering convenience for healthcare providers and patients. These strips are particularly favored in clinical settings as they allow for comprehensive assessments of kidney function, glucose levels, and urinary tract infections, all within a single test. The growing preference for home monitoring solutions also enhances the market penetration of multi-parameter test strips, especially with the advancement of digital devices.
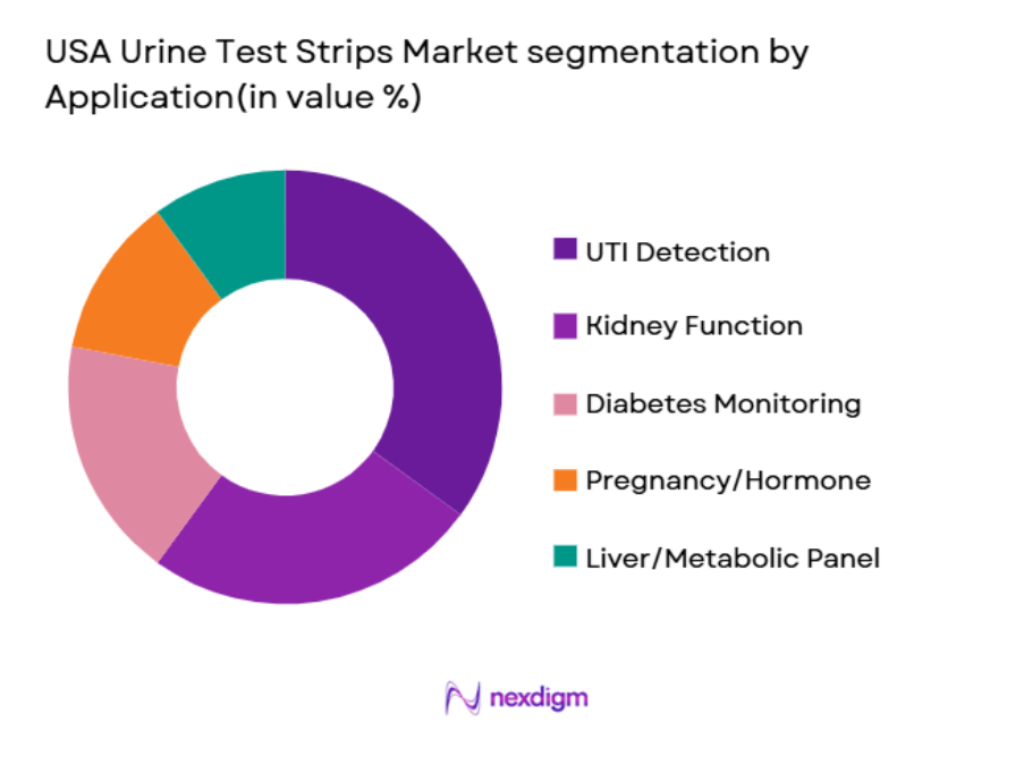
By Application
The market is also segmented by application into urinary tract infections (UTIs), kidney function, diabetes monitoring, pregnancy and hormone screening, and liver/metabolic panels. Among these, UTI detection is the leading application, accounting for the largest market share. The high incidence of UTIs, particularly among women and the elderly, drives this dominance. Additionally, UTIs often require frequent monitoring, making urine test strips a convenient and cost-effective tool for both clinicians and patients. As the demand for home-based diagnostic solutions grows, this segment is expected to continue leading the market in terms of volume and revenue.
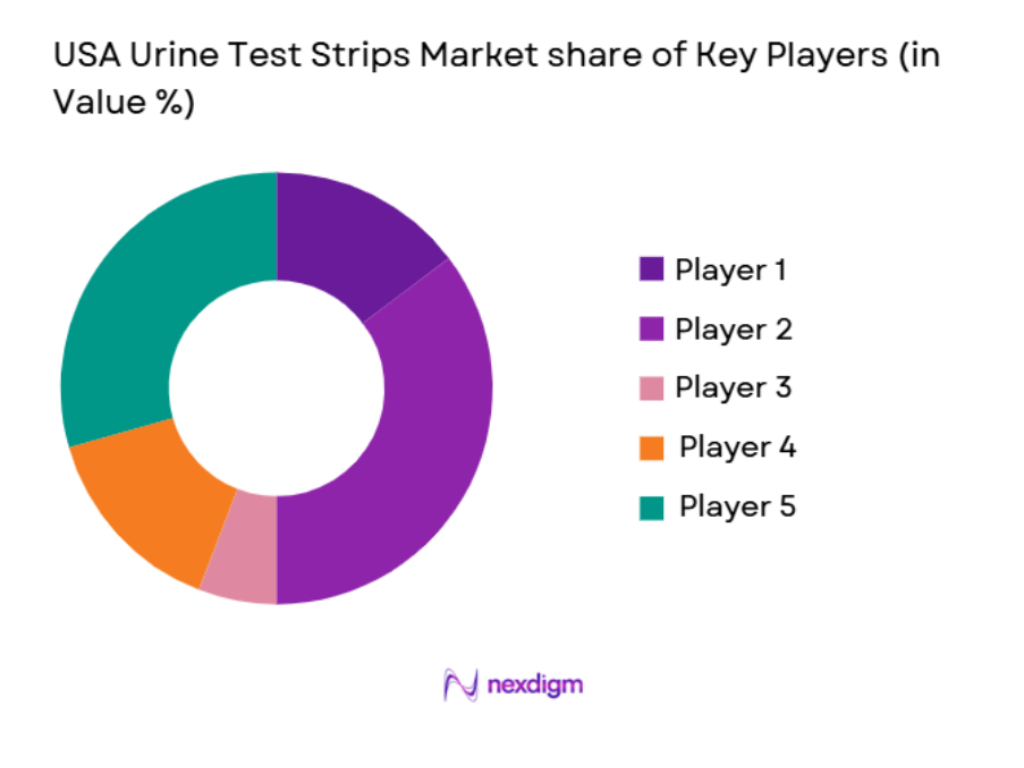
Competitive Landscape
The USA urine test strips market is dominated by a few key players who leverage their technological advancements and robust distribution networks to maintain a competitive edge. Companies such as Roche Diagnostics, Abbott Laboratories, and Siemens Healthineers lead the market due to their strong product portfolios, including both traditional and digital urine test strips. These companies also benefit from well-established relationships with healthcare providers and distribution channels. Additionally, smaller specialized players are gaining traction with innovative products that cater to niche markets, such as home healthcare and telemedicine.
| Company | Establishment Year | Headquarters | Product Portfolio | Market Penetration | Technological Innovations | Distribution Channels |
| Roche Diagnostics | 1896 | Basel, Switzerland | Digital, Multi-parameter Strips | High | Advanced digital urine strips | Hospitals, Clinics, E-commerce |
| Abbott Laboratories | 1888 | Abbott Park, USA | Quantitative, Semi-quantitative | Very High | Integration with mobile health | Pharmacies, Online Retail |
| Siemens Healthineers | 1847 | Erlangen, Germany | Multi-parameter Strips | High | AI-enabled interpretation tools | Healthcare Providers, Labs |
| ARKRAY, Inc. | 1954 | Kyoto, Japan | Semi-quantitative Strips | Moderate | Strip and reader combo products | Clinics, Hospitals |
| ACON Laboratories, Inc. | 1990 | San Diego, USA | Single-parameter Strips | Moderate | Low-cost diagnostics solutions | Retail, E-commerce |
USA Urine Test Strips Market Analysis
Growth Drivers
Rising Incidence of Chronic Diseases (Diabetes, Kidney Disorders)
The rising prevalence of chronic diseases, particularly diabetes and kidney disorders, plays a critical role in driving the demand for urine test strips. In 2022, over 37 million people in the United States were diagnosed with diabetes, and more than 20 million adults suffered from chronic kidney disease (CKD). The U.S. Centers for Disease Control and Prevention (CDC) reports that the incidence of diabetes continues to increase annually, with new diagnoses expected to grow by over 1 million every year. Furthermore, nearly 90% of individuals with CKD remain undiagnosed, making early detection through urine test strips crucial for managing kidney health. These increasing rates of chronic conditions emphasize the growing need for diagnostic tools like urine test strips.
Increased Adoption of Point-of-Care Diagnostics
Point-of-care (POC) diagnostics are becoming increasingly vital, especially in outpatient and home healthcare settings. The U.S. healthcare system is focusing on reducing hospital admissions and increasing outpatient care, which is propelling the growth of POC diagnostic tools. In 2023, the U.S. Department of Health and Human Services (HHS) reported that outpatient visits have grown substantially, with over 100 million visits recorded, showing a shift toward more decentralized healthcare delivery. This shift in healthcare paradigms is leading to a higher adoption of easy-to-use diagnostic tools like urine test strips. Additionally, POC diagnostic tests are gaining importance due to their ability to provide immediate results, reducing the need for laboratory tests.
Market Challenges
Sensitivity/Specificity Limitations
Urine test strips, while convenient, face challenges in terms of their diagnostic accuracy. In 2022, studies indicated that test strips for diabetes monitoring had a sensitivity of approximately 85%, with specificity rates varying between 75% to 90%, depending on the analyte being tested. This limitation can lead to inaccurate readings, impacting the reliability of test results. These performance challenges are particularly critical when used for diagnosing conditions such as kidney disease and infections, where precision is paramount for treatment decisions. Improving the sensitivity and specificity of test strips remains a major area of concern for manufacturers.
False Positive/Negative Risk & Confirmatory Testing Costs
Another challenge for the urine test strips market is the risk of false positives and false negatives, which can lead to unnecessary treatments or missed diagnoses. For instance, a study conducted by the U.S. Food and Drug Administration (FDA) in 2023 revealed that certain urinary test strips for detecting glucose and protein had an error margin of up to 10%, increasing the likelihood of false results. This risk is particularly high in conditions like diabetes and kidney disease, where accurate detection is critical for patient outcomes. Furthermore, confirmatory testing can incur additional costs, adding to the financial burden on healthcare systems and patients alike.
Market Opportunities
Telehealth Integration & Remote Patient Monitoring
The shift toward telehealth and remote patient monitoring offers significant growth opportunities for the urine test strips market. The use of telehealth services in the U.S. skyrocketed in 2022, with over 50 million patients utilizing virtual visits. As a result, the demand for home diagnostic tools, including urine test strips, is increasing as patients seek to manage chronic conditions remotely. In 2023, a report from the U.S. Department of Health and Human Services highlighted that remote monitoring tools are being widely adopted, with more than 75% of healthcare providers offering some form of remote monitoring for chronic disease management. The integration of urine test strips into these systems is expected to grow as healthcare providers continue to adopt digital solutions for monitoring conditions like diabetes and kidney disease.
Advanced Multiplexed Parameter Strips
The advancement of multiplexed parameter urine test strips presents a key opportunity for growth in the diagnostic market. Multiplexed strips are capable of detecting multiple health markers simultaneously, making them more efficient and appealing for both healthcare providers and consumers. These strips are gaining traction due to their ability to offer comprehensive testing for various conditions, such as diabetes, kidney disease, and infections, in a single test. Currently, over 30% of healthcare providers in the U.S. are integrating multiplexed diagnostic technologies into their workflow, as reported by the U.S. Food and Drug Administration in 2023. The shift toward more comprehensive and integrated diagnostic tools is expected to drive growth in the market for these advanced test strips.
Future Outlook
Over the next six years, the USA urine test strips market is poised for significant growth. This growth will be driven by continuous advancements in diagnostic technology, increased adoption of digital and smart strips, and a growing demand for at-home diagnostic tools. The shift towards point-of-care testing, coupled with the increasing prevalence of chronic diseases such as diabetes and kidney disorders, will further stimulate market expansion. Additionally, the rising demand for affordable, easy-to-use diagnostic solutions will fuel innovation in both product design and functionality.
Major Players
- Roche Diagnostics
- Abbott Laboratories
- Siemens Healthineers
- ARKRAY, Inc.
- ACON Laboratories, Inc.
- Bio‑Rad Laboratories
- QuidelOrtho Corporation
- Sysmex Corporation
- Becton, Dickinson and Company
- Nova Biomedical
- Thermo Fisher Scientific
- TaiDoc Technology Corporation
- OraSure Technologies, Inc.
- Medline Industries
- Labcorp
Key Target Audience
- Investments and Venture Capitalist Firms
- Government and Regulatory Bodies (FDA, CMS)
- Diagnostic Equipment Manufacturers
- Healthcare Providers and Hospitals
- Pharmaceutical Companies
- Health Insurance Providers
- Private Healthcare Networks
- E-commerce Retailers for Home Healthcare Products
Research Methodology
Step 1: Identification of Key Variables
The first phase involves identifying key market drivers and barriers within the USA urine test strips market. This is achieved through secondary research using reliable sources such as government publications, industry reports, and academic journals. Key variables such as market trends, growth drivers, and competitive dynamics are mapped out, providing a framework for the subsequent analysis.
Step 2: Market Analysis and Construction
This phase focuses on gathering historical data for the market, including pricing models, revenue statistics, and adoption rates of different urine test strip products. Comprehensive segmentation and trend analysis help in understanding the market dynamics, especially the rise of digital and multi-parameter test strips.
Step 3: Hypothesis Validation and Expert Consultation
Market hypotheses are developed and tested through interviews with industry experts, including clinicians, product developers, and healthcare providers. These consultations validate the initial data and provide deeper insights into operational challenges, technological advancements, and market trends.
Step 4: Research Synthesis and Final Output
In the final phase, we conduct primary research through surveys and direct interactions with leading manufacturers. The data collected from stakeholders in the healthcare and diagnostic sectors allows us to refine the report’s findings, ensuring that the analysis is accurate, current, and relevant to market participants.
- Executive Summary
- Research Methodology (Market Definitions and Scope, Abbreviations, Analytical Framework, Data Sources and Validation Protocols, Statistical Assumptions, Model Inputs, Survey and Field Data, Estimation Boundaries, Triangulation Approach, Limitations and Confidence Intervals)
- Definition and Scope
- Product Taxonomy and Analyte Parameter Classification, (Chemical, Biochemical, Multi‑parameter Panels)
- Clinical Purpose Mapping, (UTI, Kidney Profile, Diabetes, Pregnancy, Liver Profile)
- Healthcare Delivery Pathway, (Hospitals, Clinics, Diagnostic Labs, Home Care)
- Testing Formats, (Dipsticks, Reagent Strips, Semi‑Quantitative vs Quantitative)
Component and Raw Material Ecosystem
- Growth Drivers
Rising Incidence of Chronic Diseases (Diabetes, Kidney Disorders)
Increased Adoption of Point‑of‑Care Diagnostics
Regulatory Approvals & Reimbursement Policies
Digital Health Integration & Connected Diagnostics - Market Challenges (Analytical Specific)
Sensitivity/Specificity Limitations
False Positive/Negative Risk & Confirmatory Testing Costs
Supply Chain Constraints and Raw Material Costs - Market Opportunities
Telehealth Integration & Remote Patient Monitoring
Advanced Multiplexed Parameter Strips
AI/ML for Automated Interpretation - Industry Trends
Shift to Smartphone‑Linked Readouts
Pricing & Reimbursement Dynamics
Adoption in Chronic Disease Management Programs
- Regulatory Environment
FDA Clearance (510(k), LDT Impacts)
CLIA Waiver Adoption Trends
Reimbursement Framework (Medicare/Commercial CPT Codes)
- Porter’s Five Forces
- Value Chain & Diagnostic Pathway Mapping
- By Market Revenue (USD), 2026-2035
- By Unit Volume, 2026-2035
- By Market Penetration Index, 2026-2035
- By Average Selling Price (ASP) Trends, 2026-2035
- By Test Type (In Value %)
Singleparameter
Multiparameter
Semiquantitative
Quantitative
Digital/Smart Strips (Deviceenabled) - By Application (In Value %)
Urinary Tract Infection (UTI) Detection
Kidney Function Assessment
Diabetes Monitoring
Pregnancy & Hormone Screening
Liver/Metabolic Panel Assessment - By End User (In Value %)
Hospitals & Clinical Labs
Diagnostic Chains & Independent Labs
Home/PointofCare Testing
Pharmacy/OTC Retail
Telehealth/Remote Diagnostics - By Distribution Channel (In Value %)
Hospital Procurement
Retail Pharmacies
Ecommerce & Online Channels
Diagnostic Lab Supply Agreements
Direct OEM Contracts - By Geographical Region (USA State Level) (In Value %)
Northeast
Midwest
South
West
- Market Share by Revenue/Volume (Key Companies)
- Cross Comparison Parameters (Company Overview; Product Portfolio Breadth; Parameter Count per SKU; Clinical Accuracy Metrics; Distribution Network Penetration; Reimbursement Support; R&D Expense %; Regulatory Approvals)
Competitor SWOT Profiles
Pricing Matrix (SKU Level Price Stratification)
Go‑to‑Market Strategy Profiles - Major Competitors
Roche Diagnostics
Siemens Healthineers
Abbott Laboratories
Thermo Fisher Scientific
Bayer Diagnostics
ARKRAY, Inc.
ACON Laboratories, Inc.
QuidelOrtho Corporation
Sysmex Corporation
Bio‑Rad Laboratories
Nova Biomedical
BTNX Inc.
TaiDoc Technology Corporation
OraSure Technologies, Inc.
Becton, Dickinson and Company
- Clinical Ordering Patterns (Diagnostic Labs vs Hospitals)
- Consumer Adoption (Home Testing Growth Metrics)
- Reimbursement and Coverage Influence
- Price Elasticity and Demand Sensitivity
- By Market Revenue (USD), 2026-2035
- By Unit Volume, 2026-2035
- By Market Penetration Index, 2026-2035
- By Average Selling Price, 2026-2035